🎬 Video Lesson Available
Watch the full 7-slide video lesson for Photoelectric Effect with AI teacher narration and visual explanations.
01The Carrom Analogy: Defining the Photoelectric Effect in Atomic Structure
“Imagine sunlight hitting a metal plate like a game of carrom! When light hits a metal surface, it kicks out electrons. This is the Photoelectric Effect. It’s like magic, but it’s actually physics! Let’s dive into how this tiny miracle powers our modern world.”
Imagine you are playing a game of carrom. You aim your striker at a coin, and the moment the striker hits, the coin flies off towards the pocket. In the world of atomic structure, the photoelectric effect operates on the exact same principle. When light of a certain frequency strikes the surface of a metal, it behaves like that striker, knocking electrons—our carrom coins—out of the metal surface. This phenomenon was first observed by Heinrich Hertz in 1887 during his experiments on electromagnetic waves. He noticed that sparks occurred more easily between two metal electrodes when they were illuminated with ultraviolet light. This was the first hint that light had the power to liberate charge carriers from solid matter.
What makes this process truly fascinating is its 'all-or-nothing' nature. In a carrom game, if the striker is moving too slowly, the coin won't budge. Similarly, if the incident light doesn't have enough 'punch,' no electrons are ejected, regardless of how long you shine the light or how bright it is. This observation completely baffled the scientists of that era because they believed light was a continuous wave. If light were a wave, energy should eventually accumulate on the metal surface and kick the electron out over time. But reality showed something different: the process is instantaneous. There is no measurable time lag between the arrival of the photon and the emission of the electron, occurring in less than $10^{-9}$ seconds.
This instantaneous ejection forced the scientific community to reconsider the very nature of light. It suggested that light isn't just a smooth ripple in a pond, but a stream of discrete 'bullets' or particles. This shift toward the dual nature of light—behaving as both a wave and a particle—is a cornerstone of modern chemistry and physics, particularly for NEET aspirants who need to understand how subatomic interactions dictate macroscopic observations.
Quick Revision Points
- Observed by Hertz; electrons are ejected from metal surfaces when light of specific frequency hits them.
- The process is strictly instantaneous, occurring in less than $10^{-9}$ seconds.
- Classical wave theory could not explain the lack of a time lag.
- The 'Carrom Analogy' helps visualize the particle-like collision between light (striker) and electron (coin).
- Ejected electrons are technically called 'photoelectrons,' though they are identical to normal electrons.
NEET Exam Angle
- Conceptual questions often focus on why classical wave theory failed (the time-lag problem).
- Remember that this effect provides primary evidence for the particle nature of electromagnetic radiation.
- NEET frequently tests the definition of 'instantaneous emission' as a characteristic feature of this effect.
02Threshold Frequency: The Minimum 'Punch' Required for Emission
“Not every light works! Think of it like a coconut stall at a fair; you need a specific strength to break a coconut. If the light's frequency is too low, no electrons come out. We call this minimum requirement the 'Threshold Frequency'. You need enough 'punch'!”
One of the most critical concepts in the photoelectric effect is the Threshold Frequency, denoted by the symbol $\nu_0$ (nu-zero). Think of it like a coconut stall at a village fair. To break a coconut and win a prize, your throw must have a certain minimum force. If you throw the ball weakly, the coconut stays intact. Even if you throw a thousand weak balls, the coconut won't break because none of them individual hits have the necessary 'punch.' In this analogy, the strength of your throw represents the frequency of light, and the number of balls represents the intensity (brightness).
For every metal, there is a characteristic minimum frequency below which no photoelectrons are emitted, no matter how intense the light is. If the incident frequency $\nu$ is less than $\nu_0$, the electrons remain bound to the metal nuclei. This is a crucial distinction for NEET aspirants: the frequency determines whether emission happens, while intensity determines how many electrons come out once the threshold is met. For example, red light (low frequency) might fail to eject electrons from a potassium surface even if it is incredibly bright, whereas even a dim violet light (high frequency) will succeed immediately.
This threshold varies from metal to metal. Alkali metals like Cesium and Potassium have very low threshold frequencies, making them highly sensitive to visible light. This is why they are commonly used in photoelectric cells. Heavier or more stable metals like Copper or Platinum require much higher frequencies, often in the ultraviolet range, to trigger the effect. Understanding this relationship helps in calculating the energy of incident photons using Planck's equation, $E = h\nu$.
| Metal Type | Typical Threshold Frequency | Relative Ionization Energy |
|---|---|---|
| Alkali Metals (Cs, K) | Low (Visible Range) | Low |
| Transition Metals (Cu, Fe) | High (UV Range) | Moderate |
| Noble Metals (Pt, Au) | Very High (Deep UV) | High |
Quick Revision Points
- Threshold Frequency ($\nu_0$) is the minimum frequency required to eject an electron.
- If incident frequency $\nu < \nu_0$, zero photoelectrons are emitted.
- Threshold frequency is a characteristic property of the specific metal used.
- The relationship between energy and frequency is given by $E = h\nu$.
- Alkali metals exhibit the effect even with visible light due to low ionization enthalpies.
NEET Exam Angle
- Numerical problems often ask you to identify if emission will occur given a specific wavelength and threshold frequency.
- Remember: $\nu = c/\lambda$. A higher frequency means a shorter wavelength. Emission requires $\lambda < \lambda_{threshold}$.
- Comparison questions between metals like Cesium and Zinc are common; Cesium is more 'photo-active.'
03The Work Function: Understanding the 'Rent' for Electron Liberation
“Every metal has a 'Work Function'—the minimum energy needed to free an electron. It’s like the rent you pay to leave your house. If the photon doesn't have at least that much energy, the electron won't budge, no matter how bright the light is.”
While threshold frequency describes the 'speed' or 'quality' of the light, the Work Function ($\Phi$ or $W_0$) describes the energy requirement from the electron's perspective. Think of the Work Function as the 'rent' an electron must pay to leave the metal surface. Electrons are naturally attracted to the positive nuclei within the metal lattice. To break free from this electrostatic pull and escape into the vacuum, the electron needs a specific amount of energy. If the incoming photon provides exactly that amount of energy, the electron is liberated but has no speed—it just 'sits' on the surface.
Mathematically, the Work Function is defined as $W_0 = h\nu_0$. It represents the minimum energy binding the electron to the metal. In NEET chemistry and physics, you will frequently encounter energy units in Electron Volts (eV) rather than Joules. This is because Joules are too large for subatomic scales. One eV is the energy gained by an electron when accelerated through a potential difference of 1 Volt ($1 eV = 1.602 \times 10^{-19}$ Joules). Converting between these units is a vital skill for solving numerical problems quickly during the exam.
Different metals have different 'rents.' Metals with low ionization enthalpies have low work functions. For instance, Cesium has a work function of approximately 2.14 eV, making it one of the easiest metals to trigger. Platinum, on the other hand, has a work function of about 5.65 eV. This difference is why certain materials are preferred for solar panels while others are used for high-stability electrodes. When solving for energy, always ensure your units are consistent—either keep everything in Joules or convert your final answer to eV as per the options provided.
| Constant/Value | Symbol | Magnitude |
|---|---|---|
| Planck's Constant | $h$ | $6.626 \times 10^{-34} J\cdot s$ |
| Charge of Electron | $e$ | $1.602 \times 10^{-19} C$ |
| Energy Conversion | $1 eV$ | $1.602 \times 10^{-19} J$ |
| Velocity of Light | $c$ | $3 \times 10^8 m/s$ |
Quick Revision Points
- Work Function ($W_0$) is the minimum energy needed to liberate an electron.
- Formula: $W_0 = h\nu_0 = hc/\lambda_0$.
- Unit conversion: Always remember $1 eV = 1.6 \times 10^{-19}$ Joules.
- Higher Work Function means the electron is more tightly bound to the nucleus.
- The Work Function is independent of the intensity of the incident light.
NEET Exam Angle
- Many questions require finding the maximum wavelength ($\lambda_0$) for emission given the Work Function in eV.
- Useful Shortcut: $E (in eV) \approx 12400 / \lambda (in \mathring{A})$ or $1240 / \lambda (in nm)$. This saves massive calculation time!
- Pay attention to the metal's position in the periodic table; s-block metals usually have lower work functions.
04Wave-Particle Duality: How Einstein’s Photons Solved the Classical Paradox

“Classical physics got stumped! They thought light was just a wave, but it couldn't explain this effect. Enter Albert Einstein! He proposed light acts like a stream of 'packets' of energy called photons. He basically proved light is a particle, not just a ripple.”
By the late 19th century, physics was in a state of crisis. The classical wave theory of light, which successfully explained interference and diffraction, failed miserably when applied to the photoelectric effect. According to wave theory, the energy of light was proportional to its intensity (amplitude). If you shone a very bright light on a metal, the electrons should eventually absorb enough energy to jump out. But experiments showed that if the frequency was wrong, even the brightest light did nothing. Conversely, even the dimmest light of high frequency caused immediate emission. Classical physics simply couldn't explain this 'threshold' behavior.
Enter Albert Einstein in 1905. He proposed a revolutionary idea based on Max Planck’s quantum hypothesis: light does not travel as a continuous wave, but as discrete 'packets' or 'quanta' of energy, which he called photons. Each photon carries energy proportional to its frequency ($E = h\nu$). In Einstein’s view, the photoelectric effect is a one-on-one collision between a single photon and a single electron. If the individual photon doesn't have enough energy to overcome the metal's work function, it cannot 'team up' with other photons to kick the electron out.
This conceptual shift was massive. It proved that light has a dual nature—it behaves like a wave when traveling through space, but behaves like a particle when interacting with matter. Einstein’s explanation was so significant that it earned him the Nobel Prize in Physics in 1921. For a NEET student, understanding this duality is essential for mastering the later chapters on atomic structure and the De Broglie hypothesis, which suggests that matter itself (like electrons) also has wave-like properties.
Quick Revision Points
- Classical Wave Theory predicted emission depends on intensity; experiments proved it depends on frequency.
- Einstein proposed the 'Photon'—a discrete packet of energy.
- Photoelectric effect involves a 1:1 interaction between a photon and an electron.
- Energy of light is 'quantized' ($E = nh\nu$, where $n$ is an integer).
- Dual nature: Light exhibits both wave-like and particle-like characteristics.
NEET Exam Angle
- Questions often test why wave theory failed. The keywords are 'intensity dependence' vs 'frequency dependence.'
- Understand that 'Quantization-postulates-neet-chemistry)-postulates-neet-chemistry)-postulates-neet-chemistry)-postulates-neet-chemistry)' means energy exists in specific, fixed amounts, not as a continuous flow.
- Einstein’s Nobel Prize was specifically for the Photoelectric Effect, not Relativity—a common trivia question in exams!
05Intensity vs. Frequency: The Golden Rule for NEET Aspirants
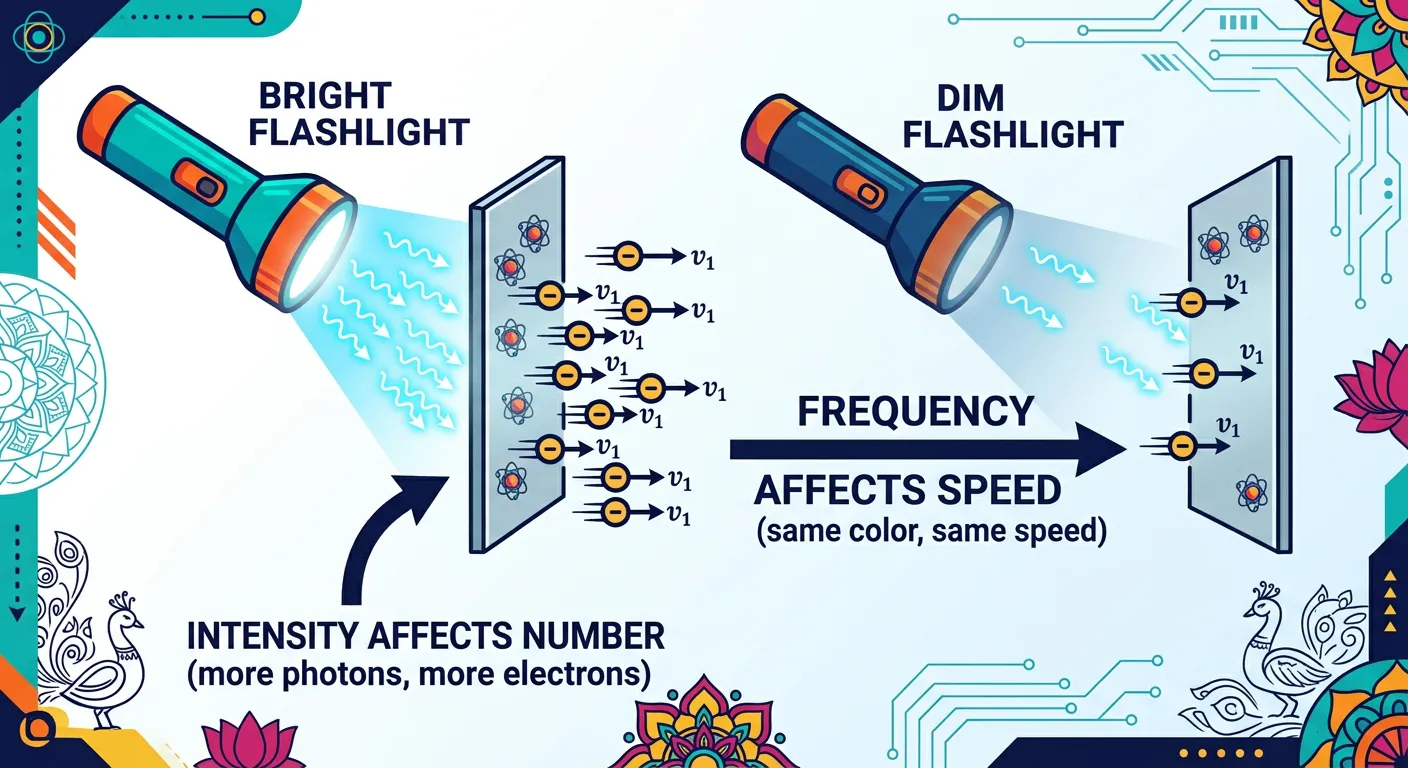
“Here is the NEET secret: Brightness (intensity) only increases the number of electrons, not their speed! If you want faster electrons, you need higher frequency light (like UV). Remember: Frequency is the boss of speed, while intensity is the boss of quantity.”
One of the most frequent 'trap' areas in NEET is the distinction between light intensity and light frequency. Aspirants often confuse 'brightness' with 'energy.' Let’s clarify this once and for all: Frequency is about the 'quality' of the photon (how much energy each one carries), while Intensity is about the 'quantity' of photons (how many are hitting the surface per second). If the frequency of light is below the threshold, you can increase the intensity to infinity, but you will still get zero electrons.
However, once you are above the threshold frequency, increasing the intensity has a very specific effect: it increases the number of photoelectrons emitted. More photons hitting the surface means more 1-on-1 collisions, leading to a higher 'photocurrent.' But—and this is the part NEET loves to test—increasing the intensity does NOT increase the kinetic energy or the speed of the ejected electrons. If you want the electrons to move faster, you must increase the frequency (the energy of each individual photon).
Think of it like rain. Frequency is the size of the individual raindrops, while intensity is how heavily it is raining. If the raindrops are too small (low frequency) to break a leaf, it doesn't matter if it's a torrential downpour (high intensity)—the leaf won't break. But if the raindrops are large enough (high frequency) to break the leaf, then a heavier downpour (high intensity) will simply break more leaves per minute. This 'Golden Rule' is the key to solving conceptual MCQs in seconds.
| Property | If Increased (at $\nu > \nu_0$) | Effect on Electrons |
|---|---|---|
| Intensity | Number of Photons ↑ | Number of Photoelectrons ↑ (Current ↑) |
| Intensity | Number of Photons ↑ | Maximum Kinetic Energy: No Change |
| Frequency | Energy per Photon ↑ | Maximum Kinetic Energy ↑ (Speed ↑) |
| Frequency | Energy per Photon ↑ | Number of Photoelectrons: No Change (if intensity is constant) |
Quick Revision Points
- Intensity = Number of photons per unit area per unit time.
- Frequency = Energy of each individual photon.
- Photocurrent is directly proportional to Intensity (provided $\nu > \nu_0$).
- Maximum Kinetic Energy is independent of Intensity but directly dependent on Frequency.
- Common Trap: 'Bright red light' (low $\nu$) vs 'Dim UV light' (high $\nu$).
NEET Exam Angle
- Graphical questions on Current vs. Intensity are very common; the graph is a straight line through the origin.
- Remember: If frequency is doubled, K.E. more than doubles (since K.E. = $h\nu - W_0$).
- A question might ask: 'If intensity is doubled, what happens to the stopping potential?' Answer: It remains the same!
06Einstein’s Photoelectric Equation: The Conservation of Energy Formula
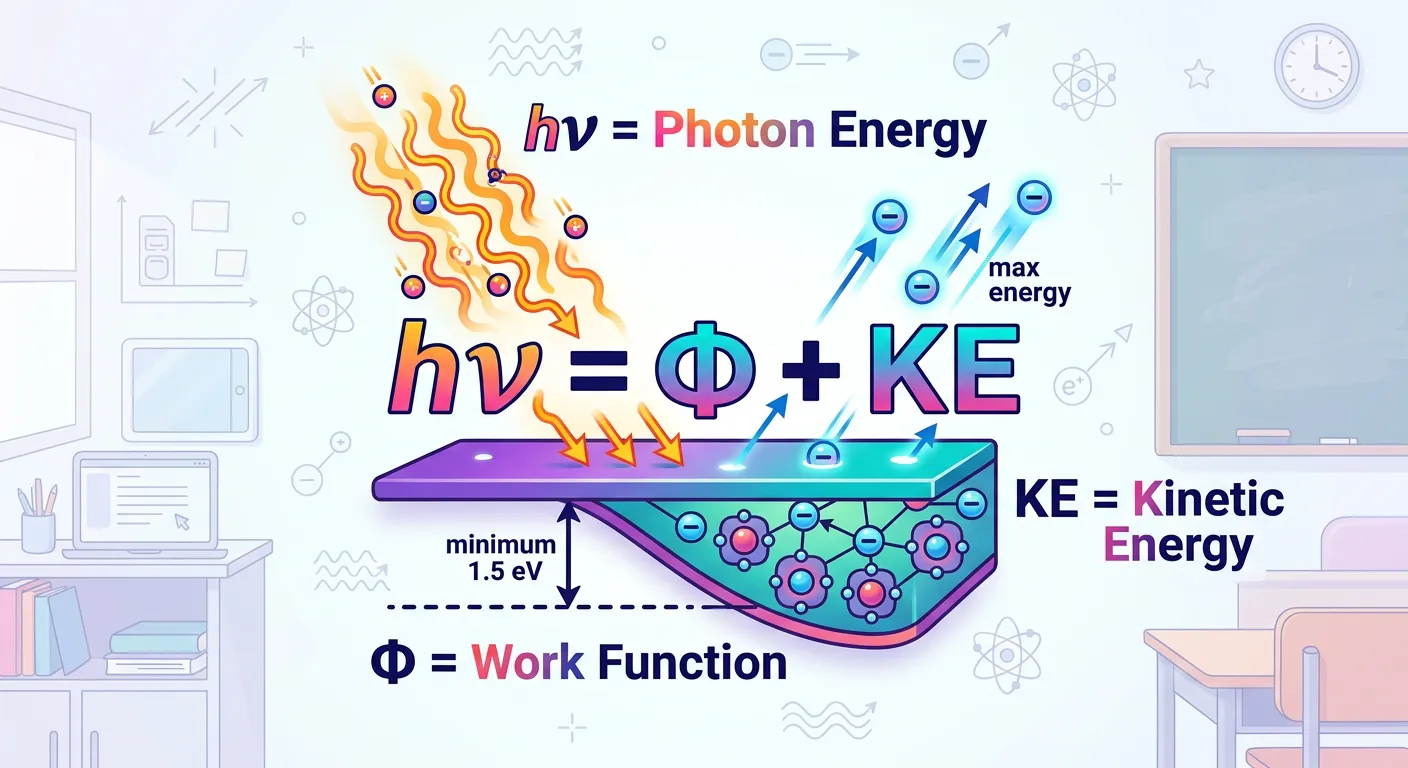
“Einstein’s famous equation is just a simple budget: Energy in equals Work Function plus Kinetic Energy. Whatever is left over from the photon's energy after 'paying the rent' becomes the electron's speed. It’s simple energy conservation at the subatomic level!”
Einstein’s photoelectric equation is essentially a statement of the Law of Conservation of Energy applied at the atomic level. It is the most important formula for this topic and a staple of NEET numericals. The equation is: $h\nu = W_0 + K.E._{max}$. In simple terms, this tells us that the total energy brought in by a photon ($h\nu$) is used in two parts. First, a portion is used to pay the 'rent' or binding energy of the electron ($W_0$). Second, whatever energy remains is converted into the kinetic energy of the escaping electron ($1/2 mv^2$).
We can rewrite this equation in several ways to suit different problem types. Since $W_0 = h\nu_0$, the equation becomes $h\nu = h\nu_0 + K.E.{max}$, which simplifies to $K.E.{max} = h(\nu - \nu_0)$. If you are working with wavelengths, remember that $\nu = c/\lambda$. This leads to $K.E._{max} = hc(1/\lambda - 1/\lambda_0)$. These variations are vital because NEET questions rarely give you the energy directly; they usually provide frequency or wavelength and expect you to derive the kinetic energy.
Graphing this equation is another high-yield area. If you plot Maximum Kinetic Energy ($y$-axis) against Frequency ($x$-axis), the equation follows the form $y = mx + c$. The slope of the line is always Planck’s constant ($h$), and the $x$-intercept is the threshold frequency ($\nu_0$). The negative $y$-intercept represents the Work Function ($W_0$). Interestingly, the slope ($h$) is universal—it will be the same for all metals, while the intercepts will shift depending on the specific metal used.
| Graphical Feature | Physical Significance |
|---|---|
| Slope of K.E. vs. $\nu$ | Planck’s Constant ($h$) |
| X-intercept of K.E. vs. $\nu$ | Threshold Frequency ($\nu_0$) |
| Y-intercept of K.E. vs. $\nu$ | Negative Work Function ($-W_0$) |
| Slope of Stopping Potential vs. $\nu$ | $h/e$ |
Quick Revision Points
- Einstein’s Equation: $h\nu = W_0 + K.E._{max}$.
- Kinetic energy is the 'leftover' energy after the work function hurdle is cleared.
- If $h\nu = W_0$, then $K.E. = 0$ (electron is just released).
- The maximum kinetic energy depends linearly on the frequency of incident light.
- Electrons are emitted with a range of kinetic energies, but Einstein's equation calculates the 'maximum' possible.
NEET Exam Angle
- Always convert your units! If $h\nu$ is in Joules and $W_0$ is in eV, you must equalize them before subtracting.
- Be prepared for 'Stopping Potential' ($V_0$) questions: $K.E._{max} = eV_0$.
- Graphical interpretation questions are frequent—identify the slope and intercepts correctly to avoid negative marking.
07Modern Applications and Exam Strategy: From Solar Cells to NEET Success
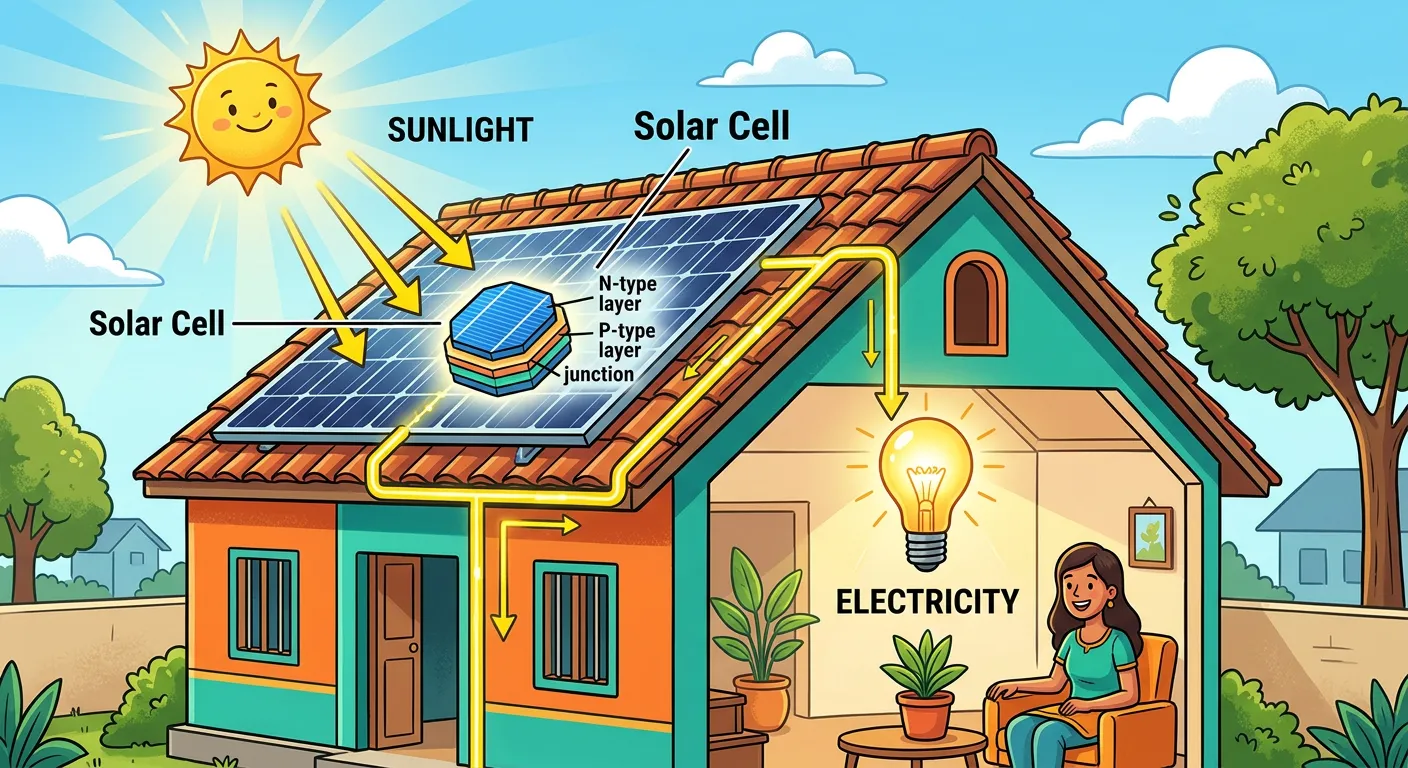
“From automatic sliding doors at malls to solar panels on your roof, the photoelectric effect is everywhere. You’ve just mastered a concept that earned Einstein his Nobel Prize! Keep this logic clear, and you will crush the physics section of your NEET exam.”
The photoelectric effect isn't just a theoretical concept in a textbook; it powers the world around you. The most visible application is in solar panels (photovoltaic cells), where sunlight directly knocks electrons loose in semiconductor materials to generate electricity. Similarly, the automatic doors at shopping malls utilize 'photoelectric sensors.' When you walk through the doorway, you block a beam of light, stopping the flow of photoelectrons and triggering a circuit that opens the door. From burglar alarms to smoke detectors and the light meters in high-end cameras, this 100-year-old discovery remains a pillar of modern electronics.
For your NEET preparation, this topic is what we call 'low-hanging fruit.' It is conceptually straightforward, the formulas are few, and the questions are often repetitive. Between the Physics and Chemistry sections of the NEET exam, you can expect at least 1-2 questions from this area. To maximize your score, you must master the conversion between wavelength (nanometers/Angstroms) and energy (eV). Most students lose marks here due to calculation errors, not conceptual misunderstanding.
Final checklist for the 2025 exam: Memorize the value of Planck's constant and the mass of an electron. Practice sketching the four main graphs (Current vs Intensity, K.E. vs Frequency, Stopping Potential vs Frequency, and Current vs Potential). Remember that while the photoelectric effect proves the particle nature of light, it does not disprove the wave nature—light is simply 'both.' Keep your logic focused on the energy budget, and you will find this topic to be one of the easiest ways to secure 4-8 marks.
Quick Revision Points
- Applications: Solar cells, automatic doors, phototubes, and light sensors.
- Used to measure the intensity of light accurately.
- Key focus for NEET: Numericals on $h\nu = W_0 + K.E.$
- Kinetic energy can be represented as $eV_0$ (Stopping Potential).
- Particle nature of light is the primary conclusion drawn from this effect.
NEET Exam Angle
- Calculation Tip: Use $hc = 1240 eV\cdot nm$ or $12400 eV\cdot \mathring{A}$ to find energy quickly.
- Don't confuse 'Photoelectric Effect' with 'Thermionic Emission' (heat-based) or 'Field Emission' (electric field-based).
- Graphical questions on the effect of changing the anode potential are frequent in the Physics section—study the 'saturation current' concept carefully.
Recommended Reading
Explore related Biology topics to build deeper chapter connections for NEET.
- Nature of Electromagnetic Radiation · Topic 2.1
- Quantum Mechanical Model · Topic 2.12
- Atomic Orbitals · Topic 2.13
- Significance of Quantum Numbers · Topic 2.16
- Bohr Model · Topic 2.4
- de Broglie's Relationship · Topic 2.9
- Jump to Key Terms (Quick Revision)
- Review Common NEET Mistakes
- Read Topic FAQs
- Check PYQ Pattern Notes
- Practice NEET MCQs
- Solve NEET PYQs
📚 Key Terms
⚠️ Common NEET Mistakes
- 1Thinking that increasing intensity will eventually cause electron emission even if the frequency is below the threshold.
- 2Forgetting to convert units, such as subtracting a work function in eV from photon energy in Joules without conversion.
- 3Confusing threshold frequency with threshold wavelength; remember that frequency must be 'above' threshold, but wavelength must be 'below' threshold.
- 4Assuming that 'number of photoelectrons' and 'kinetic energy' are the same thing; intensity affects the number, frequency affects the energy.
- 5Misinterpreting the K.E. vs. Frequency graph by assuming the slope changes for different metals (the slope is always Planck's constant 'h').
📝 NEET PYQ Pattern
Between 2018 and 2024, NEET has consistently featured numerical problems based on Einstein's equation, often requiring conversion between eV and Joules. Graphical questions representing the variation of stopping potential or kinetic energy against frequency are also frequent, appearing in roughly 60% of the past five years' papers.
❓ Frequently Asked Questions
Does the intensity of light affect the kinetic energy of the emitted photoelectrons?
No. The maximum kinetic energy of photoelectrons depends solely on the frequency of the incident light and the nature of the metal (work function). Intensity only increases the number of electrons emitted per second, not their individual energy.
What happens if the frequency of incident light is exactly equal to the threshold frequency?
If the incident frequency equals the threshold frequency ($\nu = \nu_0$), the energy of the photon is just enough to liberate the electron from the metal surface. The emitted electron will have zero kinetic energy ($K.E. = 0$).
Why is the photoelectric effect considered evidence for the particle nature of light?
Because it shows that light energy is delivered in discrete packets (photons). Classical wave theory fails to explain why there is no time lag for emission and why a minimum frequency is required, both of which are explained by particle-like 1-on-1 collisions.
Can the photoelectric effect occur in non-metals? If not, why?
While it can theoretically occur in non-metals, it is much harder to observe. Non-metals have very high ionization energies (and thus high work functions), meaning they require very high-frequency radiation (like X-rays) to eject electrons compared to the visible/UV light used for metals.
How does the stopping potential change if we increase the frequency of the incident light?
Increasing the frequency increases the maximum kinetic energy of the photoelectrons. Since stopping potential ($V_0$) is directly proportional to kinetic energy ($K.E._{max} = eV_0$), the stopping potential will also increase (become more negative).
What is the relationship between the wavelength of light and the photoelectric effect?
Energy is inversely proportional to wavelength ($E = hc/\lambda$). Therefore, for emission to occur, the incident wavelength must be less than or equal to a certain 'threshold wavelength' ($\lambda_0$). Longer wavelengths (like infrared) usually lack the energy to trigger the effect.
Written By
NEET Content Strategist & Biology Expert
Sangita Kumari is a NEET educator and content strategist with over 6 years of experience teaching Biology, Chemistry, and Physics to Class 11 and 12 aspirants. She helps bridge the gap between traditional NCERT preparation and modern AI-powered learning. Her content is trusted by thousands of NEET aspirants across India.