🎬 Video Lesson Available
Watch the full 7-slide video lesson for Dalton's Atomic Theory with AI teacher narration and visual explanations.
01The Dawn of Modern Chemistry: Dalton's Revolutionary Idea

“Welcome, future NEET stars! Ever wondered what everything is made of? Imagine your favorite LEGO set. Just like you build big structures with tiny bricks, John Dalton proposed that all matter is made of tiny, indivisible building blocks called atoms. Let's dive into his revolutionary theory!”
Imagine the scientific landscape of 1808. Chemistry was essentially a collection of fragmented observations without a central, unifying narrative. While Alchemists had spent centuries mixing substances, they lacked a logical 'why' behind their results. John Dalton, a visionary British schoolteacher and polymath, changed the course of history by proposing his atomic theory. While the concept of 'atoms' dated back to ancient Greek philosophers like Democritus and Leucippus, these were purely speculative ideas. It was Dalton who transitioned these ideas from the realm of abstract philosophy to a rigorous, evidence-based scientific framework. He provided the first logical explanation for why matter behaves the way it does during chemical transformations. By treating atoms as the fundamental, indestructible building blocks of the universe, he unified the existing laws of chemical combination into one cohesive and predictive theory.
For a NEET aspirant, understanding Dalton's starting point is crucial because it sets the stage for everything that follows in stoichiometry, thermodynamics, and the structure of the atom. Dalton viewed the atom as the smallest unit of an element that retains its unique chemical identity. He didn't have access to the sophisticated electron microscopes or particle accelerators we have today; instead, he used pure logic and the data from the Law of Definite Proportions to deduce that matter must be discrete (grainy) rather than continuous (fluid-like). This 'LEGO block' approach to the universe is the foundation of the molecular world you study today. In this section, we'll explore how this theory acted as the 'grammar' of chemical science, providing the vocabulary for everything from simple reactions to complex organic synthesis.
| Historical Context | Contribution |
|---|---|
| Ancient Philosophy | Atoms as a theoretical concept without proof or measurement. |
| Antoine Lavoisier | Established the Law of Conservation of Mass through combustion. |
| Joseph Proust | Established the Law of Definite Proportions in compounds. |
| John Dalton | Synthesized these laws into a single, cohesive Atomic Theory. |
Quick Revision Points
- Dalton published his findings in 'A New System of Chemical Philosophy' in 1808.
- His theory provided a theoretical justification for experimental laws observed by his predecessors.
- He shifted the focus of chemistry from qualitative descriptions to quantitative measurements.
- Dalton imagined atoms as solid, hard, spherical particles that couldn't be further divided.
- This theory is the cornerstone of the Unit: 'Some Basic Concepts in Chemistry'.
- He was the first to realize that chemical reactions are just the rearrangement of these units.
NEET Exam Angle
- Focus on the historical timeline: Dalton's work followed the laws of Lavoisier and Proust.
- Question Tip: If a question asks which theory first explained the laws of chemical combination, the answer is Dalton's Atomic Theory.
- Remember that while his model was simple, it was the first to assign relative weights to atoms, which is the precursor to the Modern Atomic Mass unit.
02Postulate One: The Indivisibility of Atoms

“Dalton's first pillar: Atoms are indivisible. Think of them like the smallest, hardest stone in your collection. You can't break it further, cut it, or crush it! This was the starting point of understanding chemistry—the idea that atoms are the ultimate, solid foundation of our universe.”
The first pillar of Dalton's atomic theory states that all matter is composed of extremely small, indivisible particles called atoms. The word 'atom' comes from the Greek word 'atomos', which literally translates to 'uncuttable' or 'that which cannot be divided'. In Dalton's view, you could keep cutting a piece of gold into smaller and smaller bits, but you wouldn't be able to do this infinitely. Eventually, you would reach a point where you reached the 'ultimate' particle that couldn't be cut anymore. That final, indestructible particle was the atom. He envisioned them as hard, solid spheres, much like miniature billiard balls that could bounce around and collide but never be crushed, broken, or pierced into smaller pieces. This was a radical departure from the 'continuous' view of matter where substance was thought to be like a smooth fluid.
From a modern perspective, we know that atoms are not actually indivisible; they contain subatomic particles like protons, neutrons, and electrons. However, it is vital for your NEET preparation to understand that in the context of a chemical reaction, the atom remains the smallest unit of exchange. Dalton's genius lay in recognizing that while you can rearrange atoms to form new substances, the atoms themselves do not break apart during ordinary chemical processes like burning wood or rusting iron. This postulate provided the first clear definition of what an element actually is: a substance made entirely of these ultimate, indivisible units. Even though we now know about nuclear fission (which splits atoms), Dalton’s rule still holds true for the vast majority of chemical interactions you will study in your Class 11 and 12 syllabus.
Quick Revision Points
- Atoms are the basic units of matter that cannot be created, divided, or destroyed according to Dalton.
- This postulate directly supports the idea that matter has a granular, not continuous, nature.
- Dalton's 'billiard ball' model implies atoms have no internal structure or empty space.
- In chemical reactions, atoms are the discrete units that participate in bonding and separation.
- Indivisibility was a core assumption that simplified the calculation of atomic weights during the 19th century.
- This postulate effectively linked the microscopic world of particles to the macroscopic world of mass.
NEET Exam Angle
- Be extremely careful with 'True/False' statements in MCQs: Dalton's postulate of indivisibility is now considered incorrect due to the discovery of subatomic particles.
- Application: If a problem mentions 'Daltonian atoms', assume they are spherical and indivisible for the sake of the problem's logic.
- This postulate is the reason why Dalton's theory failed to explain things like isotopes, radioactivity, and the existence of subatomic particles later on in history.
03The Identity of Elements: Mass and Properties
“Secondly, atoms of a specific element are identical in mass and properties. Think of it like a packet of 5-star chocolates; every single piece tastes and weighs exactly the same. Gold atoms are always gold, and oxygen atoms are always oxygen—consistent and reliable.”
Dalton’s second major postulate focuses on the consistency and identity of chemical elements. He stated that all atoms of a given element are identical in mass and chemical properties, while atoms of different elements differ significantly in their mass and properties. This was a massive leap forward for the periodic classification of matter. Think about it this way: if you have a block of pure silver, every single silver atom in that block is exactly the same as every other silver atom in the universe. They all weigh exactly the same and react in the exact same way. This consistency is what allows us to have a Modern Periodic Table today where each element has a distinct, predictable identity based on its atomic nature.
This postulate is what makes chemical formulas like $H_2O$ or $NaCl$ meaningful and constant. If oxygen atoms varied in mass randomly, we could never calculate a reliable molecular weight for water. Dalton used this principle to create the first-ever table of relative atomic weights. Although he made some experimental errors—for instance, he mistakenly thought hydrogen's weight was 1 and oxygen's was 7 (due to measuring ratios incorrectly)—the underlying logic was incredibly sound. He realized that if atoms of an element are identical, we can assign a 'characteristic weight' to that element. This concept is a foundational pillar for stoichiometry and mole concepts in NEET Chemistry. It means that the chemical behavior of an element is an inherent property of its atoms, which are all 'cast from the same mold'.
| Comparison | Atoms of Same Element | Atoms of Different Elements |
|---|---|---|
| Mass | Identical (according to Dalton) | Different |
| Chemical Properties | Identical | Different |
| Size/Volume | Identical | Distinct to that element |
| Reactivity | Identical | Unique to that element's nature |
Quick Revision Points
- Every atom of Oxygen is exactly like every other atom of Oxygen in mass and behavior.
- An atom of Gold is fundamentally different from an atom of Silver because their individual masses differ.
- This postulate allows for the practical calculation of 'Atomic Mass'.
- It explains why elements have specific, predictable chemical behaviors regardless of their source.
- Dalton used hydrogen as the standard reference for comparing atomic weights because it was the lightest.
NEET Exam Angle
- Critical Exception: Isotopes (atoms of the same element with different masses) prove this postulate partially wrong in modern science.
- NEET often asks which postulate was challenged by the discovery of isotopes—it is this specific one regarding identical mass.
- Remember: Chemical properties are mostly determined by valence electrons, but Dalton linked them primarily to the inherent 'nature' of the atom's mass.
04Compound Formation: Simple Whole-Number Ratios
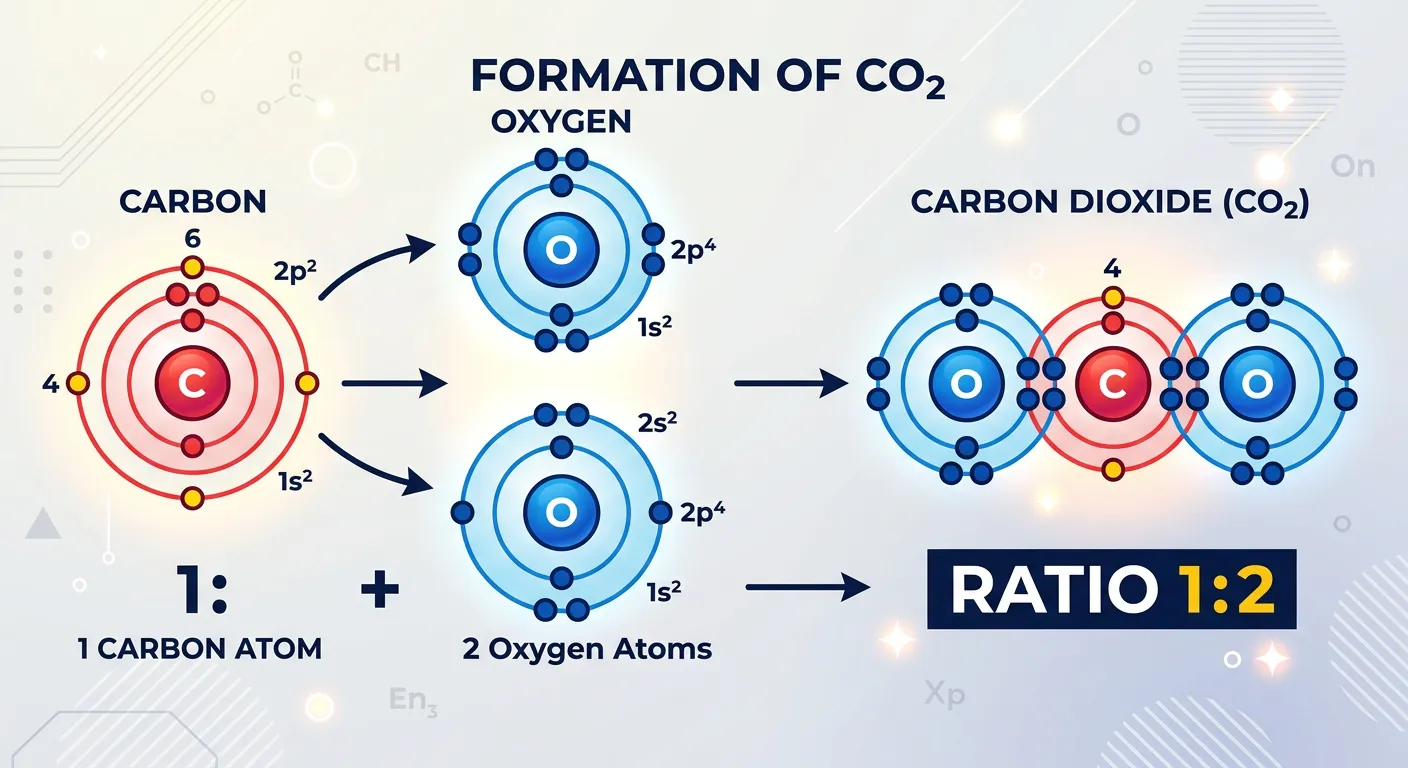
“Atoms combine in simple whole-number ratios to form compounds. It’s like a recipe for tea: you need a specific number of cups of water to sugar. Atoms follow strict, simple math rules. If you change the ratio, you change the entire chemical recipe!”
One of the most mathematically satisfying and elegant parts of Dalton's theory is how atoms combine to form compounds. Dalton postulated that compounds are formed when atoms of different elements combine in simple, fixed, whole-number ratios. In Dalton's universe, you won't find half an atom of Carbon joining with 1.3 atoms of Oxygen. It’s always a clean integer ratio like 1:1 (to form Carbon Monoxide) or 1:2 (to form Carbon Dioxide). This principle provides the physical basis for both the Law of Definite Proportions and the Law of Multiple Proportions, which are key topics in your Class 11 Chemistry syllabus.
Think of this like a digital system rather than an analog one. Chemistry works in 'clicks' or discrete 'steps'. When you react Hydrogen and Oxygen to form water, the ratio is always 2 atoms of Hydrogen to 1 atom of Oxygen. This 'recipe' is universal across the cosmos. Whether the water comes from a tap in Delhi, a moon of Jupiter, or a glacier in Antarctica, the atomic ratio remains exactly $2:1$. For NEET students, this is the origin of the empirical formula and molecular formula concepts. If you understand that atoms combine in integers, stoichiometry becomes a game of simple arithmetic rather than complex calculus. Dalton’s rule explains why formulas like $C_{12}H_{22}O_{11}$ exist and why we never see fractional subscripts in a standard molecular formula. It highlights the 'quantized' nature of chemical bonding long before quantum mechanics was even a thought.
| Compound | Atomic Ratio (A:B) | Simplified Whole Number |
|---|---|---|
| Water ($H_2O$) | 2 Hydrogen : 1 Oxygen | 2:1 |
| Ammonia ($NH_3$) | 1 Nitrogen : 3 Hydrogen | 1:3 |
| Methane ($CH_4$) | 1 Carbon : 4 Hydrogen | 1:4 |
| Carbon Dioxide ($CO_2$) | 1 Carbon : 2 Oxygen | 1:2 |
| Glucose ($C_6H_{12}O_6$) | 6 Carbon : 12 Hydrogen : 6 Oxygen | 1:2:1 (Empirical) |
Quick Revision Points
- Atoms do not combine in fractions; they combine as whole units because they are indivisible.
- This explains why chemical formulas always use integers (e.g., $C_6H_{12}O_6$).
- Different compounds of the same elements (like $CO$ and $CO_2$) result from different whole-number ratios.
- This postulate is the foundation of modern molecular stoichiometry and formula writing.
- It successfully explained the Law of Multiple Proportions, which Dalton himself proposed.
NEET Exam Angle
- Numerical Tip: If a problem gives you a mass ratio and asks for a formula, you are essentially applying this postulate.
- NEET MCQ Trick: Be wary of 'Berthollides' or non-stoichiometric compounds (like $Fe_{0.95}O$) which are modern exceptions found in solid-state chemistry.
- Focus on the link between this postulate and the Law of Definite Proportions—it’s a common 'Assertion-Reason' target.
05Chemical Reactions: The Great Rearrangement
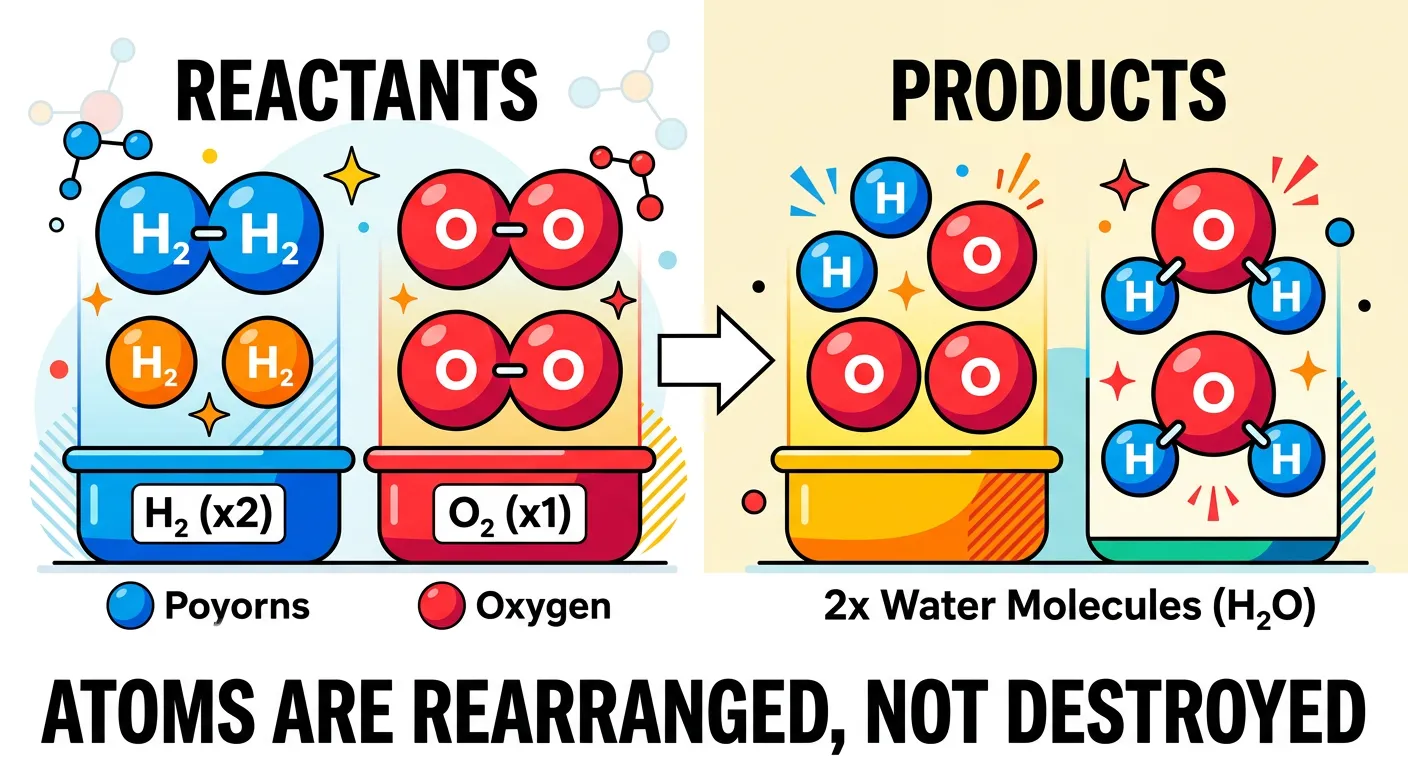
“In chemical reactions, atoms are neither created nor destroyed—they just rearrange. It's like rearranging your bedroom furniture. The chairs and tables are still there, but in new spots! Atoms simply swap partners to create new compounds while keeping the total count the same.”
Dalton’s fourth postulate offers a brilliant and intuitive explanation for what actually happens during a chemical reaction. He stated that chemical reactions involve the reorganization of atoms. Crucially, atoms are neither created nor destroyed in a chemical reaction; they only change how they are grouped together. This is the atomic-level explanation for the Law of Conservation of Mass. If you start with 10 atoms of reactant A and 10 atoms of reactant B, you will end up with exactly 20 atoms in your products, regardless of how complex the new bonding arrangement might be. No mass is lost because no atoms are lost.
Imagine a ballroom dance. At the start of the song, everyone has a partner. When the music changes (representing the reaction occurring), people switch partners. No one leaves the ballroom, and no new people enter from the street. The 'composition' of the pairs changes—some might even form groups of three—but the total number of dancers in the room remains constant. In a chemical reaction like the combustion of methane ($CH_4 + 2O_2 \rightarrow CO_2 + 2H_2O$), the bonds between Carbon and Hydrogen break, the bonds between Oxygen atoms break, and new bonds form to create Carbon Dioxide and Water. The atoms are perfectly conserved. This is precisely why we must always 'balance' chemical equations in our NEET practice problems. Balancing an equation is not just a mathematical chore; it is a literal reflection of Dalton’s reality that matter is permanent and only the 'partnership' of atoms is temporary during a reaction.
Quick Revision Points
- Chemical reactions are essentially 'shuffling' atoms from one combination to another.
- The total mass of reactants always equals the total mass of products because the atom count is constant.
- Atoms cannot be converted into atoms of another element during a chemical reaction (transmutation is chemically impossible).
- This postulate justifies the use of balanced chemical equations in all stoichiometry problems.
- It provides a physical reason for the Law of Conservation of Mass discovered by Lavoisier.
- It implies that the total weight of the universe (neglecting nuclear changes) remains constant.
NEET Exam Angle
- Remember: In nuclear reactions, mass can be converted to energy ($E=mc^2$), but in the chemical reactions covered by Dalton, mass and atoms are always conserved.
- Expect questions asking which postulate explains why we balance equations—it is this one regarding 'reorganization'.
- Balancing an equation is literally a mathematical application of Dalton's conservation of atoms. If you fail to balance, you are effectively 'destroying' atoms in your calculation!
06The Limitations and Evolution of Atomic Theory

“But wait, no theory is perfect! Modern science discovered subatomic particles like protons, electrons, and neutrons. Dalton didn't know atoms could be split. This is why in science, we respect the foundation while always staying curious about the new discoveries that follow. Keep questioning!”
No scientific theory is perfect, and Dalton's was no exception. As science progressed into the late 19th and early 20th centuries, new discoveries began to poke significant holes in his original postulates. For instance, Dalton believed atoms were indivisible, but the discovery of electrons by J.J. Thomson in 1897, protons by Rutherford, and neutrons by Chadwick proved that the atom has a complex internal structure. Furthermore, the discovery of isotopes (atoms of the same element with different masses, like Carbon-12 and Carbon-14) directly disproved Dalton's idea that all atoms of an element have identical mass. We now know that while the number of protons defines an element, the number of neutrons can vary.
Despite these flaws, Dalton’s theory is not 'wrong'—it is simply 'incomplete' for the subatomic scale. It served as the vital stepping stone that led us toward the Modern Atomic Theory, which incorporates subatomic particles and quantum mechanics. However, for most of the chemistry you will perform in the NEET syllabus (especially stoichiometry and gas laws), Dalton's model works perfectly fine. It is essential to learn exactly where Dalton succeeded and where he missed the mark, as the 'limitations' of his theory are a favorite topic for examiners who want to test your understanding of scientific progress.
| Postulate | Modern Correction |
|---|---|
| Atoms are indivisible | Atoms are made of protons, neutrons, and electrons. |
| Atoms of an element are identical | Isotopes exist (same protons, different neutrons/masses). |
| Atoms are indestructible | Nuclear fission and fusion can split or create atoms. |
| Simple whole-number ratios | Non-stoichiometric compounds exist in the solid state. |
| Atoms are solid spheres | Atoms are mostly empty space with a tiny nucleus and electron cloud. |
Quick Revision Points
- Limitations arose due to the discovery of isotopes and subatomic particles.
- Isobars (different elements with the same mass) also challenged Dalton’s logic.
- Dalton could not explain why atoms of different elements have different valencies (combining capacities).
- He failed to distinguish between the 'ultimate particle' of an element (atom) and a compound (molecule).
- The theory didn't account for Gay-Lussac's Law of Gaseous Volumes, which Avogadro later solved.
NEET Exam Angle
- Direct Question: Which of Dalton's postulates is contradicted by the existence of isotopes? (Postulate: All atoms of an element are identical in mass).
- Understand the difference between chemical and nuclear changes. Dalton applies strictly to the former.
- Modern Atomic Theory doesn't replace Dalton; it refines him. Think of Dalton as the 'Classic' version and Modern theory as the 'HD' version.
07Summary: Mastering the Foundation for NEET

“Dalton’s theory was the seed that grew into the vast forest of modern chemistry. By understanding these basics, you’ve mastered the grammar of the universe. Keep these rules in mind as you ace your NEET preparations. See you in the next lesson!”
As we wrap up our study of Dalton's atomic theory, remember that you are looking at the 'DNA' of chemistry. Every complex topic you will encounter later—from Bohr's model of the atom to the complex mechanisms of Organic Chemistry—relies on the fundamental assumption that matter is composed of discrete atoms. Dalton gave us a language to describe the invisible world. By defining the atom as a tangible unit with a specific weight, he allowed scientists to finally start weighing and measuring the universe at a microscopic level. It was the birth of quantitative chemistry as we know it today.
For your NEET exam, this topic is usually high-yield because it connects historical chemistry with modern calculation methods. Don't just memorize the postulates as a list; try to understand the 'why' behind each one. Understand how they directly explain the laws of chemical combination. When you look at a chemical equation, try to see the atoms rearranging as Dalton described. When you solve a mass-mass relationship problem, remember his postulate about identical masses and fixed ratios. Mastery of these basics ensures that you don't make 'silly mistakes' in more advanced chapters like Chemical Bonding or Thermodynamics. You've now built a strong foundation. This section is the 'root' from which the 'tree' of your chemical knowledge will grow. Keep this momentum going as you dive deeper into the actual structure of the atom in the upcoming lessons. Success in NEET comes from a solid understanding of these 'Level 1' concepts before jumping into complex problem-solving.
Quick Revision Points
- Dalton's theory was the first to provide a physical basis for the laws of chemical combination.
- It treats atoms as discrete, mass-bearing units that are the smallest parts of elements.
- The theory explains the Law of Conservation of Mass and the Law of Definite Proportions perfectly.
- It paved the way for Avogadro's Hypothesis and Berzelius's system of atomic symbols.
- Even with its known limitations, it remains the standard starting point for Class 11 Chemistry.
- The theory failed to explain gaseous volumes and the internal structure of atoms.
NEET Exam Angle
- Focus on the conceptual link: Dalton's Theory + Law of Conservation of Mass = Modern Balanced Equations.
- The 'Indivisibility' and 'Identical Mass' parts are the most commonly tested limitations in MCQ formats.
- Revision Tip: Draw a mind map connecting each of Dalton's 4 main postulates to the specific law of chemical combination it explains.
- Prepare for 'Assertion-Reason' questions based on the limitations (isotopes/subatomic particles) versus the original postulates.
Recommended Reading
Explore related Biology topics to build deeper chapter connections for NEET.
- Chemical Equations · Topic 1.10
- Laws of Chemical Combination · Topic 1.4
- Mole Concept · Topic 1.6
- Molar Mass · Topic 1.7
- Nature of Electromagnetic Radiation · Topic 2.1
- Photoelectric Effect · Topic 2.2
- Jump to Key Terms (Quick Revision)
- Review Common NEET Mistakes
- Read Topic FAQs
- Check PYQ Pattern Notes
- Practice NEET MCQs
- Solve NEET PYQs
📚 Key Terms
⚠️ Common NEET Mistakes
- 1Thinking Dalton discovered protons and neutrons (he didn't know they existed).
- 2Confusing Dalton's Theory with Bohr's Model (Dalton had no concept of orbits or energy levels).
- 3Assuming Dalton's theory is 'completely wrong'—it is actually the basis for most chemical calculations even today.
- 4Forgetting that Dalton proposed the Law of Multiple Proportions as a direct result of his atomic theory.
- 5Believing Dalton's theory explains why atoms bond (it only explains that they do bond, not the 'why' of electronic forces).
📝 NEET PYQ Pattern
NEET questions on this topic usually focus on identifying which postulate was proven wrong by modern science (specifically isotopes and subatomic particles). There are also frequent 'Assertion-Reason' questions linking Dalton's postulates to the Laws of Chemical Combination. It is a 'Level 1' topic, meaning questions are usually direct but require precise knowledge of his specific wording.
❓ Frequently Asked Questions
Did John Dalton discover the electron?
No, John Dalton believed atoms were indivisible and had no internal structure. The electron was discovered much later, in 1897, by J.J. Thomson using cathode ray experiments.
Which law of chemical combination was not explained by Dalton's theory?
Dalton's theory primarily failed to explain Gay-Lussac's Law of Gaseous Volumes, as it did not distinguish between atoms and molecules clearly.
How do isotopes disprove Dalton's theory?
Dalton stated that all atoms of an element have identical mass. Isotopes are atoms of the same element (same atomic number) with different masses (different neutron counts), which directly contradicts Dalton's postulate.
Is Dalton's theory still relevant in modern chemistry?
Yes, while refined, the core idea that matter is composed of discrete atoms that rearrange during chemical reactions remains the fundamental basis of stoichiometry and chemical logic.
What is the 'Billiard Ball Model'?
This is a nickname for Dalton's model because he envisioned atoms as solid, hard, indivisible spheres with no internal parts, similar to billiard balls.
What was Dalton's main error in determining atomic weights?
Dalton assumed the simplest ratio for compounds was always 1:1. For example, he thought water was OH instead of H2O, leading to incorrect relative weights for oxygen.
Written By
NEET Content Strategist & Biology Expert
Sangita Kumari is a NEET educator and content strategist with over 6 years of experience teaching Biology, Chemistry, and Physics to Class 11 and 12 aspirants. She helps bridge the gap between traditional NCERT preparation and modern AI-powered learning. Her content is trusted by thousands of NEET aspirants across India.