🎬 Video Lesson Available
Watch the full 7-slide video lesson for Bohr Model with AI teacher narration and visual explanations.
01Beyond Rutherford: The Genesis of Bohr’s Atomic Paradigm

“Welcome, NEET warriors! Ever wondered how the hydrogen atom holds its secrets? After Rutherford's model hit a dead end, Niels Bohr stepped in with a brilliant idea. He treated the electron not like a chaotic planet, but like a disciplined student following a strict path.”
To understand where Niels Bohr was coming from in 1913, we first have to look at the wreckage left behind by Rutherford’s model. While Rutherford correctly identified the nucleus, his model faced a massive crisis thanks to Maxwell’s electromagnetic theory. According to classical physics, any charged particle (like an electron) moving in a circular path should accelerate and constantly radiate energy. If an electron lost energy this way, it would spiral into the nucleus in less than a nanosecond. Clearly, that doesn't happen, or we wouldn't exist! This 'instability' was the wall that classical physics hit.
Niels Bohr took a leap of faith. He realized that to explain the atom, we had to stop thinking like classical engineers and start thinking like quantum pioneers. He didn't just tweak the existing model; he introduced a revolutionary hypothesis specifically for the hydrogen atom. Instead of the chaotic, unpredictable motion allowed by Rutherford, Bohr suggested that electrons are highly disciplined. They don't just fly anywhere; they are restricted to specific paths. This was the shift from the 'chaotic' planetary model to a 'disciplined' orbital system where the laws of classical physics were intentionally set aside to explain atomic reality.
Comparing planetary motion to electronic motion helps, but only to a point. While planets stay in orbit due to gravity, Bohr’s electrons are held by electrostatic forces. However, unlike planets which could theoretically orbit at any distance, Bohr’s electrons are 'invited' only to specific distances. This section sets the stage for the five postulates that redefined chemistry and physics for the 20th century.
| Feature | Rutherford Model | Bohr Model |
|---|---|---|
| Electron Path | Any circular orbit | Specific 'Stationary' orbits |
| Stability | Unexplained (Atom should collapse) | Postulated as stable |
| Energy | Continuous loss predicted | No loss while in orbit |
| Spectral Prediction | Continuous spectrum | Discrete line spectrum |
Quick Revision Points
- Rutherford's model failed because it couldn't explain why electrons don't spiral into the nucleus.
- Maxwell’s theory predicted that accelerating charges must emit radiation.
- Bohr introduced his model in 1913, specifically targeting the hydrogen atom's behavior.
- The model represents a transition point between classical mechanics and early quantum theory.
- Bohr’s genius was in accepting that classical physics simply doesn't apply at the atomic scale.
NEET Exam Angle
- Remember that the Bohr model is strictly valid only for 'hydrogen-like' species (1 electron systems).
- Be ready for questions asking why Rutherford’s model was discarded; the answer is always 'atomic instability' and 'failure to explain the line spectrum.'
02Postulate I: Stationary Orbits and the Resolution of Atomic Collapse
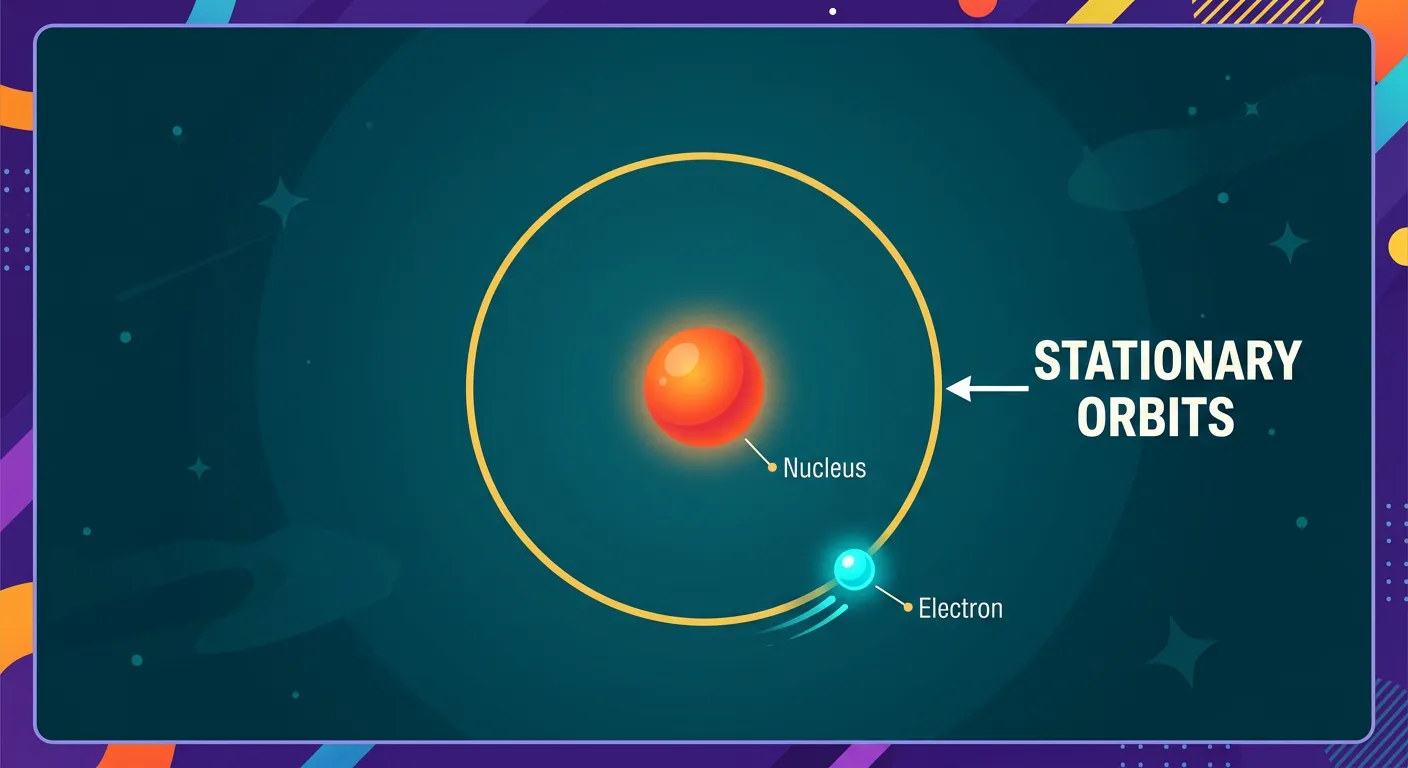
“Bohr's first big postulate: electrons travel in fixed, circular paths called stationary orbits or shells. Think of these like the specific tracks in a stadium. An electron stays in its own lane and doesn't lose energy, preventing it from spiraling into the nucleus. Simple, right?”
Bohr’s first postulate is the ultimate 'rule-breaker.' He stated that electrons revolve around the nucleus only in certain allowed circular paths. He called these 'stationary states' or 'stationary orbits.' The term 'stationary' doesn't mean the electron isn't moving; it means the energy of the electron in that orbit is constant. This was a direct challenge to classical electromagnetism. Bohr essentially said, 'I know the laws of physics say it should radiate energy, but in these specific orbits, it simply doesn't.'
This postulate provides the physics behind why atoms are stable. Inside these allowed orbits, the centrifugal force arising from the electron’s circular motion perfectly balances the electrostatic attraction (Coulombic force) from the nucleus. Since the electron does not radiate energy while it stays in its specific track, it never loses the kinetic energy needed to stay aloft. It is the atomic equivalent of a 'frictionless' environment where the electron can maintain its momentum indefinitely.
Understanding this 'non-radiating' condition is vital for your NEET prep. It explains why the atom doesn't collapse and sets the foundation for everything else in quantum chemistry. Without this postulate, the concept of energy levels would be impossible to define. It essentially gives the electron a permanent 'parking spot' where it is safe from the spiral of death predicted by Maxwell.
Quick Revision Points
- Electrons move in fixed circular paths called stationary orbits.
- Energy is neither emitted nor absorbed as long as an electron stays in its orbit.
- The centrifugal force is balanced by the Coulombic (electrostatic) attraction.
- These orbits are also referred to as 'shells' or 'energy levels.'
- This postulate directly solves the problem of atomic collapse mentioned in Rutherford's limitations.
NEET Exam Angle
- Focus on the term 'Stationary States'—it’s a favorite in MCQ options.
- Expect conceptual questions on the balance of forces: $mv^2/r = kZe^2/r^2$. Knowing this equilibrium is the key to deriving radius and velocity formulas later.
- Keep in mind that Bohr assumed circular orbits, which was later modified by Sommerfeld to include elliptical paths (though not needed for basic Bohr postulates).
03Postulate II: Energy Quantization and the Principal Quantum Number (n)
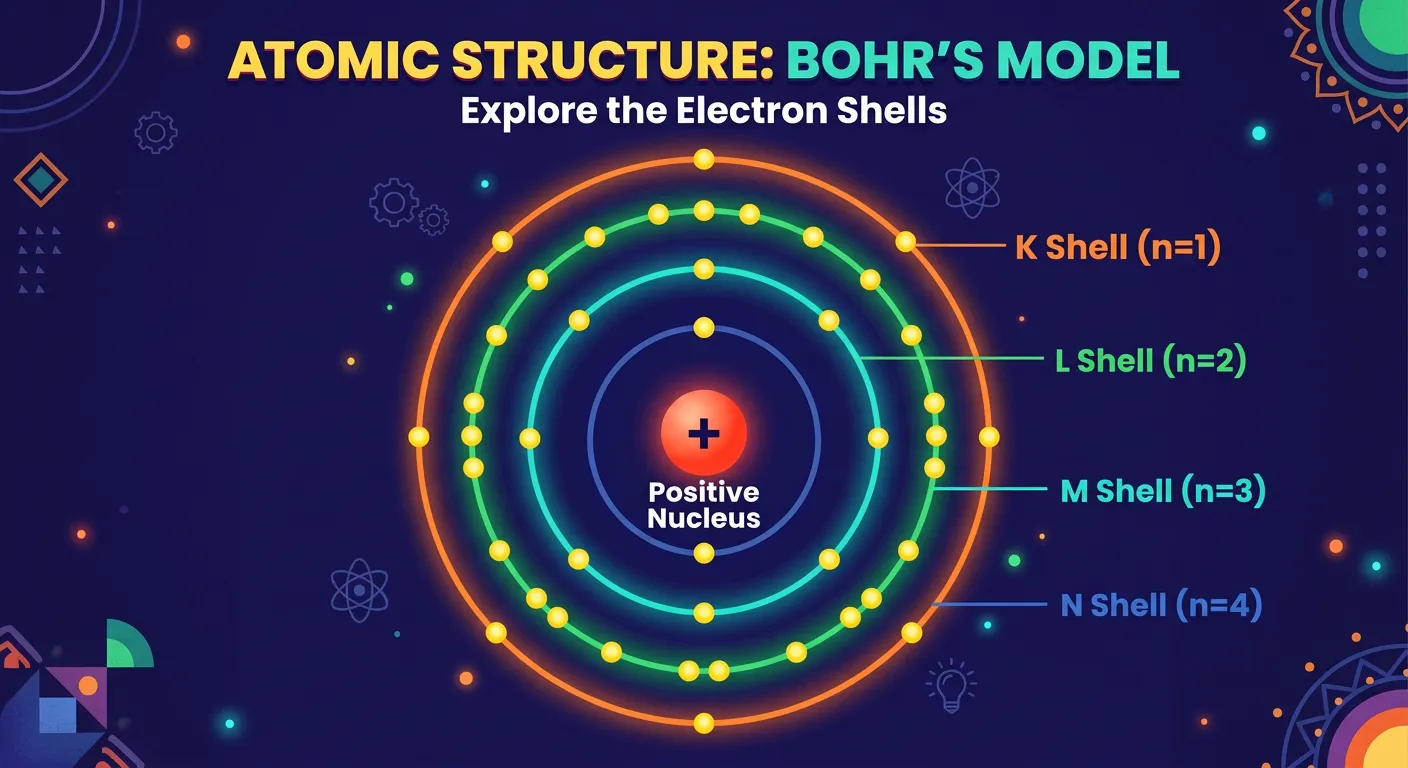
“These orbits are quantized! We label them as n = 1, 2, 3, or K, L, M, N shells. As the shell number increases, so does the distance from the nucleus. Each orbit has a specific, fixed amount of energy—just like energy levels in a tiered auditorium.”
In the Bohr model, not all orbits are created equal. This brings us to the second postulate: the concept of quantization. Bohr introduced the Principal Quantum Number, denoted by the letter 'n'. These orbits are numbered as integers: n=1, n=2, n=3, and so on. Alternatively, they are labeled with capital letters starting from the nucleus outward: K, L, M, N, O... This integer 'n' is the 'ID card' for the electron's position and energy state.
The distance of the shell from the nucleus increases as 'n' increases. In the hydrogen atom, n=1 is called the Ground State—this is where the electron is most stable and has the lowest energy. Any level higher than n=1 is considered an 'Excited State.' A crucial point for NEET aspirants is to realize that energy levels are 'tiered.' Just like a ladder has rungs but no space to stand between them, an electron can be in n=1 or n=2, but it can never exist at n=1.5.
This 'all-or-nothing' existence at discrete energy values is what we mean by 'quantization.' The energy of these shells is not random; it follows a specific mathematical relationship where energy is inversely proportional to the square of n. As n increases, the orbits get further apart in distance but closer together in energy.
| Shell Name | Principal Quantum Number (n) | Relative Distance from Nucleus | Energy State |
|---|---|---|---|
| K | 1 | Closest | Ground State (Lowest Energy) |
| L | 2 | Farther | 1st Excited State |
| M | 3 | Even Farther | 2nd Excited State |
| N | 4 | Most Distant | 3rd Excited State |
Quick Revision Points
- Orbits are identified by the Principal Quantum Number 'n' (1, 2, 3...).
- Shell nomenclature: K=1, L=2, M=3, N=4.
- n=1 is the 'Ground State,' representing the lowest possible energy.
- Energy levels are discrete (quantized), meaning no 'middle' states exist.
- As 'n' increases, the radius of the orbit increases ($r \propto n^2$).
NEET Exam Angle
- A common trick question: 'What is the value of n for the 2nd excited state?' The answer is n=3. Always remember: nth excited state = n+1 shell.
- Notice the relationship between n and radius. In numericals, you will often use $r_n = 0.529 \times n^2 / Z$ Angstroms.
04Postulate III: Electronic Excitation and Energy Absorption Mechanics
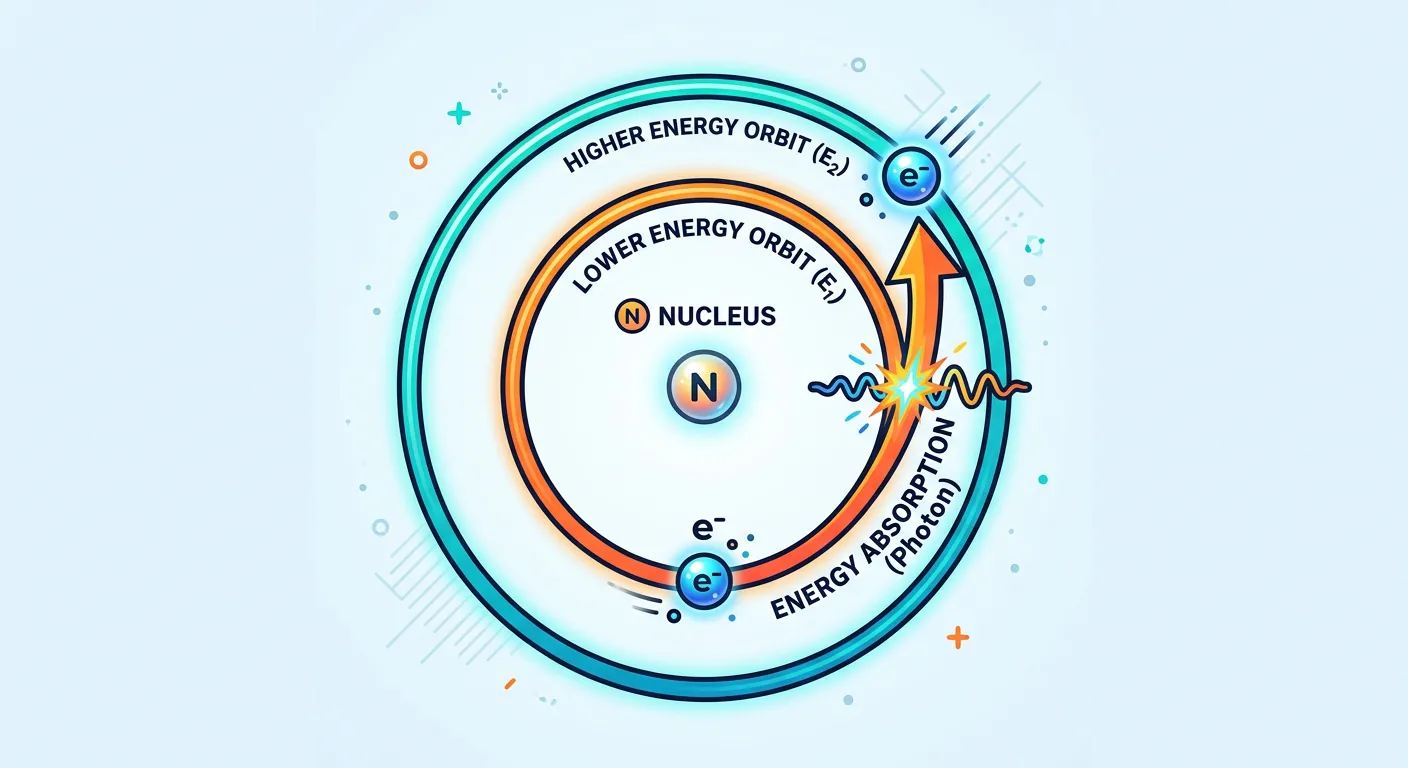
“What happens when an electron gets excited? It absorbs energy and jumps to a higher orbit! It's like taking a lift to a higher floor. But remember, it can only occupy these specific levels—no hovering in between. It is either on the first floor or the second!”
How does an electron move if it’s trapped in these fixed orbits? Bohr explained this through 'quantum leaps.' His third postulate describes electronic excitation. An electron can move from a lower energy level (lower n) to a higher energy level (higher n) only if it absorbs a very specific amount of energy. This energy must be exactly equal to the difference between the two levels. If you provide less energy than the gap, the electron stays put. It's a binary choice: jump or stay.
Think of this like a lift in a building. To get from the 1st floor to the 3rd floor, you need a specific amount of electrical energy. You can't stop the lift halfway between floors and live there. Similarly, an electron in the ground state (n=1) can jump to n=2 if it absorbs energy equal to $E_2 - E_1$. This energy is usually provided by heat, electricity, or light (photons).
This postulate is the foundation of 'Absorption Spectra.' When white light passes through a gas, the gas atoms 'pick out' and absorb the specific wavelengths (energies) that match their internal jumps. This leaves dark lines in the spectrum. For NEET, you must be comfortable with the formula $\Delta E = E_{final} - E_{initial}$. If $\Delta E$ is positive, energy is being absorbed. This process is temporary, as the electron in a higher energy level is unstable and will eventually want to return home.
Quick Revision Points
- Electrons jump to higher levels by absorbing energy.
- The energy absorbed must be exactly $\Delta E = E_2 - E_1$.
- This process is called 'Excitation.'
- It explains the 'dark lines' seen in atomic absorption spectra.
- The electron cannot exist in the 'forbidden zones' between the orbits.
NEET Exam Angle
- Numerical Tip: Energy gaps decrease as you move further from the nucleus. The jump from n=1 to n=2 requires much more energy than the jump from n=2 to n=3.
- Keep an eye out for terms like 'excitation potential'—this is the energy required (in eV) to move an electron from the ground state to an excited state.
05Postulate IV: Photon Emission and the Origin of Hydrogen Spectra
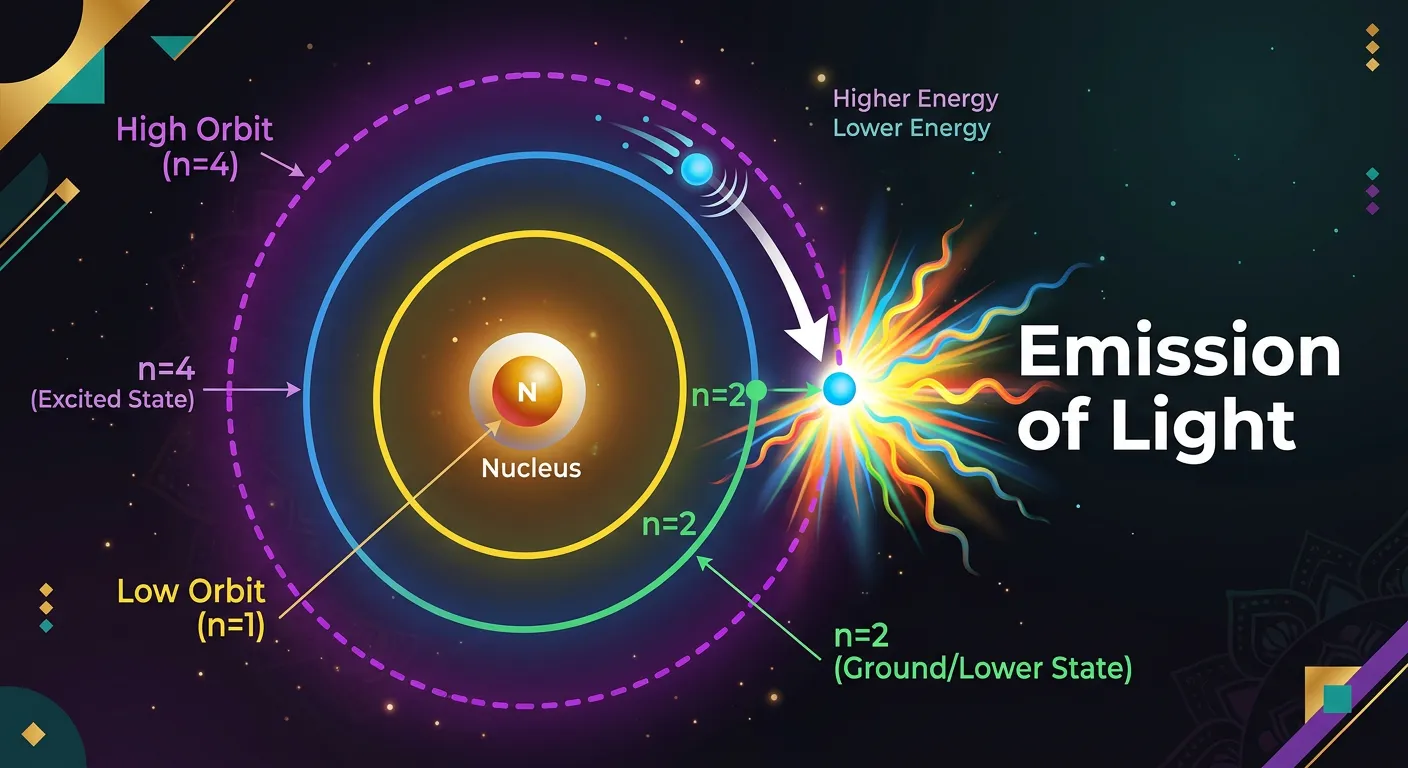
“When the electron drops back to a lower energy state, it releases that extra energy as a photon of light. The energy difference between the orbits determines the color of the light emitted. This is the secret behind the beautiful spectra we observe in the lab!”
What goes up must come down. When an electron is in an excited state, it is inherently unstable. It eventually 'falls' back to a lower energy state—a process called de-excitation. But according to the Law of Conservation of Energy, that energy can't just vanish. Bohr’s fourth postulate states that when an electron drops from a higher orbit to a lower one, it releases the energy difference as a single photon of electromagnetic radiation.
The energy of this emitted photon is given by Planck's equation: $E = h\nu = hc/\lambda$. By setting this equal to the energy difference between the shells ($\Delta E = E_{higher} - E_{lower}$), Bohr could predict the exact frequency ($\nu$) and wavelength ($\lambda$) of light emitted. This was the 'Eureka!' moment for the model. It perfectly explained the 'line spectra' of hydrogen—those beautiful, discrete colors that scientists had seen for years but couldn't explain.
Because the energy levels are fixed (quantized), the energy jumps are also fixed. Since the energy jumps are fixed, the colors emitted are fixed. This is why every element has a unique 'fingerprint' spectrum. In the hydrogen atom, transitions ending at n=2 produce the visible Balmer series, while transitions ending at n=1 produce the ultraviolet Lyman series. This direct link between electronic transitions and light is the cornerstone of modern spectroscopy.
| Series | Transition to ($n_{lower}$) | Region of Spectrum |
|---|---|---|
| Lyman | n = 1 | Ultraviolet |
| Balmer | n = 2 | Visible |
| Paschen | n = 3 | Infrared |
| Brackett | n = 4 | Infrared |
| Pfund | n = 5 | Infrared |
Quick Revision Points
- De-excitation occurs when an electron falls to a lower energy shell.
- The energy difference is emitted as a photon: $\Delta E = h\nu$.
- This postulate explains the origin of emission line spectra.
- Each transition corresponds to a specific line in the hydrogen spectrum.
- Higher energy drops (e.g., n=5 to n=1) result in higher frequency light.
NEET Exam Angle
- You will frequently be asked to calculate the number of spectral lines possible when an electron drops from n to the ground state. Formula: $N = n(n-1)/2$.
- Be careful with the units! Energy is often in electron-volts (eV), while Planck's constant (h) is in Joules. Know the conversion: $1 eV = 1.6 \times 10^{-19} J$.
06Postulate V: Quantization of Angular Momentum and the mvr Equation
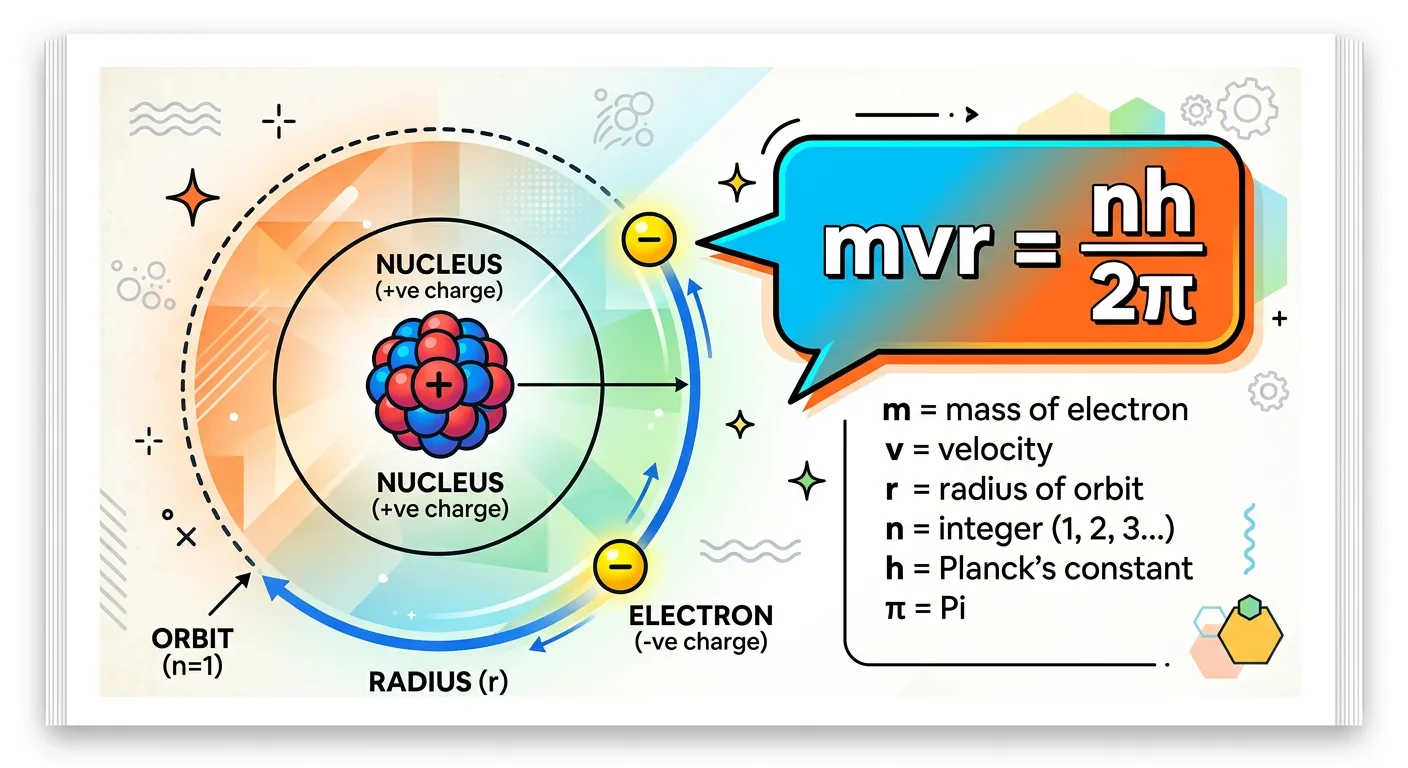
“Bohr added a mathematical rule: angular momentum is quantized! The electron can only move in orbits where its angular momentum, mvr, is an integral multiple of h/2π. This elegant equation acts as the gatekeeper for where an electron is actually allowed to exist.”
The fifth and perhaps most famous postulate is the mathematical 'gatekeeper.' Bohr needed a way to explain why only certain radii were allowed. He proposed that the angular momentum of an electron is quantized. In simple terms, he said an electron can only exist in an orbit where its angular momentum ($L$) is an integral multiple of $h/2\pi$ (where $h$ is Planck's constant).
The mathematical form is: $mvr = n(h/2\pi)$, where $m$ is the mass of the electron, $v$ is its velocity, $r$ is the radius of the orbit, and $n$ is an integer (1, 2, 3...). This means angular momentum can be $1h/2\pi$, $2h/2\pi$, $3h/2\pi$, but it can never be $1.5h/2\pi$. This restriction on angular momentum is what forces the orbits to exist only at specific distances from the nucleus.
This postulate was purely empirical at first—Bohr chose it because it made the math work. It wasn't until a decade later that Louis de Broglie explained why this must be true (linked to the wave-particle duality of the electron). For your NEET exams, this formula is a goldmine. It connects the physical properties of the electron (mass, velocity, radius) to the quantum world (Planck's constant, n). Whenever you see a question about the 'angular momentum of an electron in the 3rd shell,' this is the equation you need.
| Shell (n) | Angular Momentum ($L = mvr$) |
|---|---|
| n = 1 | $h/2\pi$ or $0.159h$ |
| n = 2 | $2h/2\pi$ or $h/\pi$ |
| n = 3 | $3h/2\pi$ or $1.5h/\pi$ |
| n = 4 | $4h/2\pi$ or $2h/\pi$ |
Quick Revision Points
- Angular momentum ($mvr$) is quantized.
- Only orbits satisfying $mvr = nh/2\pi$ are allowed.
- The value of 'n' must be a positive integer (1, 2, 3...).
- This quantization explains why electrons are restricted to specific radii.
- 'h' is Planck’s constant ($6.626 \times 10^{-34} J\cdot s$).
NEET Exam Angle
- A recurring MCQ asks for the angular momentum in the 'L shell' or 'M shell.' Don't forget to translate the letter to 'n' first! (L means n=2, so $L = 2h/2\pi = h/\pi$).
- Note that in modern quantum mechanics, the angular momentum formula is actually $\sqrt{l(l+1)}h/2\pi$, but for Bohr Model questions, always stick to the $nh/2\pi$ formula unless orbital quantum numbers (l) are mentioned.
07Synthesizing Bohr’s Model: NEET Readiness and Legacy
“To wrap up: fixed orbits, quantized energy, and specific transitions. Bohr's model perfectly explained the hydrogen spectrum and laid the foundation for quantum mechanics. Keep this structure in your mind, and you'll ace those NEET physics and chemistry questions on atomic structure with ease!”
To master the Bohr model for NEET, you need to see it as a complete package built on three pillars: fixed orbits (Postulate 1 & 2), energy jumps (Postulate 3 & 4), and quantized momentum (Postulate 5). The model was a massive success because it allowed scientists to calculate the Rydberg constant from first principles and precisely predict the wavelengths of light emitted by hydrogen. It turned atomic physics from a guessing game into a precise science.
However, as a NEET aspirant, you must also be aware of where Bohr’s model falls short. It is a 'one-electron' model. It works perfectly for H, $He^+$, $Li^{2+}$, and $Be^{3+}$, but it fails completely for multi-electron atoms like Helium ($He$) or Carbon ($C$). It doesn't explain the 'fine structure' of spectral lines (when one line appears as two under a powerful microscope) or the Zeeman effect (splitting of lines in a magnetic field). It also treats the electron as a particle moving in a well-defined path, which violates Heisenberg's Uncertainty Principle.
When preparing for the exam, don't just memorize the postulates—understand the 'why' behind them. Use the 'lift' analogy for energy jumps and the 'stadium tracks' for stationary orbits. This mental scaffolding will help you tackle complex numericals on energy transitions and angular momentum with confidence. The Bohr model may be an 'old' model, but it is the primary gateway to the modern quantum mechanical model you will study next.
Quick Revision Points
- Successes: Explained atomic stability and the hydrogen line spectrum.
- Successes: Calculated the Rydberg constant and radius of the H-atom ($0.529$ Å).
- Limitations: Fails for multi-electron atoms (cannot explain electron-electron repulsion).
- Limitations: Does not account for the wave nature of electrons or the Uncertainty Principle.
- Limitations: Cannot explain the Zeeman effect or the Stark effect.
NEET Exam Angle
- Frequently tested: Identify which species Bohr's model applies to. Look for 'Hydrogenic' species (single electron only).
- Strategy: If a question asks for the 'shortest wavelength' in a series, it corresponds to the transition from $n = \infty$ to the base shell of that series (the highest energy drop).
- Strategy: If a question asks for the 'longest wavelength,' it's the smallest energy drop (e.g., n=3 to n=2 for the Balmer series).
Recommended Reading
Explore related Biology topics to build deeper chapter connections for NEET.
- Nature of Electromagnetic Radiation · Topic 2.1
- Quantum Mechanical Model · Topic 2.12
- Atomic Orbitals · Topic 2.13
- Significance of Quantum Numbers · Topic 2.16
- Photoelectric Effect · Topic 2.2
- de Broglie's Relationship · Topic 2.9
- Jump to Key Terms (Quick Revision)
- Review Common NEET Mistakes
- Read Topic FAQs
- Check PYQ Pattern Notes
- Practice NEET MCQs
- Solve NEET PYQs
📚 Key Terms
⚠️ Common NEET Mistakes
- 1Thinking 'stationary' means the electron is not moving; it actually means the energy level is constant.
- 2Applying Bohr's formulas to multi-electron atoms like He, O, or Fe (it only works for 1-electron systems).
- 3Confusing 'n=2' with the '2nd excited state'; n=2 is actually the 1st excited state.
- 4Forgetting that energy is absorbed when moving to a higher n and emitted when moving to a lower n.
- 5Using the wrong units for Planck's constant (h) or forgetting to convert eV to Joules in numericals.
📝 NEET PYQ Pattern
NEET questions from 2018–2024 frequently focus on the calculation of angular momentum (mvr = nh/2π) and the energy associated with transitions between levels (n1 to n2). A recurring pattern involves identifying which transitions produce the shortest/longest wavelength in the hydrogen spectrum, or identifying 'hydrogenic' species (like He+, Li2+) to which Bohr's model applies.
❓ Frequently Asked Questions
What was the main reason Bohr's model was preferred over Rutherford's model?
Bohr's model was preferred because it explained the stability of the atom and the discrete nature of the hydrogen spectrum, both of which Rutherford's model failed to do according to classical electromagnetic theory.
Why does an electron in a stationary orbit not lose energy according to Bohr?
Bohr postulated that stationary orbits are 'non-radiating' states. In these specific orbits, the classical laws of electromagnetism do not apply, allowing the electron to maintain constant energy without spiraling into the nucleus.
What is the mathematical condition for the quantization of angular momentum?
The condition is that the angular momentum (L = mvr) must be an integral multiple of h/2π, expressed as mvr = nh/2π, where n is a positive integer (1, 2, 3...).
Can the Bohr model be applied to calculate the properties of a Lithium (Li) atom?
No, it cannot be applied to a neutral Lithium atom (3 electrons). It only applies to 'hydrogen-like' species which have exactly one electron, such as the Li2+ ion.
How does the energy difference between two orbits relate to the wavelength of light emitted?
The energy difference (ΔE) is inversely proportional to the wavelength (λ). According to the formula ΔE = hc/λ, a larger energy jump produces light with a shorter wavelength (higher frequency).
What happens to the radius of the orbit as the principal quantum number 'n' increases?
The radius (r) increases proportionally to the square of n (r ∝ n²). This means the orbits get significantly further away from the nucleus as the shell number increases.
Written By
NEET Content Strategist & Biology Expert
Sangita Kumari is a NEET educator and content strategist with over 6 years of experience teaching Biology, Chemistry, and Physics to Class 11 and 12 aspirants. She helps bridge the gap between traditional NCERT preparation and modern AI-powered learning. Her content is trusted by thousands of NEET aspirants across India.