🎬 Video Lesson Available
Watch the full 7-slide video lesson for Nature of Electromagnetic Radiation with AI teacher narration and visual explanations.
01Introduction to Electromagnetic Radiation: The Energy Driving the Universe

“Welcome, future doctors! Imagine the sunlight hitting your study table. Did you know that light isn't just a glow? It is a powerful wave of energy traveling through space! Today, we are decoding the electromagnetic radiation that makes our universe visible and drives every chemical reaction.”
Understanding the nature of electromagnetic radiation is the first step toward mastering atomic structure for NEET. In the mid-19th century, James Clerk Maxwell revolutionized our understanding of light by proposing that it is not merely a beam of illumination but a complex form of energy transmission consisting of oscillating electric and magnetic fields. These fields move perpendicular to each other and to the direction of propagation. This discovery was pivotal because it explained how energy could travel through the vast vacuum of space without requiring a medium like air or water. For a medical aspirant, this isn't just physics; it is the foundation of how light interacts with biological tissues and chemical molecules.
Electromagnetic radiation (EMR) encompasses a massive range of energies, from the low-energy radio waves used in communication to the high-energy gamma rays emitted during nuclear decay. In the context of chemistry, EMR is the 'trigger' for many transformations. Whether it is the photosynthesis occurring in a leaf or the breaking of a chemical bond in a laboratory flask, electromagnetic radiation provides the necessary energy to bridge the gap between reactants and products. This section sets the stage for understanding how electrons within an atom respond to these external energy pulses.
One of the most striking features of EMR is its universal speed. In a vacuum, every form of electromagnetic radiation—be it visible light, X-rays, or heat-carrying infrared rays—travels at a staggering speed of approximately 300,000 kilometers per second. This constant, denoted as 'c', serves as a cosmic speed limit and a crucial constant in every chemical calculation involving light. As we dive deeper, we will see how this energy is quantified and how it dictates the behavior of the subatomic world.
Quick Revision Points
- Electromagnetic radiation consists of oscillating electric and magnetic fields perpendicular to each other.
- Maxwell's theory established that EMR does not require a medium to propagate through space.
- Radiation is a primary driver of chemical transformations by transferring energy to matter.
- Visible light is only a tiny fraction of the broader electromagnetic spectrum.
NEET Exam Angle
- Conceptual questions often ask about the nature of the fields in EMR (they are mutually perpendicular).
- Understanding that all EMR travels at the same speed (c) in a vacuum is a common point of confusion in multiple-choice questions.
- Connect this to Thermodynamics, as EMR is a key mode of energy transfer (radiation).
02The Anatomy of a Wave: Wavelength, Amplitude, and Oscillation

“Think of a ripple in a calm pond after dropping a pebble. Electromagnetic waves oscillate like that! We have a peak and a trough. The distance between two consecutive peaks is the 'wavelength', and the height of the wave is its 'amplitude'. Simple, right?”
To describe any wave, we must first understand its physical architecture. Imagine looking at a cross-section of the ocean; you see peaks and valleys. In wave mechanics, the peak of a wave is called a crest, and the lowest point is known as a trough. The most fundamental characteristic of a wave is its wavelength, represented by the Greek letter lambda (λ). The wavelength is defined as the distance between two consecutive crests or two consecutive troughs. In the subatomic world, these distances can be as large as kilometers or as small as the diameter of an atomic nucleus.
Another critical parameter is the amplitude. If you think of a wave as a ripple, the amplitude is the height of the crest or the depth of the trough measured from the central line of propagation. In the context of light, amplitude determines the intensity or brightness of the radiation. A wave with a larger amplitude carries more 'punch' or intensity, though not necessarily more energy per individual particle (as we will learn later in Planck’s theory). This distinction between intensity and energy is a frequent pitfall for students during the NEET exam.
Because wavelengths vary so drastically across the spectrum, we use several units to measure them. For visible light, we often use nanometers (nm) or Angstroms (Å). One nanometer is one-billionth of a meter ($10^{-9}$ m), and one Angstrom is $10^{-10}$ m. Mastering these conversions is non-negotiable for solving numerical problems. If you cannot fluently switch between meters, nanometers, and centimeters, you will likely lose marks on simple calculation-based questions.
Quick Revision Points
- Crest and Trough: The highest and lowest points of a wave oscillation.
- Wavelength (λ): The spatial distance between identical points on consecutive waves.
- Amplitude (A): The maximum displacement from the equilibrium position; determines intensity.
- Units: 1 nm = $10^{-9}$ m; 1 Å = $10^{-10}$ m; 1 pm = $10^{-12}$ m.
NEET Exam Angle
- Be prepared for unit conversion traps: Questions may give λ in nm but require meters for use with the speed of light in SI units.
- Relationship between amplitude and intensity: Intensity is proportional to the square of the amplitude ($I ∝ A^2$).
- Practice drawing wave diagrams to visualize how wavelength and frequency change relatively.
03Frequency and the Hertz: Quantifying Wave Periodicity
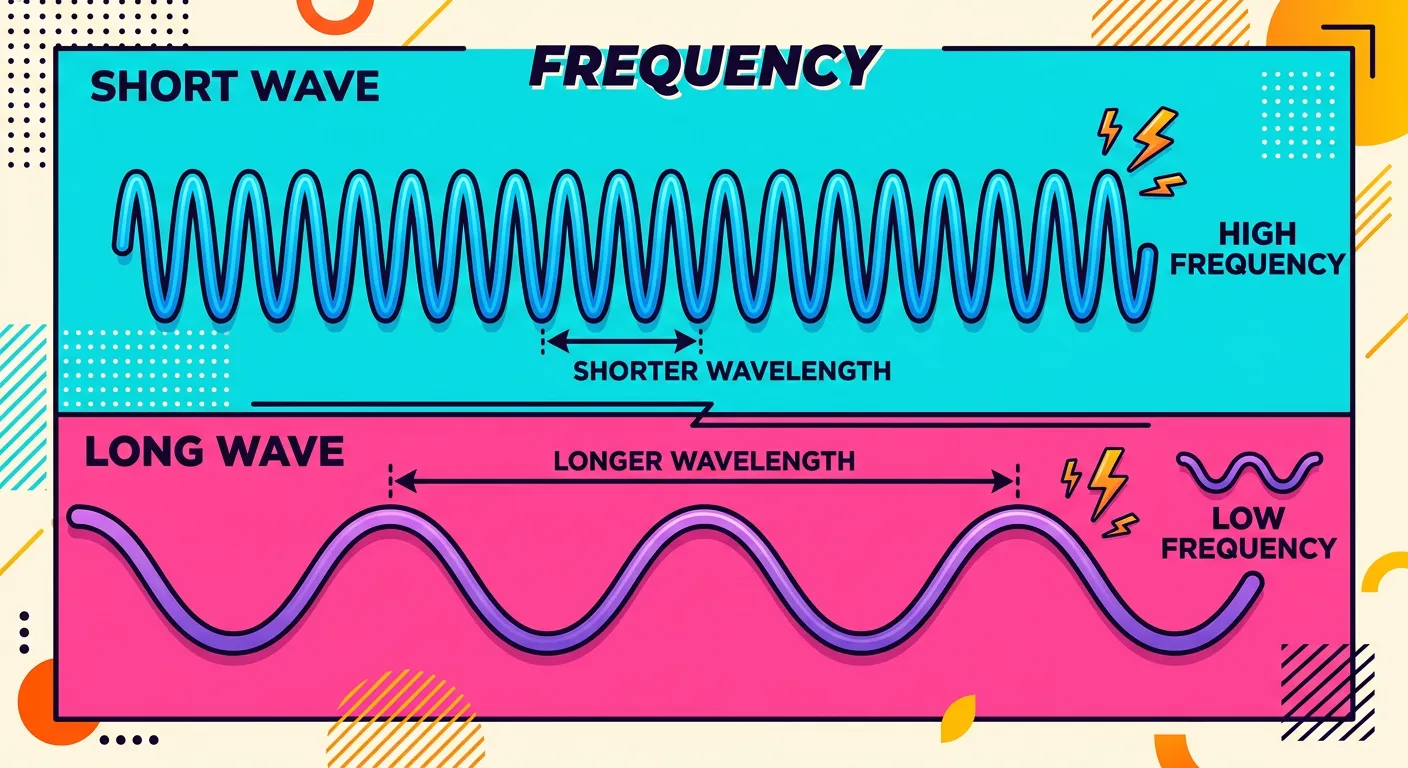
“Frequency is just how many waves pass a point in one second. Imagine a fast-tapping drummer versus a slow one. High frequency means more taps, or waves, per second. Remember, it is measured in Hertz. If the wave is faster, the frequency is definitely higher!”
While wavelength describes the spatial size of a wave, frequency describes its temporal rhythm. Frequency, represented by the Greek letter nu (ν), is the number of wave cycles that pass a fixed point in one second. Think of it as the 'heartbeat' of the radiation. A high-frequency wave is one that is vibrating very rapidly, sending many cycles through space every second. Conversely, a low-frequency wave oscillates slowly. Frequency is the primary factor that determines the type of radiation we are dealing with—whether it is 'soft' radio waves or 'hard' X-rays.
The International System (SI) unit for frequency is the Hertz (Hz), named after Heinrich Hertz. One Hertz is defined as one cycle per second ($s^{-1}$). In many chemical problems, you will see frequency expressed simply as $s^{-1}$, which is functionally identical to Hz. Another related term is the time period (T), which is the time taken for one complete wave cycle to pass a point. Mathematically, frequency and the time period are reciprocals of each other ($ν = 1/T$).
In the NEET syllabus, frequency is often the bridge between the wave properties and the energy properties of light. When an electron in an atom absorbs energy, it is often expressed in terms of the frequency of the radiation required to excite it. High-frequency radiation carries more energy, making it capable of ionizing atoms or damaging DNA—a concept vital for medical students to understand when studying radiology or oncology. Understanding that frequency is a count of 'events per unit time' helps demystify the complex equations that follow.
Quick Revision Points
- Frequency (ν): The number of complete wave cycles passing a point per second.
- Unit: Hertz (Hz) or $s^{-1}$ (cycles per second).
- Time Period (T): The duration of one single wave cycle ($T = 1/ν$).
- Significance: Frequency is a characteristic property of the source and does not change when light moves between media.
NEET Exam Angle
- Calculate frequency from the time period: If $T = 2.0 × 10^{-10}$ s, then $ν = 5.0 × 10^9$ Hz.
- Note that in a medium other than vacuum, velocity changes, but frequency remains constant.
- Expect questions comparing the frequencies of different colors in the visible spectrum (Violet has the highest frequency).
04The Inverse Proportionality Law: Wavelength vs. Frequency
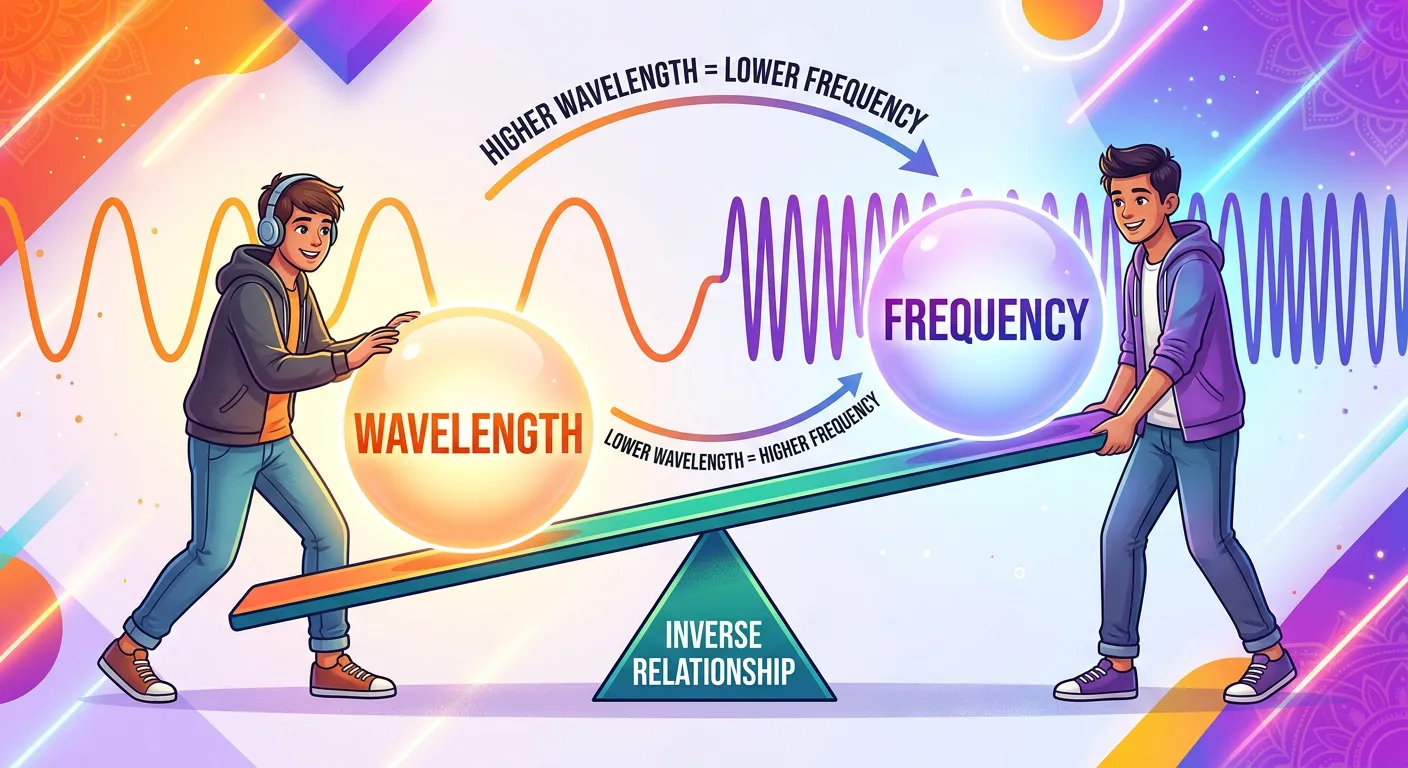
“Here is the golden rule for your NEET exams: wavelength and frequency are inversely proportional. Long, lazy waves have low frequency, while short, energetic waves have high frequency. Just like a long, slow cricket swing versus a short, sharp flick of the wrist!”
One of the most fundamental mathematical relationships in chemistry is the link between wavelength (λ) and frequency (ν). Since all electromagnetic waves travel at a constant speed (c) in a vacuum, their spatial length and temporal frequency must balance each other out. This is expressed by the elegant equation: $c = λ ν$. Because 'c' is a constant ($3.0 × 10^8$ m/s), if the wavelength increases, the frequency must decrease to keep the product the same. This is known as an inverse proportionality.
Visualizing this is easy: if you have very long waves (large λ), fewer of them can pass a point in a second. If you have tiny, short waves (small λ), many more of them will zip past that same point in the same amount of time. This relationship is why radio waves (long λ) have low frequencies, while Gamma rays (short λ) have incredibly high frequencies. In NEET, you are frequently asked to compare two waves. If Wave A has twice the wavelength of Wave B, you must immediately recognize that Wave A has half the frequency of Wave B.
This inverse relationship also has profound energy implications. As we will see in Planck's Law, energy is directly proportional to frequency. Therefore, energy is inversely proportional to wavelength. Short-wavelength radiation is high-energy radiation. This explains why Ultraviolet rays (short λ) can cause sunburn, while Infrared rays (long λ) only make us feel warm. Master this concept, and you will find the hydrogen spectrum and Bohr’s model much easier to comprehend.
| Radiation Type | Wavelength (λ) | Frequency (ν) | Relative Energy |
|---|---|---|---|
| Radio Waves | Longest | Lowest | Lowest |
| Visible Light | Intermediate | Intermediate | Intermediate |
| Gamma Rays | Shortest | Highest | Highest |
Quick Revision Points
- Formula: $c = λν$, where $c ≈ 3 × 10^8$ m/s.
- Inverse Relationship: $λ ∝ 1/ν$. Doubling one halves the other.
- Energy Link: Short wavelengths = High Frequency = High Energy.
- Consistency: This law applies to all electromagnetic radiation in a vacuum.
NEET Exam Angle
- Direct numericals: Given λ, find ν. Use $3 × 10^8$ as the default value for c unless stated otherwise.
- Comparison questions: "Which has higher energy: $400$ nm light or $700$ nm light?" (Answer: 400 nm because shorter λ means higher ν).
- Recall the 'cricket swing' hook: Short, sharp movements are high frequency and high energy.
05The Electromagnetic Spectrum: Mapping the Energy Family Tree
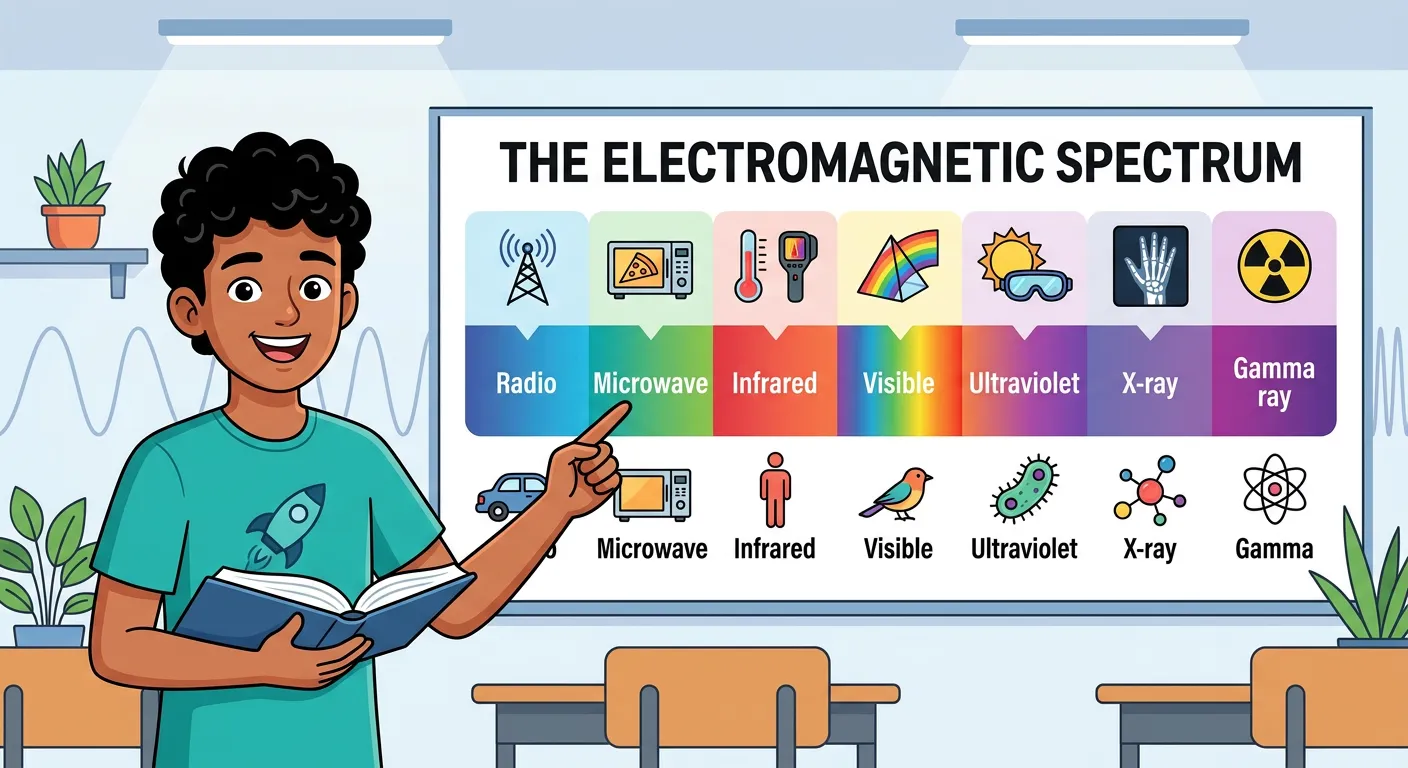
“The electromagnetic spectrum is our complete family tree! From giant Radio waves to tiny, lethal Gamma rays, they all travel at the speed of light—three lakh kilometers per second. It is the same speed for every single one of them in a vacuum!”
The electromagnetic spectrum is the complete range of all possible frequencies of electromagnetic radiation. It is like a giant family tree where every member, despite having different energies and properties, shares the same DNA: the speed of light. The spectrum is typically organized from the longest wavelengths (Radio waves) to the shortest (Gamma rays). For the NEET exam, you must memorize the general order of these regions. A common mnemonic to remember the spectrum in increasing order of frequency (or decreasing wavelength) is: "Raging Martians Invaded Venus Using X-ray Guns" (Radio, Microwave, Infrared, Visible, Ultraviolet, X-ray, Gamma).
The visible region is only a sliver of the total spectrum, ranging roughly from 400 nm (violet) to 750 nm (red). This is the only part our eyes can detect. The sequence of colors within this region is the famous VIBGYOR (Violet, Indigo, Blue, Green, Yellow, Orange, Red). It is essential to remember that Violet has the shortest wavelength (~400 nm) and the highest energy, while Red has the longest wavelength (~750 nm) and the lowest energy.
Beyond the visible range, different radiations require different detection methods. Infrared radiation is detected as heat, while X-rays require specialized photographic plates. In chemistry, we use these different regions to probe the structure of matter. For example, Infrared (IR) spectroscopy helps us identify functional groups in organic molecules by observing how they vibrate when they absorb specific IR frequencies. The spectrum is not just a list; it is a diagnostic tool for both doctors and chemists.
| Region | Approx. Wavelength Range | Common Use/Observation |
|---|---|---|
| Radio | $> 0.1$ m | Broadcasting, MRI |
| Microwave | $1$ mm to $0.1$ m | Radar, Cooking |
| Infrared | $750$ nm to $1$ mm | Heat sensors, Molecular vibrations |
| Visible | $400$ nm to $750$ nm | Human vision, Photosynthesis |
| Ultraviolet | $10$ nm to $400$ nm | Sterilization, Sunburns |
| X-ray | $0.01$ nm to $10$ nm | Medical imaging |
| Gamma | $< 0.01$ nm | Cancer treatment (radiotherapy) |
Quick Revision Points
- Sequence: Radio > Microwave > IR > Visible > UV > X-ray > Gamma (Wavelength order).
- Visible range: ~400 nm (Violet) to ~750 nm (Red).
- Speed: All EM waves travel at $c = 3.0 × 10^8$ m/s in a vacuum.
- Energy: Increases as we move toward Gamma rays.
NEET Exam Angle
- Ordering questions: You may be given four types of radiation and asked to arrange them by increasing frequency.
- Atmospheric connection: UV is absorbed by the Ozone layer; IR is trapped by greenhouse gases.
- VIBGYOR knowledge: Knowing that Red has the lowest frequency is vital for solving problems on the photoelectric effect.
06Wave-Particle Duality: The Concept of Photons and Quanta

“Is light a wave or a particle? It is both! This is the 'Wave-Particle Duality'. Think of it like a cricket ball; it travels as a wave through the air but hits the bat as a single, solid object. In physics, we call these energy packets 'photons'.”
By the end of the 19th century, scientists believed the wave theory of light was complete. However, certain phenomena—like black body radiation and the photoelectric effect—could not be explained if light was only a continuous wave. In 1900, Max Planck proposed a radical idea: energy is not emitted or absorbed continuously but in discrete, small 'packets' or bundles. He called these packets quanta (singular: quantum). Albert Einstein later applied this to light, proposing that light behaves like a stream of particles called photons.
This gave birth to the concept of Wave-Particle Duality. It suggests that light has a dual nature. In phenomena like interference and diffraction, light acts as a wave. In interactions with matter, such as when light hits a metal surface and ejects an electron (photoelectric effect), it acts as a particle. Think of it like a stream of water. From a distance, it looks like a continuous flow (wave), but if you look closely enough, it is made of individual droplets (particles).
For NEET aspirants, the 'particle' nature is where the math becomes critical. Each photon carries a specific amount of energy that depends solely on its frequency. This explains why some types of light can trigger chemical reactions while others cannot, regardless of how 'bright' or intense the light is. If the individual photons don't have enough energy (threshold frequency) to break a bond or move an electron, no amount of intense light will make the reaction happen. This is the cornerstone of modern quantum chemistry.
| Property | Wave Theory | Particle Theory (Photon) |
|---|---|---|
| Propagation | Continuous oscillation in fields | Discrete energy packets |
| Energy | Proportional to Intensity ($A^2$) | Proportional to Frequency ($ν$) |
| Phenomena Explained | Interference, Diffraction | Photoelectric Effect, Black Body Radiation |
| Analogy | A continuous ocean wave | A stream of bullets/baseballs |
Quick Revision Points
- Quantum: The smallest discrete unit of energy.
- Photon: A quantum of electromagnetic radiation (light particle).
- Wave-Particle Duality: Light exhibits both wave-like and particle-like properties.
- Failure of Wave Theory: Could not explain the Photoelectric Effect or Black Body Radiation.
NEET Exam Angle
- Definition of a photon: A massless packet of energy traveling at speed 'c'.
- Conceptual shift: Realize that increasing intensity means more photons, but increasing frequency means more energy per photon.
- Application: Connect this to the 'Dual Nature of Matter' in Physics.
07Mastering the Math: Planck’s Equation and NEET Problem Solving

“To ace your NEET paper, memorize these two magic equations: c equals wavelength times frequency, and energy equals Planck's constant times frequency. Keep these in your pocket, and you'll solve any radiation problem with ease. Great work today, future healers. Keep shining bright!”
The final piece of the puzzle is calculating the actual energy of these photons. Max Planck provided the mathematical tool for this: $E = hν$. In this equation, $E$ is the energy of a single photon, $ν$ is the frequency, and $h$ is Planck's constant. The value of $h$ is approximately $6.626 × 10^{-34}$ Joule-seconds (J·s). Because we know that $ν = c/λ$, we can also write the energy equation as $E = hc/λ$. This second version is extremely popular in NEET exams because wavelength is usually easier to measure than frequency.
When solving these problems, precision is everything. Planck’s constant is an incredibly small number, and the speed of light is very large. When you multiply them ($h × c$), you get roughly $1.989 × 10^{-25}$ J·m. A useful shortcut for NEET (where time is of the essence) is to remember the combined constant in terms of electron-volts (eV) and nanometers. The energy of a photon in eV is roughly $1240 / λ$ (where $λ$ is in nm). This can save you minutes of tedious power-of-ten calculations.
Always ensure your units are consistent. If you use $c$ in $m/s$, your wavelength must be in meters. If the question asks for the energy of a 'mole' of photons, you must multiply the energy of one photon by Avogadro’s number ($N_A = 6.022 × 10^{23} mol^{-1}$). This total energy is often called an 'Einstein' of radiation. Mastery of these two formulas—$c = λν$ and $E = hν$—guarantees success in the Atomic Structure unit.
| Variable | Symbol | Standard Value (SI Units) |
|---|---|---|
| Planck’s Constant | $h$ | $6.626 × 10^{-34}$ J·s |
| Speed of Light | $c$ | $3.0 × 10^8$ m/s |
| Avogadro’s Number | $N_A$ | $6.022 × 10^{23}$ $mol^{-1}$ |
| Energy of 1 Photon | $E$ | $hν$ or $hc/λ$ (Joules) |
Quick Revision Points
- Fundamental Formula: $E = hν = hc/λ$.
- Constant h: $6.626 × 10^{-34}$ J·s.
- Total Energy: For 'n' photons, $E_{total} = n × hν$.
- Energy per mole: Multiply single photon energy by Avogadro's number.
NEET Exam Angle
- Numerical Trend: Calculating energy when λ is given in Angstroms. Remember: $10 Å = 1 nm = 10^{-9} m$.
- Ratio problems: "If the frequency of radiation is doubled, the energy of the photon is..." (Answer: Doubled).
- Units: Look out for answers in ergs ($1 J = 10^7$ ergs) or eV ($1 eV = 1.602 × 10^{-19}$ J).
Recommended Reading
Explore related Biology topics to build deeper chapter connections for NEET.
- Quantum Mechanical Model · Topic 2.12
- Atomic Orbitals · Topic 2.13
- Significance of Quantum Numbers · Topic 2.16
- Photoelectric Effect · Topic 2.2
- Bohr Model · Topic 2.4
- de Broglie's Relationship · Topic 2.9
- Jump to Key Terms (Quick Revision)
- Review Common NEET Mistakes
- Read Topic FAQs
- Check PYQ Pattern Notes
- Practice NEET MCQs
- Solve NEET PYQs
📚 Key Terms
⚠️ Common NEET Mistakes
- 1Using the wrong units: Forgetting to convert wavelength from nm or Å to meters before using $c = 3.0 × 10^8$ m/s.
- 2Confusing intensity with energy: Assuming a brighter light (higher intensity/amplitude) has higher energy per photon (it doesn't; energy depends only on frequency).
- 3Inverting the proportionality: Thinking that longer wavelengths have higher energy, when they actually have lower energy.
- 4Ignoring Avogadro's Number: Calculating the energy for only one photon when the question asks for the energy of a mole of photons.
- 5Confusing 'nu' (ν) with 'v': Mistaking the Greek symbol for frequency with the symbol for velocity in physics equations.
📝 NEET PYQ Pattern
In NEET 2018–2024, questions from this topic primarily focus on numerical applications of E = hc/λ and E = hν. There is a frequent trend of asking students to compare the energies or frequencies of different regions of the EM spectrum, such as comparing Infrared to Ultraviolet.
❓ Frequently Asked Questions
What is the relationship between frequency and wavelength in a vacuum?
In a vacuum, frequency (ν) and wavelength (λ) are inversely proportional. Their product always equals the constant speed of light (c), expressed by the formula c = λν. This means as wavelength increases, frequency must decrease.
How does the energy of a photon change if its frequency is doubled?
According to Planck’s equation (E = hν), the energy of a photon is directly proportional to its frequency. Therefore, if the frequency is doubled, the energy of the photon also doubles.
Why do all electromagnetic waves travel at the same speed in a vacuum?
All electromagnetic waves are composed of oscillating electric and magnetic fields that propagate through space without a medium. In a vacuum, there are no particles to interfere with or slow down these oscillations, allowing all frequencies to travel at the universal constant speed 'c' (3.0 x 10⁸ m/s).
What is the value and significance of Planck’s constant in NEET chemistry?
The value of Planck’s constant (h) is 6.626 x 10⁻³⁴ J·s. It is significant because it represents the proportionality constant between the energy of a photon and its frequency, effectively linking the wave-like and particle-like properties of light.
Which part of the electromagnetic spectrum has the highest ionizing power?
Gamma rays have the highest ionizing power. This is because they have the shortest wavelengths and highest frequencies, which corresponds to the highest energy per photon, allowing them to easily remove electrons from atoms.
How do you convert wavelength from nanometers to meters for calculation?
To convert nanometers (nm) to meters (m), multiply the value by 10⁻⁹. For example, 500 nm becomes 500 x 10⁻⁹ m or 5.0 x 10⁻⁷ m. This conversion is essential when using the speed of light in SI units (m/s).
Written By
NEET Content Strategist & Biology Expert
Sangita Kumari is a NEET educator and content strategist with over 6 years of experience teaching Biology, Chemistry, and Physics to Class 11 and 12 aspirants. She helps bridge the gap between traditional NCERT preparation and modern AI-powered learning. Her content is trusted by thousands of NEET aspirants across India.