🎬 Video Lesson Available
Watch the full 7-slide video lesson for Significance of Quantum Numbers with AI teacher narration and visual explanations.
01The Quantum Map: Defining the Electron's Stadium Address
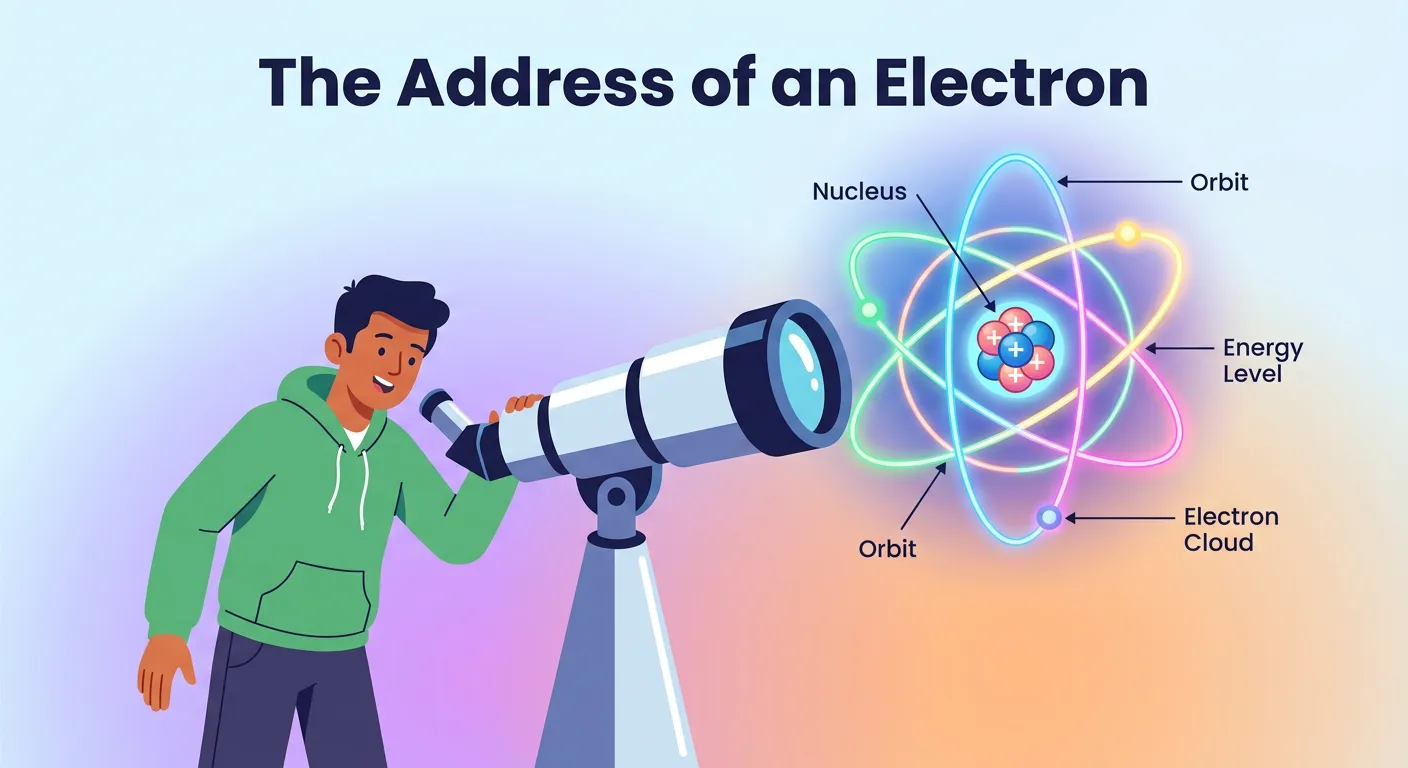
“Welcome! Imagine finding a friend in a massive stadium. To locate them, you need the gate, block, row, and seat. In chemistry, quantum numbers are exactly that—they provide the precise address of an electron within an atom. Let's decode this electronic map together!”
Transitioning from the classical Bohr model-postulates-neet-chemistry)-postulates-neet-chemistry) to the quantum mechanical model of the atom is like moving from a simple 2D drawing to a complex 3D virtual reality. In the Bohr model, we imagined electrons moving in fixed, circular orbits like planets around a sun. However, Heisenberg’s Uncertainty Principle and de Broglie’s hypothesis changed everything. We realized we couldn’t pinpoint an electron's exact path; instead, we talk about 'orbitals'—regions in 3D space where the probability of finding an electron is highest. To describe these regions and the electrons within them, we use four specific coordinates known as quantum numbers. These aren't just arbitrary digits; they are the mathematical solutions derived from the Schrödinger Wave Equation.
Think of the atom as a massive stadium. If you want to find a friend (the electron), you need their gate number, block, row, and seat. Without all four pieces of information, your search would be inefficient or impossible. In the atomic world, these coordinates represent the shell, subshell, orbital, and the intrinsic spin. By understanding the significance of quantum numbers, you aren't just memorizing values; you are learning the language of electron density and chemical reactivity. This 'electronic address' defines everything from why an atom bonds to how it behaves in a magnetic field.
Quick Revision Points
- Quantum numbers arise from the wave-like properties of matter described by Schrödinger.
- There are four quantum numbers: Principal (n), Azimuthal (l), Magnetic (ml), and Spin (ms).
- The first three quantum numbers define the orbital, while the fourth defines the electron itself.
- This framework replaces Bohr's deterministic 'orbits' with probabilistic 'orbitals'.
- Quantum numbers are essential for understanding the stability and electronic configuration of elements.
NEET Exam Angle
- NEET frequently asks conceptual questions regarding the difference between an 'orbit' (2D) and an 'orbital' (3D).
- Remember: The first three quantum numbers (n, l, ml) are derived from the Schrödinger wave equation, but the Spin quantum number (ms) was added later to explain spectral doublets and magnetism.
- Direct questions often involve identifying which set of coordinates describes a specific region of electron density.
02Principal Quantum Number (n): Defining Shell Size and Energy
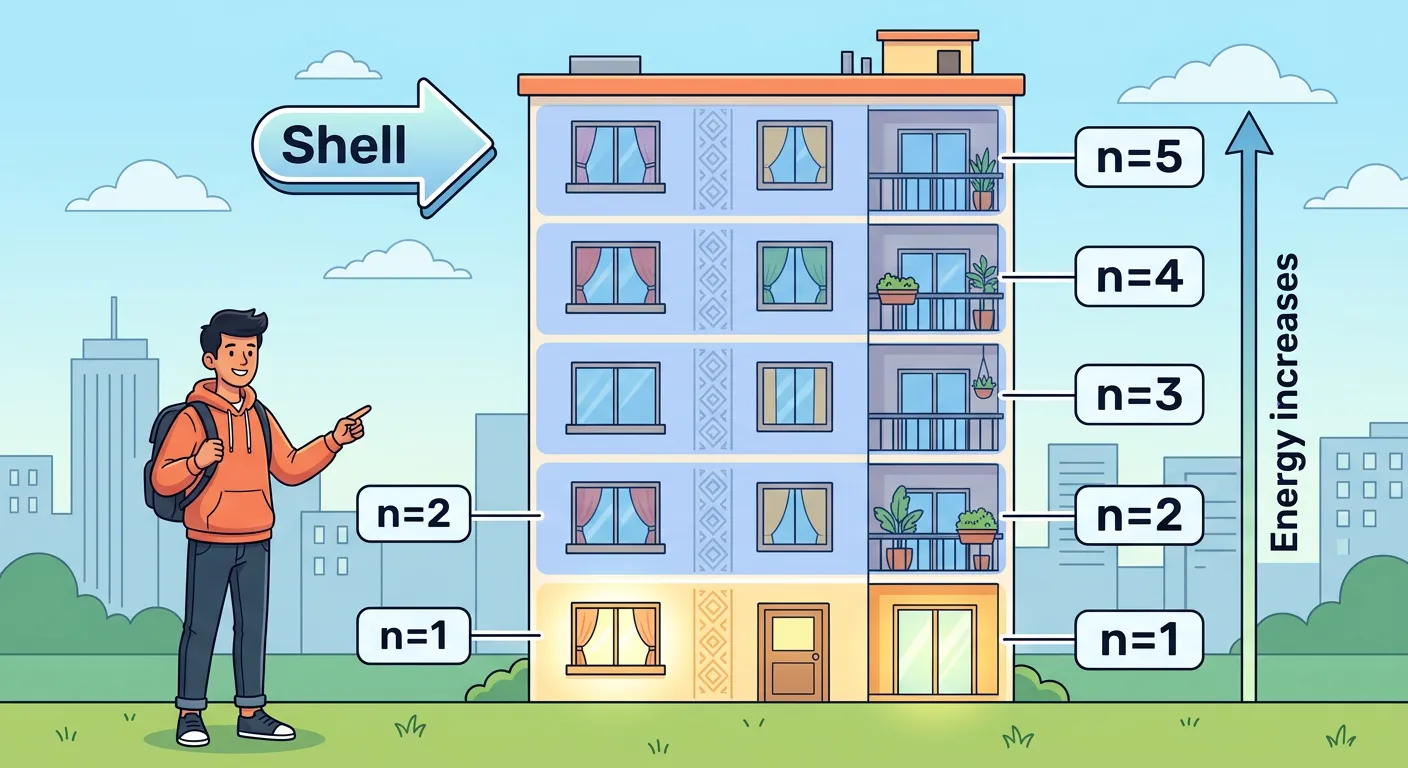
“First, the Principal Quantum Number 'n'. Think of it as the floor number in an apartment building. As 'n' increases, the floor gets higher, the distance from the nucleus grows, and the energy level rises. It tells us the size of the electron's shell.”
The Principal Quantum Number, denoted by 'n', is the primary indicator of an electron's energy level and its average distance from the nucleus. Think of 'n' as the main floor number in an atomic apartment complex. It can take any positive integer value (n = 1, 2, 3...). As the value of 'n' increases, the electron moves further away from the nucleus, and the shell size expands. This is why a 3s orbital is much larger than a 1s orbital. In terms of energy, as 'n' increases, the electrostatic attraction between the nucleus and the electron weakens, meaning the electron exists at a higher (less negative) energy state.
For a single-electron system like Hydrogen, the energy depends solely on 'n'. However, in multi-electron atoms, 'n' works alongside 'l' to determine the overall energy level. One of the most important mathematical rules to remember for NEET is the relationship between 'n' and the maximum number of electrons: a shell can hold up to 2n² electrons. Furthermore, 'n' corresponds directly to the periods in the Modern Periodic Table. If an element’s valence electron is in the n=4 shell, you will find that element in the fourth period. Understanding 'n' is the first step in predicting atomic size and ionization enthalpy trends.
Quick Revision Points
- 'n' designates the main shell (K, L, M, N...) and determines the size of the electron cloud.
- Energy levels: As 'n' increases, energy increases and the shell becomes more distant from the nucleus.
- Maximum number of electrons in a shell = 2n².
- Total number of orbitals in a shell = n².
- The value of 'n' can never be zero or negative.
NEET Exam Angle
- Numerical problems often ask for the total number of orbitals or electrons in a shell (e.g., How many orbitals in the M shell? Answer: 3² = 9).
- Correlation with Periodic Table: Elements in the same period have the same value of 'n' for their outermost shell.
| Shell Letter | n Value | Max Orbitals (n²) | Max Electrons (2n²) |
|---|---|---|---|
| K | 1 | 1 | 2 |
| L | 2 | 4 | 8 |
| M | 3 | 9 | 18 |
| N | 4 | 16 | 32 |
03Azimuthal Quantum Number (l): Decoding Subshells and Orbital Shapes

“Next is the Azimuthal Quantum Number 'l'. If 'n' is the floor, 'l' is the shape of the rooms on that floor. It defines the subshell. Whether it's a simple sphere or a complex clover, 'l' tells us the shape of the orbital where our electron hangs out.”
While the principal quantum number tells us which 'floor' the electron is on, the Azimuthal Quantum Number (also called the Orbital Angular Momentum Quantum Number), denoted by 'l', tells us the shape of the room. It defines the subshell. For any given value of 'n', 'l' can have integer values ranging from 0 to (n-1). Each value of 'l' is associated with a specific letter: 0 for 's', 1 for 'p', 2 for 'd', and 3 for 'f'. These letters originated from spectroscopic terms: Sharp, Principal, Diffuse, and Fundamental.
The physical significance of 'l' is two-fold: it determines the three-dimensional geometric shape of the orbital and identifies the orbital angular momentum of the electron. The formula for orbital angular momentum is L = √[l(l+1)] * (h/2π). This is a favorite for NEET examiners! Geometrically, an 's' orbital (l=0) is a sphere, 'p' (l=1) is a dumbbell, 'd' (l=2) is a double-dumbbell (cloverleaf), and 'f' (l=3) has a complex eight-lobed structure. Because the shape of an orbital dictates how atoms overlap, 'l' is fundamentally responsible for the geometry of molecules and chemical bonding.
Quick Revision Points
- 'l' defines the subshell and the 3D shape of the orbital.
- Range: l = 0 to (n-1). If n=3, l can be 0, 1, or 2.
- Orbital notation: l=0 (s), l=1 (p), l=2 (d), l=3 (f).
- Angular momentum calculation: L = √(l(l+1)) h/2π.
- It determines the number of angular nodes (Number of angular nodes = l).
NEET Exam Angle
- Calculation of angular momentum is a high-yield topic. Note that for any 's' orbital, the angular momentum is always zero because l=0.
- Pay attention to 'n' and 'l' relationships. For example, a '2d' orbital is impossible because for n=2, 'l' can only be 0 or 1.
| Subshell Value (l) | Letter Designation | Shape Description | Max Electrons (2(2l+1)) |
|---|---|---|---|
| 0 | s | Spherical | 2 |
| 1 | p | Dumbbell | 6 |
| 2 | d | Double Dumbbell | 10 |
| 3 | f | Complex/Diffuse | 14 |
04Magnetic Quantum Number (ml): Orbital Orientation in Space
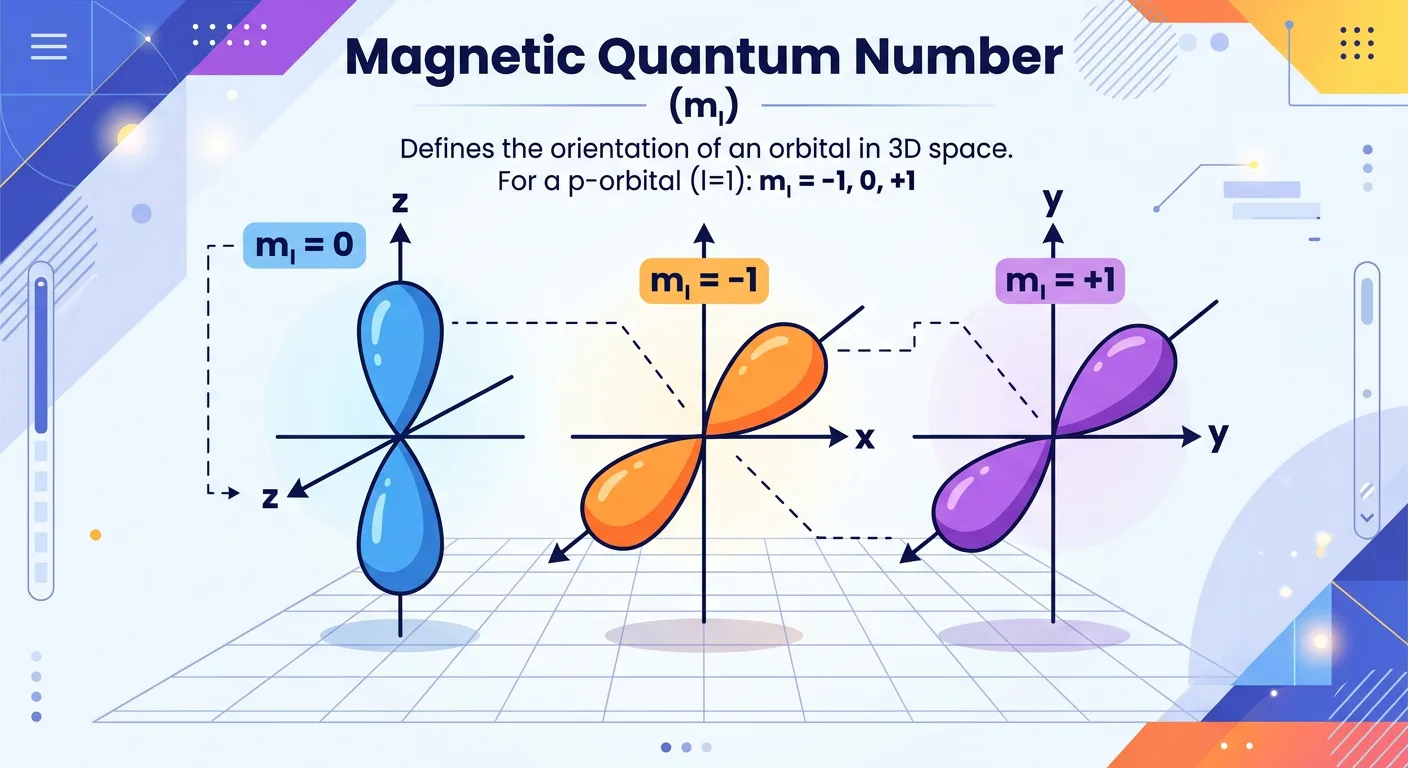
“Meet the Magnetic Quantum Number 'ml'. If 'l' gives us the room shape, 'ml' tells us its orientation in space—like pointing a dumbbell along the x, y, or z-axis. It defines how orbitals are aligned relative to the external magnetic field.”
If the azimuthal quantum number tells us the shape of the 'room' (subshell), the Magnetic Quantum Number (ml) tells us how that room is oriented in three-dimensional space relative to a set of Cartesian axes (x, y, z). This number becomes particularly significant when the atom is placed in an external magnetic field. Under such conditions, orbitals that previously had the same energy (degenerate orbitals) might split into different energy levels—a phenomenon known as the Zeeman Effect.
The possible values of ml depend entirely on 'l'. For a given value of 'l', ml can range from -l to +l, including zero. This means there are (2l + 1) possible orientations. For example, if l=1 (p-subshell), ml can be -1, 0, or +1. These three values correspond to the px, py, and pz orbitals. In the absence of an external magnetic field, these three orbitals have exactly the same energy and are called 'degenerate'. This concept is crucial for understanding how coordination compounds split their d-orbitals and how atoms interact during chemical reactions. It provides the spatial alignment necessary for hybridization and directional bonding.
Quick Revision Points
- 'ml' determines the number of orbitals in a subshell and their spatial orientation.
- Number of orbitals per subshell = 2l + 1.
- Range: -l through 0 to +l.
- Degenerate orbitals: Orbitals with the same 'n' and 'l' values that possess identical energy in a field-free environment.
- Explains the Zeeman effect (splitting of spectral lines in a magnetic field).
NEET Exam Angle
- Questions often ask to identify the number of orbitals for a given 'l' value (e.g., how many orientations for l=2? Answer: 2(2)+1 = 5).
- Understand that for the d-subshell (l=2), the five orbitals are dxy, dyz, dxz, dx²-y², and dz². Each corresponds to a specific ml value.
- A common trick question: "How many orbitals have n=3, l=1?" The answer is 3 (corresponding to ml = -1, 0, +1).
05Spin Quantum Number (ms): The Intrinsic Rotation of Electrons
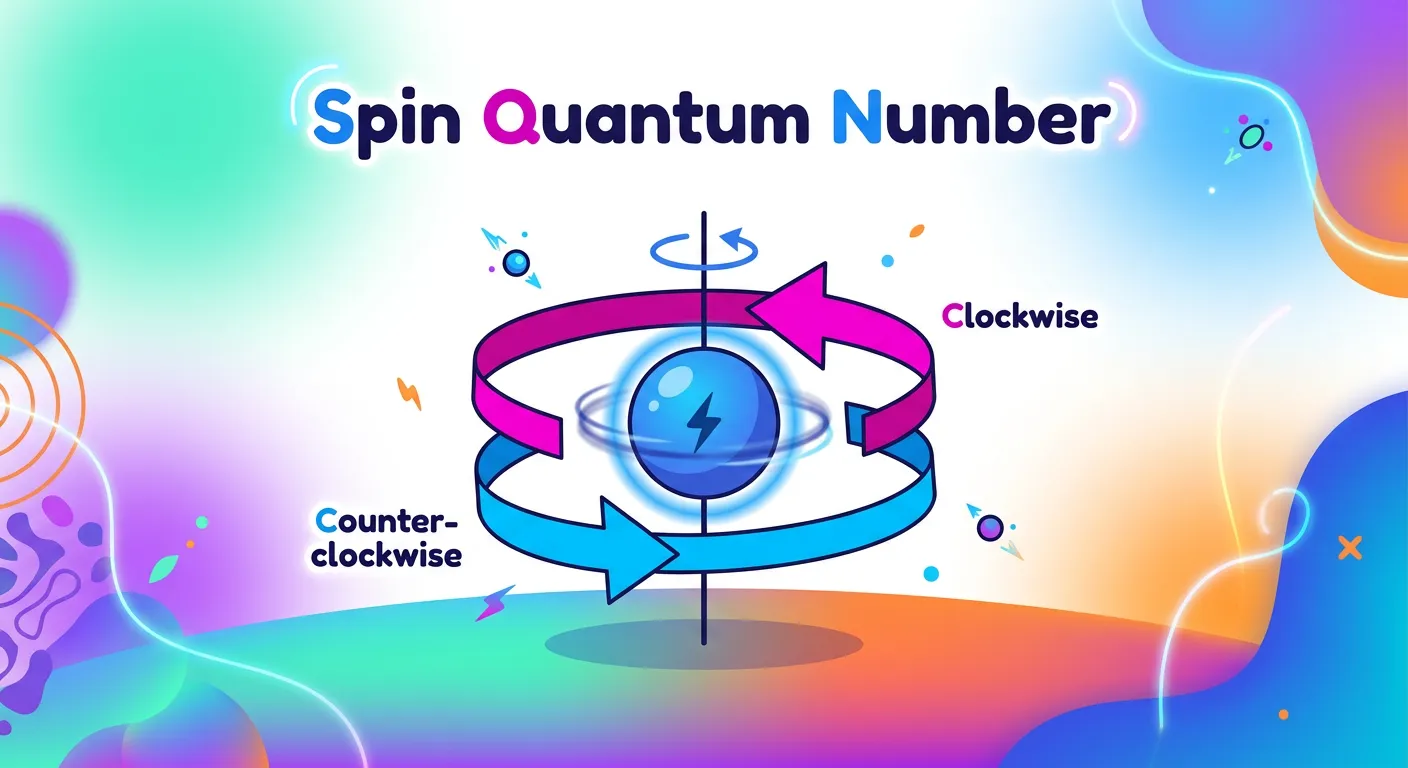
“Finally, the Spin Quantum Number 'ms'. Think of an electron as a tiny spinning top. It can rotate clockwise or counter-clockwise. Represented as plus or minus one-half, this tells us the intrinsic angular momentum of the electron. It is the final piece of our address puzzle!”
The first three quantum numbers describe the 'house' (orbital), but the Spin Quantum Number (ms) describes the 'occupant' (the electron). Long after Schrödinger's equation was developed, scientists noticed that certain spectral lines appeared as 'doublets' (two closely spaced lines). This led to the discovery that an electron behaves as if it is spinning on its own axis, much like the Earth rotates as it orbits the Sun. This spin generates a tiny magnetic field, giving the electron an intrinsic magnetic moment.
There are only two possible values for 'ms': +1/2 (represented as an upward arrow ↑, often called 'spin up' or clockwise) and -1/2 (represented as a downward arrow ↓, 'spin down' or counter-clockwise). This quantum number is the foundation of Pauli’s Exclusion Principle, which states that no two electrons in the same atom can have the same set of all four quantum numbers. If two electrons share the same orbital (meaning they have identical n, l, and ml), they must have opposite spins. This property explains why elements are paramagnetic (having unpaired electrons) or diamagnetic (all electrons paired). It is also the reason why each orbital can accommodate exactly two electrons.
Quick Revision Points
- 'ms' describes the intrinsic spin of an electron.
- Values: +1/2 and -1/2 only.
- Spin is a non-classical property; it was not derived from Schrödinger’s wave equation.
- Pauli’s Exclusion Principle: An orbital can hold a maximum of 2 electrons with opposite spins.
- Magnetic properties: Paramagnetism arises from unpaired spins; diamagnetism from paired spins.
NEET Exam Angle
- Remember: The Spin Quantum Number is the only one that doesn't depend on the values of n, l, or ml.
- Direct questions often ask for the 'spin magnetic moment' using the formula: μ = √[n(n+2)] Bohr Magnetons, where 'n' is the number of unpaired electrons.
| Quantum Property | Symbol | Physical Significance |
|---|---|---|
| Principal | n | Size and Energy |
| Azimuthal | l | Shape |
| Magnetic | ml | Orientation |
| Spin | ms | Direction of Spin |
06Predictive Power: Electronic Configuration and Reactivity
“Why bother? Because these numbers dictate chemistry! They explain the periodic table, predict how atoms bond, and help us understand why some elements are reactive while others are chill. Mastering these is your shortcut to acing those tricky electronic configuration questions in NEET!”
The true significance of quantum numbers lies in their predictive power. By combining these four numbers, we can write the electronic configuration of any element, which is essentially the roadmap of its chemical personality. For example, the (n+l) rule (Aufbau Principle) uses these numbers to determine the order in which orbitals are filled. The lower the value of (n+l), the lower the energy of the orbital. If two orbitals have the same (n+l) value, the one with the lower 'n' is filled first. This explains why the 4s orbital is filled before the 3d orbital, a common point of confusion for students.
Furthermore, quantum numbers help us understand the exceptional stability of half-filled and fully-filled subshells (like Chromium and Copper). When subshells are exactly half-full (e.g., d5) or completely full (e.g., d10), the distribution of charge is symmetrical and exchange energy is maximized, leading to lower total energy. For NEET, you must be able to spot 'forbidden' sets of quantum numbers immediately. Any set where l ≥ n, or |ml| > l, is physically impossible. Mastering these constraints allows you to solve configuration problems quickly and accurately, forming the basis for Inorganic Chemistry.
Quick Revision Points
- (n+l) Rule: Determines the sequence of orbital filling based on energy levels.
- Symmetry and Stability: Half-filled and fully-filled subshells are extra stable due to symmetry and exchange energy.
- Invalid Sets: Identifying impossible combinations (e.g., n=2, l=2 is impossible).
- Quantum numbers dictate the valence shell, which determines an atom’s valency and bonding capacity.
NEET Exam Angle
- A recurring NEET pattern: "Which of the following sets of quantum numbers is not allowed?"
- Check for: 1) Is l < n? 2) Is |ml| ≤ l? 3) Is ms = ±1/2?
- Example: n=3, l=3 is invalid. n=2, l=1, ml=2 is invalid.
| Set Example | n | l | ml | Valid? | Reason |
|---|---|---|---|---|---|
| Set A | 3 | 2 | 0 | Yes | l < n, ml is within range |
| Set B | 2 | 2 | 1 | No | l cannot be equal to n |
| Set C | 1 | 0 | 1 | No | ml cannot be greater than l |
| Set D | 4 | 0 | 0 | Yes | Valid 4s orbital |
07Mastering the Foundation: Quantum Mechanics for the NEET Aspirant

“You have just decoded the electron's secret address! Remember, these four numbers are the foundation of quantum mechanics. Practice visualizing these shapes and you'll easily conquer any complex chemistry problem. Keep studying, stay curious, and get ready to crush that NEET exam!”
As we conclude our exploration of quantum numbers, it is vital to synthesize these concepts into a cohesive mental model. You are no longer looking at an atom as a solid ball or a solar system, but as a dynamic cloud of probability defined by mathematical parameters. Each electron occupies a state characterized by its unique set of four quantum numbers. This understanding is the gateway to advanced topics like Molecular Orbital Theory (MOT) and organic reaction mechanisms. If you can visualize the 'p' orbital's dumbbell shape and its orientation along the x-axis, you can easily understand why pi-bonds form through lateral overlap.
To master this for NEET, focus on the visualization of nodes. Remember that a node is a region where the probability of finding an electron is zero. The number of radial nodes is given by (n - l - 1), and the number of angular nodes is simply 'l'. The total number of nodes is (n - 1). When practicing NCERT-based problems, always verify your quantum sets against these formulas. This conceptual clarity will transform Physical Chemistry from a subject of rote memorization into a logical puzzle that you can solve with confidence. Keep practicing, stay curious about the invisible world of the atom, and use these 'atomic addresses' to navigate your way to a top NEET score.
Quick Revision Points
- Radial Nodes formula: n - l - 1.
- Angular Nodes formula: l.
- Total Nodes: n - 1.
- Probability density (ψ²) is highest where the 'electron cloud' is most dense.
- All 4 quantum numbers are necessary to uniquely identify an electron in an atom.
NEET Exam Angle
- Node calculations are extremely common. Example: For a 3p orbital, n=3 and l=1. Radial nodes = 3-1-1 = 1. Angular nodes = 1.
- Focus on matching the quantum number to its physical attribute: n (size), l (shape), ml (orientation), ms (spin).
- Be prepared for 'Assertion-Reason' questions based on the Pauli Exclusion Principle and the limitations of quantum numbers.
Recommended Reading
Explore related Biology topics to build deeper chapter connections for NEET.
- Nature of Electromagnetic Radiation · Topic 2.1
- Quantum Mechanical Model · Topic 2.12
- Atomic Orbitals · Topic 2.13
- Photoelectric Effect · Topic 2.2
- Bohr Model · Topic 2.4
- de Broglie's Relationship · Topic 2.9
- Jump to Key Terms (Quick Revision)
- Review Common NEET Mistakes
- Read Topic FAQs
- Check PYQ Pattern Notes
- Practice NEET MCQs
- Solve NEET PYQs
📚 Key Terms
⚠️ Common NEET Mistakes
- 1Assuming the value of 'l' can be equal to 'n'. Always remember l = 0 to (n-1).
- 2Confusing the number of orbitals (n²) with the number of electrons (2n²).
- 3Thinking that 'ms' depends on the values of 'n', 'l', or 'ml'. Spin is independent.
- 4Forgetting that the orbital angular momentum for any 's' orbital is zero because l=0.
- 5Misidentifying the range of ml. It is -l to +l, not -n to +n.
📝 NEET PYQ Pattern
NEET questions from 2018–2024 frequently involve identifying 'invalid sets' of quantum numbers (e.g., where l ≥ n). There is also a recurring pattern of matching quantum numbers to their specific physical significance (size, shape, orientation, spin).
❓ Frequently Asked Questions
What is the physical significance of the Principal Quantum Number (n)?
The Principal Quantum Number (n) determines the main energy level or shell of an electron, its average distance from the nucleus (size), and the maximum number of electrons that can be accommodated in that shell (2n²).
How does the Azimuthal Quantum Number (l) determine the shape of an orbital?
The value of 'l' (from 0 to n-1) defines the subshell and its geometric shape: l=0 is spherical (s), l=1 is dumbbell (p), l=2 is double-dumbbell (d), and l=3 is complex (f).
Can two electrons in the same atom have the same set of four quantum numbers?
No. According to Pauli's Exclusion Principle, no two electrons in the same atom can have an identical set of all four quantum numbers. They must differ in at least one, usually the Spin Quantum Number (ms).
How do you calculate the total number of orbitals in a shell with principal quantum number 'n'?
The total number of orbitals in a shell is calculated using the formula n². For example, in the M shell (n=3), there are 3² = 9 orbitals (one 3s, three 3p, and five 3d).
What does a negative value for the Magnetic Quantum Number (ml) represent?
A negative value for ml simply represents one of the specific spatial orientations of an orbital along the Cartesian axes relative to a magnetic field; it does not imply negative energy or physical quantity.
Why is the Spin Quantum Number (ms) not derived from the Schrodinger Wave Equation?
The Schrodinger equation describes the wave nature of the electron in space. Spin is an intrinsic property discovered later through experimental evidence (like the Stern-Gerlach experiment) and was incorporated to explain the fine structure of spectra.
Written By
NEET Content Strategist & Biology Expert
Sangita Kumari is a NEET educator and content strategist with over 6 years of experience teaching Biology, Chemistry, and Physics to Class 11 and 12 aspirants. She helps bridge the gap between traditional NCERT preparation and modern AI-powered learning. Her content is trusted by thousands of NEET aspirants across India.