🎬 Video Lesson Available
Watch the full 7-slide video lesson for Atomic Orbitals with AI teacher narration and visual explanations.
01Beyond Bohr’s Orbits: The Quantum Leap to Uncertainty
“Welcome, NEET warriors! Forget everything you knew about circular orbits. Bohr’s tracks were a great start, but electrons aren't just running on a racetrack. They are living in a quantum world of uncertainty! Today, we dive into the mysterious, fuzzy, and exciting world of atomic orbitals.”
For a long time, we imagined atoms as miniature solar systems. Niels Bohr’s model was revolutionary, placing electrons in fixed, circular 'orbits' with specific energies. It worked beautifully for Hydrogen, but as soon as we added more electrons, the math fell apart. The real world of the atom is far more chaotic and fascinating than Bohr’s rigid tracks could ever explain. The turning point came with Werner Heisenberg and his famous Uncertainty Principle. He proved that it is physically impossible to know both the exact position and the exact momentum of an electron simultaneously. If you pin down where it is, you lose track of where it’s going. This single realization destroyed the concept of a 'racetrack' orbit entirely.
In the quantum world, we stop asking 'Where exactly is the electron?' and start asking 'Where is it most likely to be?' This shift from deterministic paths to probabilistic regions marks the birth of the Quantum Mechanical Model. Instead of a dot moving on a line, imagine a buzzing region of energy. The electron isn't just a particle; it behaves like a wave spread out in space. This conceptual leap is the foundation of modern chemistry, and mastering it is your first step toward understanding how atoms truly interact in chemical reactions. This model incorporates the dual nature of matter proposed by de Broglie, suggesting that every moving particle has an associated wave. For NEET, remember that the failure of Bohr’s model wasn't just about multi-electron systems; it was about the fundamental nature of matter at the subatomic level which denies the existence of definite trajectories.
Quick Revision Points
- Bohr’s model viewed electrons as particles in fixed 2D circular paths called orbits.
- Heisenberg’s Uncertainty Principle states that $\Delta x \cdot \Delta p \geq h/4\pi$, invalidating precise trajectories.
- The Quantum Mechanical Model treats electrons as three-dimensional wave functions.
- Transitioned from 'certainty' (orbits) to 'probability' (orbitals).
- Bohr's model failed to account for the wave-particle duality of electrons.
NEET Exam Angle
- Direct questions often ask why Bohr’s model was discarded; the answer is usually linked to the Heisenberg Uncertainty Principle and wave nature.
- Be prepared to calculate uncertainty in position or velocity using the formula $\Delta x \cdot m\Delta v \geq h/4\pi$.
- Understand that 'trajectory' is a classical concept that does not exist in quantum mechanics.
02Schrödinger’s Equation: The Wave Function as a Mathematical Guide

“Think of an electron like a naughty kid playing hide and seek. You can't pin them down! Because electrons show wave-particle duality, we use the Schrödinger equation to describe them. It treats the electron as a wave function, giving us the probability of where it hides.”
To describe the behavior of electrons, Erwin Schrödinger developed a complex mathematical equation that treats electrons as standing waves. This is known as the Schrödinger Wave Equation. When we 'solve' this equation for an atom, we get a set of solutions called wave functions, denoted by the Greek letter Psi ($\psi$). On its own, $\psi$ doesn't represent a physical entity—it is simply a mathematical function that contains all the information about an electron in an atom. Think of it as the 'DNA' of an electron's state. It tells us about the energy levels and the spatial distribution, but it lacks a direct physical meaning in our everyday 3D world.
The solutions to the Schrödinger equation are quantized, meaning only certain energy levels are allowed. These solutions are characterized by three quantum numbers ($n, l, m_l$) which describe the size, shape, and orientation of the orbitals. The real magic happens when we square the wave function: $\psi^2$. This is known as the Probability Density. Max Born interpreted $\psi^2$ at any point as the probability of finding the electron at that specific location. If $\psi^2$ is high, there is a high chance of spotting the electron; where $\psi^2$ is zero, the electron will never be found. This mathematical framework allows us to map out 'clouds' of electron density. It is important to distinguish between the wave function (which can be positive or negative) and the probability (which is always positive because of the square). This distinction is a classic 'trap' in NEET theory questions.
Quick Revision Points
- The Schrödinger Equation $(\hat{H}\psi = E\psi)$ is the fundamental equation of quantum mechanics.
- $\psi$ (Psi) is a one-electron wave function representing an orbital.
- $\psi$ has no physical significance; it can have positive, negative, or zero values.
- $\psi^2$ (Probability Density) represents the likelihood of finding an electron in a unit volume.
- Solutions to the Schrödinger equation provide the quantum numbers $n, l,$ and $m_l$.
NEET Exam Angle
- Common Question: What is the physical significance of $\psi$? (Answer: None, but $\psi^2$ is the probability density).
- Understand that the Schrödinger equation was solved exactly for Hydrogen, but approximations are needed for multi-electron atoms.
- Pay attention to 'nodes'—points where $\psi$ crosses zero and $\psi^2$ is zero.
03Defining the Atomic Orbital: 3D Probability Density Clouds
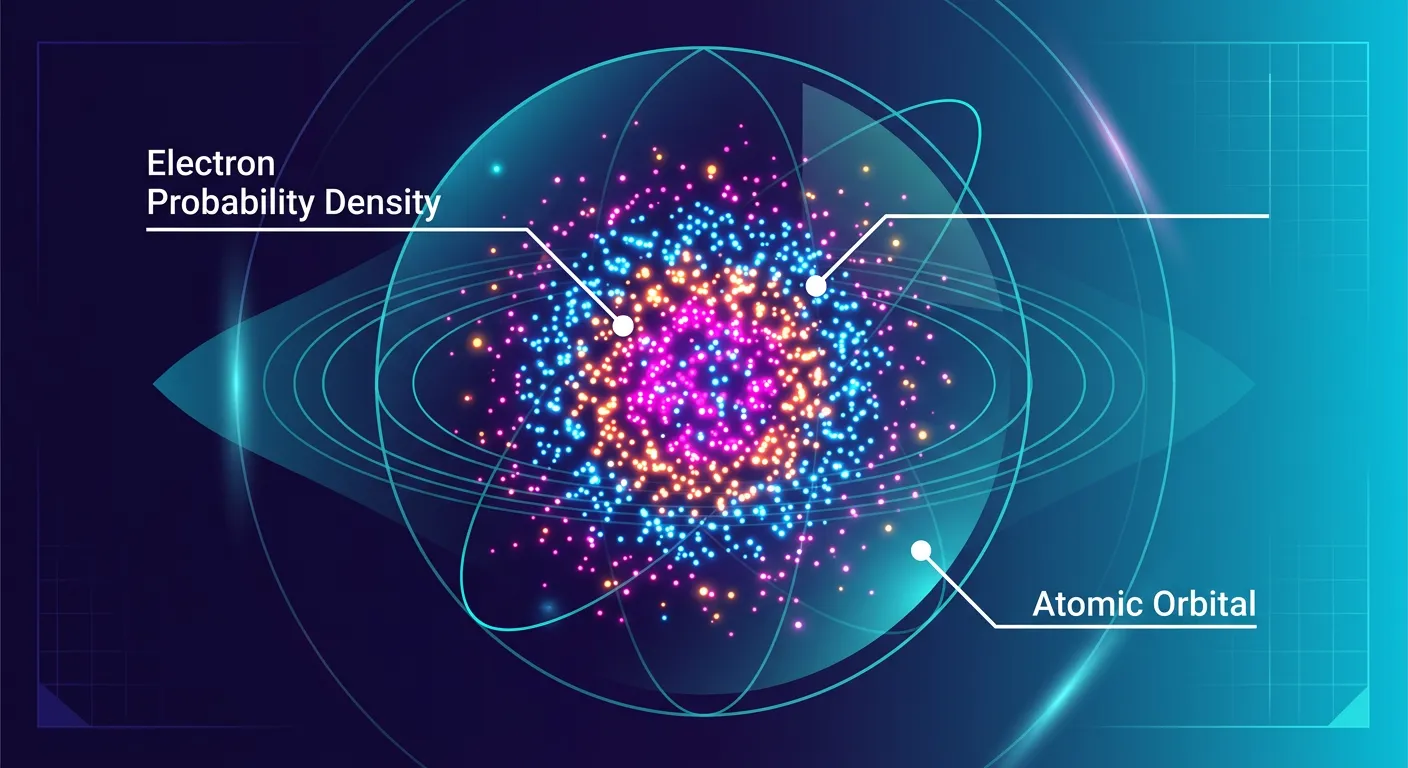
“An atomic orbital is simply the region in space where the probability of finding an electron is maximum—about 90 to 95 percent! It is not a solid shell, but a misty, three-dimensional cloud. It is where our electron prefers to spend its time.”
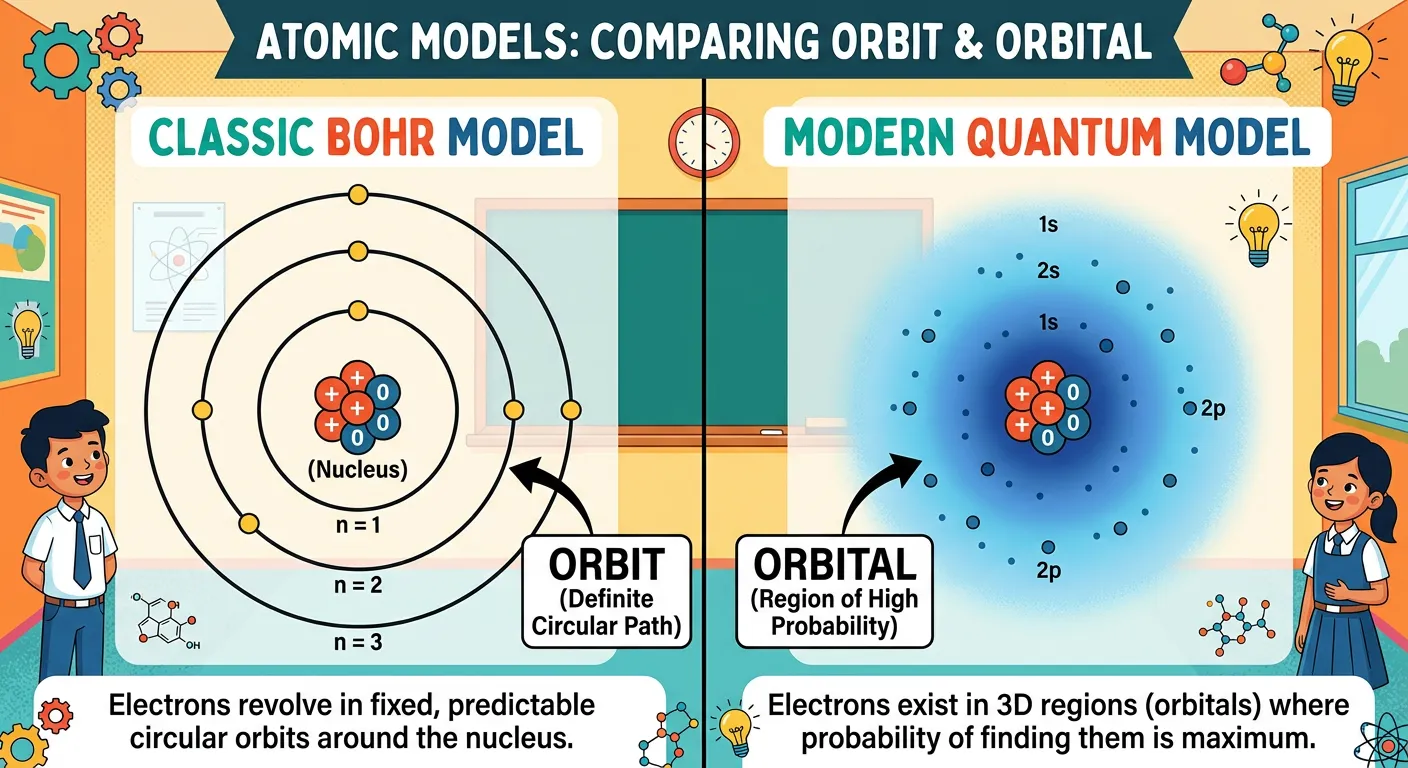
Now that we know we can't track an electron's exact path, how do we visualize it? We use the concept of an 'Atomic Orbital.' Formally, an orbital is a three-dimensional region in space around the nucleus where the probability of finding an electron is maximum—typically defined as 90 to 95 percent. Why not 100 percent? Because theoretically, there is a tiny, non-zero probability of finding an electron even a mile away from the nucleus! We draw boundary surface diagrams to represent these regions, which look like misty clouds or solid shapes. These diagrams represent a volume where the electron spends the vast majority of its time.
One of the most important things for a NEET aspirant is to distinguish between an 'Orbit' and an 'Orbital.' An orbit is a flat, 2D circle with a fixed radius where the electron's position is certain (a Bohr concept). An orbital is a 3D volume where the electron's position is a matter of chance, governed by wave functions. While an orbit can hold a specific number of electrons based on $2n^2$, a single orbital can never hold more than two electrons, and even then, they must have opposite spins according to the Pauli Exclusion Principle. This distinction is fundamental to understanding electronic configurations, bond lengths, and chemical bonding later on. In essence, while orbits were specific paths, orbitals are the 'rooms' within the electronic shells where electrons reside.
| Feature | Orbit (Bohr) | Orbital (Quantum) |
|---|---|---|
| Dimension | 2D Circular Path | 3D Space/Region |
| Concept | Deterministic/Certain | Probabilistic/Uncertain |
| Shape | Always circular/elliptical | Spherical, dumbbell, cloverleaf |
| Heisenberg | Violates Uncertainty Principle | Consistent with Uncertainty Principle |
| Electron Capacity | $2n^2$ electrons | Maximum 2 electrons |
Quick Revision Points
- Orbitals are 'electron density' clouds where findability is 90-95%.
- Boundary surface diagrams represent the shape of these orbitals.
- Orbital size and shape are determined by quantum numbers $n$ and $l$.
- An orbital is a mathematical 'one-electron wave function'.
NEET Exam Angle
- Identify the difference between 2D orbits and 3D orbitals in multiple-choice questions.
- Focus on the fact that the probability of finding an electron at an infinite distance from the nucleus is never zero.
- Remember that the 'boundary surface' is where the probability density $\psi^2$ is constant.
04Spherical Symmetry: Characteristics of s-Orbitals

“Meet the s-orbital! It is perfectly spherical, like a cricket ball. No matter which angle you look from, the electron density is uniform. It is the simplest room an electron can occupy. Remember, the larger the principal quantum number, the bigger the ball!”
The simplest type of orbital is the s-orbital, corresponding to the azimuthal quantum number $l=0$. These orbitals are perfectly spherical. This means that the probability of finding an electron is the same in all directions at a given distance from the nucleus. Because of this, s-orbitals are called non-directional. As the principal quantum number ($n$) increases, the size of the s-orbital increases, which also means the electron is likely to be found further from the nucleus. A $2s$ orbital is larger than a $1s$ orbital, and a $3s$ orbital is even larger. This increase in size also corresponds to an increase in potential energy as the electron escapes the tight pull of the nucleus.
Inside these spheres, we find fascinating regions called 'nodes.' A node is a space where the probability of finding an electron is zero ($\psi^2 = 0$). For s-orbitals, we deal primarily with radial nodes (also called spherical nodes). The number of radial nodes in any orbital is calculated using the formula $(n - l - 1)$. Since for s-orbitals $l=0$, the number of radial nodes is simply $(n-1)$. So, a $1s$ orbital has zero nodes, while a $2s$ orbital has one radial node—a region like the gap in a nested doll where the electron cannot exist. As $n$ increases, the number of these nodes also increases, creating a complex internal structure of probability peaks and valleys within the orbital sphere.
| Orbital | $n$ | $l$ | Radial Nodes $(n-l-1)$ | Angular Nodes $(l)$ | Total Nodes $(n-1)$ |
|---|---|---|---|---|---|
| 1s | 1 | 0 | 0 | 0 | 0 |
| 2s | 2 | 0 | 1 | 0 | 1 |
| 3s | 3 | 0 | 2 | 0 | 2 |
| 4s | 4 | 0 | 3 | 0 | 3 |
Quick Revision Points
- s-orbitals are spherically symmetrical and non-directional.
- The probability density $\psi^2$ is uniform across all axes ($x, y, z$).
- The size and energy of s-orbitals increase as $n$ increases.
- The number of radial nodes for an s-orbital is $(n - 1)$.
- $1s$ is the only orbital with no nodes at all.
NEET Exam Angle
- NEET loves questions on node calculation. Memorize the formula: Total Nodes $= n - 1$.
- Understand that s-orbitals are the most penetrating because of their spherical shape near the nucleus.
- Identify s-orbitals by their boundary surface diagrams which look like solid balls.
05Directional Geometry: Understanding px, py, and pz Dumbbells
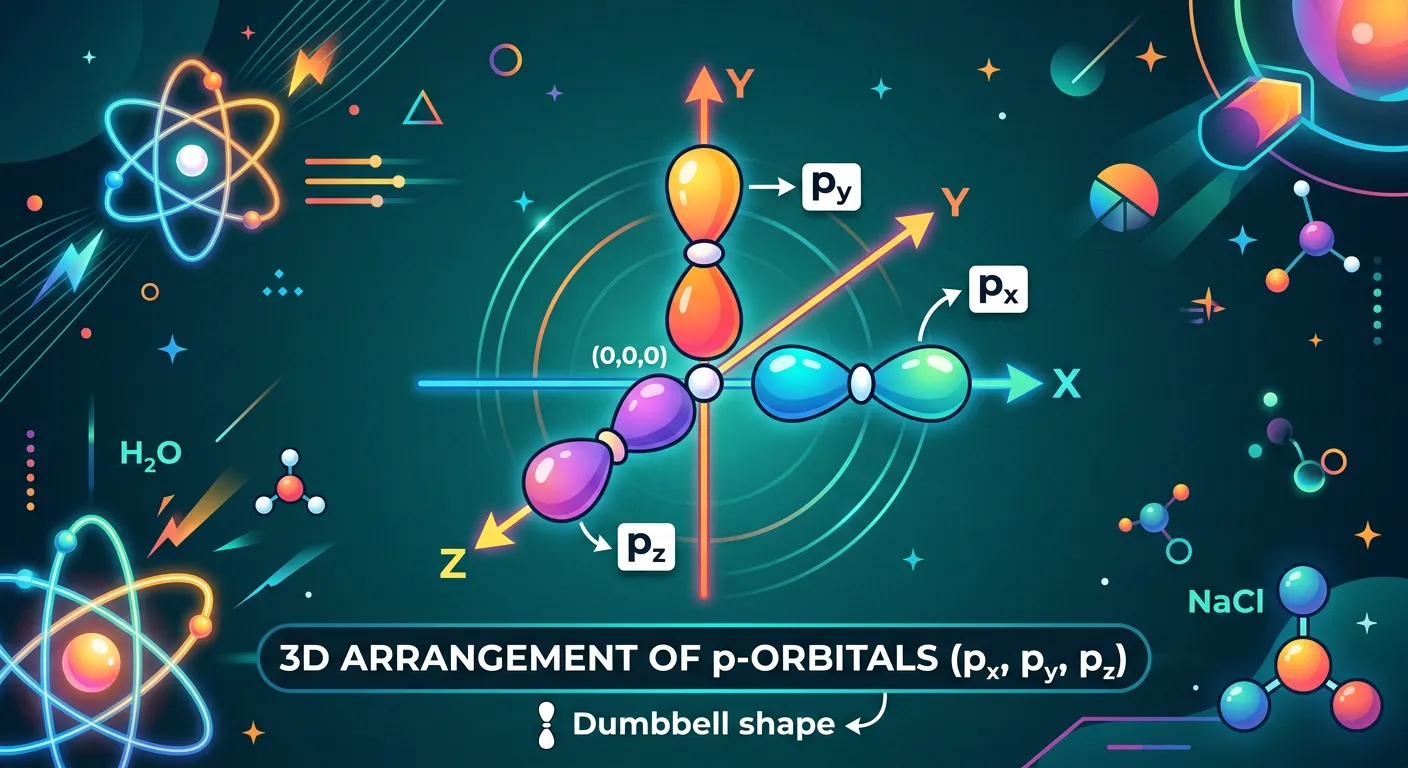
“Things get cooler with p-orbitals! They are shaped like a dumbbell. Because they have directional character, we have three of them: px, py, and pz, aligned along the axes. They are like three wings of a house, each holding two electrons in their lobes.”
When $l=1$, we enter the world of p-orbitals. Unlike s-orbitals, p-orbitals are not spherical; they are dumbbell-shaped. Each p-orbital consists of two 'lobes' of high electron density situated on either side of the nucleus, separated by a plane where the probability of finding an electron is exactly zero—this is called an angular node or a nodal plane. Because these lobes point in specific directions along the Cartesian axes, p-orbitals are described as directional. This directionality is the physical reason why atoms form specific bond angles in molecules like water or methane, as bonds tend to form in the direction of maximum orbital overlap.
There are three p-orbitals in any p-subshell, corresponding to the three possible values of the magnetic quantum number $m_l$ (-1, 0, +1). These are labeled $p_x, p_y,$ and $p_z$ because their lobes lie along the x, y, and z axes respectively. In an isolated atom, these three orbitals have the exact same energy, a state we call 'degenerate.' However, this degeneracy can be broken in the presence of external magnetic or electric fields (known as the Zeeman and Stark effects). The size and energy of p-orbitals increase as the principal quantum number $n$ increases ($2p < 3p < 4p$). Understanding the orientation of these dumbbells is vital for visualizing pi-bonds in organic chemistry, where side-on overlap of parallel p-orbitals creates the electron clouds above and below the plane of a double bond. Furthermore, note that $n=1$ has no p-orbitals because $l$ must be less than $n$. The first set of p-orbitals appears in the $L$ shell ($n=2$). Each p-orbital has one nodal plane passing through the nucleus, meaning an electron in a $p_x$ orbital will never be found on the $yz$ plane.
| Orbital | Orientation | Nodal Plane |
|---|---|---|
| $p_x$ | Along x-axis | yz-plane |
| $p_y$ | Along y-axis | xz-plane |
| $p_z$ | Along z-axis | xy-plane |
Quick Revision Points
- p-orbitals are dumbbell-shaped with two lobes.
- There are three degenerate p-orbitals ($p_x, p_y, p_z$) starting from $n=2$.
- Each p-orbital has one angular node (nodal plane) passing through the nucleus.
- Directional character leads to covalent bonding and molecular geometry.
- Radial nodes in p-orbitals are calculated as $(n - 1 - 1) = n - 2$.
NEET Exam Angle
- Practice identifying the nodal plane for each p-orbital (e.g., $p_x$ has the $yz$ nodal plane).
- Note that $1p$ orbitals do not exist because $l$ must be less than $n$.
- Questions often ask about the total number of nodes in a $3p$ orbital $(3-1=2)$.
06Complex Cloverleafs: The Geometry of d-Orbitals
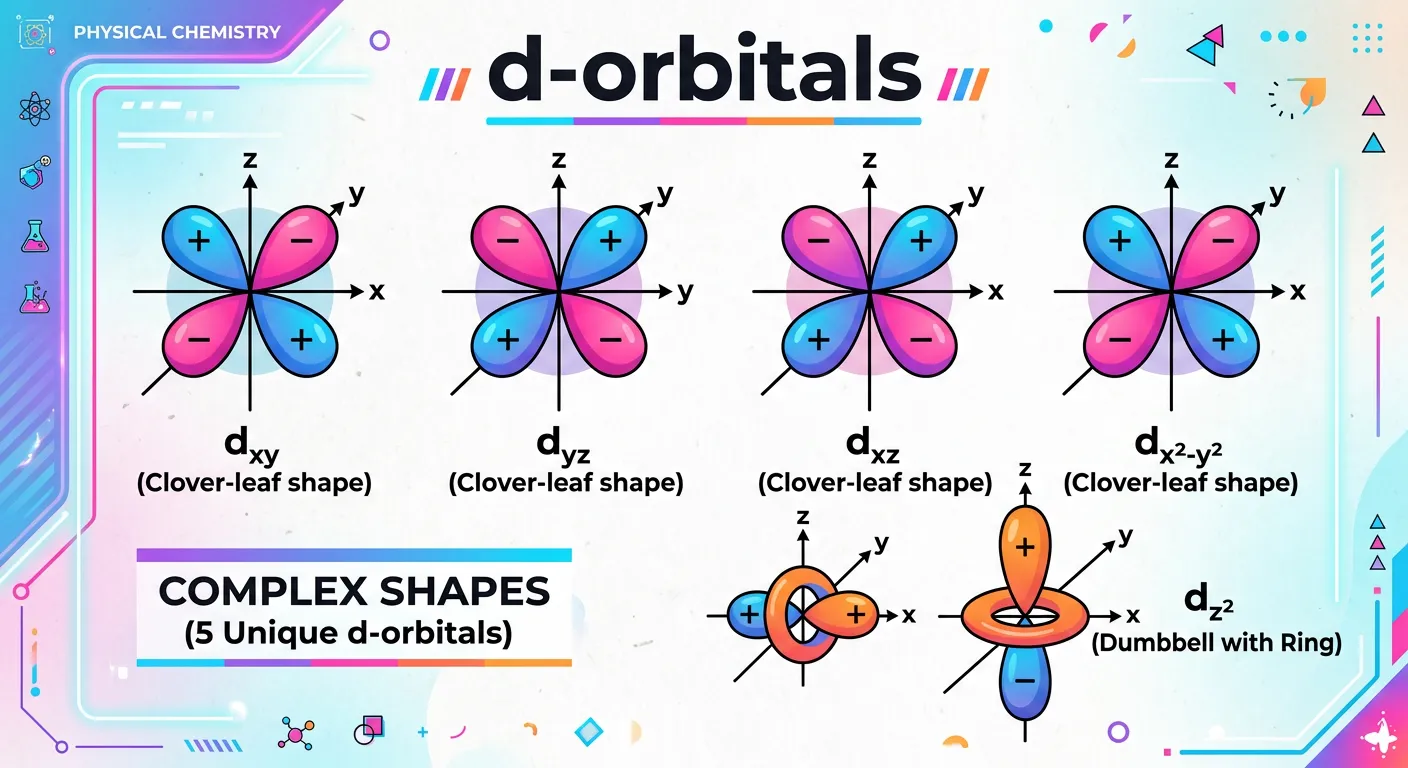
“As we move to d-orbitals, the shapes get fancy! We have four clover-leaf shapes and one with a donut ring in the middle. These are crucial for NEET, especially in understanding transition metal chemistry and coordination compounds. Stay sharp, they are quite complex!”
For $l=2$, we have the d-orbitals. These start appearing from the third shell ($n=3$) because the azimuthal quantum number $l$ can range from $0$ to $(n-1)$. There are five d-orbitals in a set, corresponding to $m_l$ values of -2, -1, 0, +1, +2. Their shapes are significantly more complex than the s or p types. Four of these orbitals—$d_{xy}, d_{yz}, d_{xz},$ and $d_{x^2-y^2}$—have a cloverleaf shape with four lobes of electron density. The first three ($d_{xy}, d_{yz}, d_{xz}$) have lobes that lie in the planes between the Cartesian axes (non-axial), while $d_{x^2-y^2}$ has lobes that lie directly on the x and y axes (axial). This spatial distinction is the fundamental key to studying Crystal Field Theory in Class 12 Coordination Chemistry.
The fifth orbital, $d_{z^2}$, is unique. It does not follow the cloverleaf pattern; instead, it consists of two large lobes pointing along the z-axis and a doughnut-shaped ring of electron density, often called a 'collar' or 'torus,' in the xy-plane. Despite its different appearance, it is energetically degenerate with the other four d-orbitals in an isolated atom. D-orbitals are responsible for the unique properties of transition metals, such as variable oxidation states, catalytic behavior, and the formation of beautifully colored complexes. They have two angular nodes each, which dictates how they interact with ligands during the formation of complex compounds. As $n$ increases ($3d, 4d, 5d$), the orbitals become larger and more diffuse, affecting the strength of the bonds they can form.
| Orbital Name | Lobe Orientation | Type |
|---|---|---|
| $d_{xy}, d_{yz}, d_{xz}$ | Between the axes | Non-axial |
| $d_{x^2-y^2}$ | Along X and Y axes | Axial |
| $d_{z^2}$ | Along Z axis with ring | Axial |
Quick Revision Points
- d-orbitals ($l=2$) exist in groups of five degenerate orbitals.
- Shapes: 4 cloverleafs and 1 'doughnut' ($d_{z^2}$).
- Radial nodes formula: $(n - 2 - 1) = n - 3$.
- Angular nodes for all d-orbitals = 2.
- The $d_{z^2}$ orbital is mathematically a combination of two other functions but is treated as a single orbital.
NEET Exam Angle
- The $d_{z^2}$ orbital is a frequent flyer in NEET exams. Know its shape and axial orientation.
- Distinguish between axial and non-axial orbitals; this is the key to understanding 'Splitting' in Coordination Chemistry.
- Calculate nodes for $3d$ (0 radial, 2 angular) vs $4d$ (1 radial, 2 angular).
07Orbital Mapping: The Key to Mastering Chemical Bonding

“So, orbitals aren't just shapes—they are the addresses of our electrons! Master these probability clouds, and you've cracked the code to chemical bonding. Keep practicing, stay curious, and you will definitely ace that NEET exam. See you in the next session!”
Understanding orbitals isn't just an exercise in spatial geometry; it’s about finding the precise 'address' and behavior of an electron within an atom. Each orbital is defined by its quantum numbers, and its specific shape dictates exactly how it will interact with other atoms to form chemical bonds. For example, the head-on overlap of two s-orbitals or an s and a p-orbital creates a strong sigma ($\sigma$) bond, while the sideways, parallel overlap of p-orbitals creates a pi ($\pi$) bond. Without a clear visualization of these 3D probability clouds, complex concepts like hybridization (such as $sp^3, sp^2,$ or $dsp^2$) would seem like abstract mathematical magic rather than the logical spatial rearrangements they truly are.
As you prepare for NEET, remember that the probability cloud approach is the only way to explain the structural diversity of the physical world. The repulsion between these electron clouds—described by VSEPR theory—and their underlying symmetry is what determines if a molecule like $CO_2$ is linear or if $H_2O$ is bent. These shapes influence macroscopic properties: whether a substance is a polar solvent, whether it exists as a gas or solid at room temperature, and how it will interact with biological receptors in your body. By mastering orbitals, you transition from rote memorization of chemical formulas to an intuitive understanding of molecular architecture. You are essentially learning the fundamental blueprints of matter. Mastery over these probability mists is your ticket to a high rank in Chemistry. Always connect the shape of the orbital to the symmetry of the molecule; for instance, the spherical nature of s-orbitals allows for rotation, while the directional nature of p and d orbitals restricts it, leading to the fascinating world of isomerism. Keep practicing the nodal formulas and the boundary surface diagrams, as these are high-yield, easy-scoring areas in the competitive exam landscape.
Quick Revision Points
- Orbital shape and orientation determine bond strength and molecular geometry.
- Degenerate orbitals have the same energy level in the absence of external fields.
- Maximum probability density is found near the nucleus for s-orbitals.
- Nodal planes are regions where the electron density is exactly zero.
- The size and volume of orbitals increase with the principal quantum number ($n$).
NEET Exam Angle
- Focus on the relationship between orbital overlap and bond energy.
- Be ready to identify orbitals based on given quantum number sets ($n, l, m_l$).
- Remember: Angular nodes = $l$, Radial nodes = $n - l - 1$, Total nodes = $n - 1$.
Recommended Reading
Explore related Biology topics to build deeper chapter connections for NEET.
- Nature of Electromagnetic Radiation · Topic 2.1
- Quantum Mechanical Model · Topic 2.12
- Significance of Quantum Numbers · Topic 2.16
- Photoelectric Effect · Topic 2.2
- Bohr Model · Topic 2.4
- de Broglie's Relationship · Topic 2.9
- Jump to Key Terms (Quick Revision)
- Review Common NEET Mistakes
- Read Topic FAQs
- Check PYQ Pattern Notes
- Practice NEET MCQs
- Solve NEET PYQs
📚 Key Terms
⚠️ Common NEET Mistakes
- 1Thinking that the wave function (ψ) itself has physical meaning; only ψ² represents probability.
- 2Confusing 'Orbit' (Bohr's 2D path) with 'Orbital' (Quantum 3D region).
- 3Forgetting to subtract 1 when calculating radial nodes (using n-l instead of n-l-1).
- 4Assuming all d-orbitals look like cloverleafs; the dz² orbital is a major exception.
- 5Believing that an orbital has a solid boundary; it is actually a fuzzy cloud where 100% probability is never reached.
📝 NEET PYQ Pattern
In NEET 2018–2024, questions frequently focus on identifying the number of radial and angular nodes in specific orbitals. There is also a recurring pattern of testing the orientation of d-orbitals, specifically the unique shape and axial alignment of the dz2 and dx2-y2 orbitals.
❓ Frequently Asked Questions
What is the physical meaning of the wave function squared (ψ²)?
The wave function squared, ψ², represents the 'Probability Density.' It gives the probability of finding an electron in a small volume around a specific point in space. It is always a positive value.
How many nodes are present in a 3s orbital vs a 2p orbital?
A 3s orbital has 2 radial nodes (3-0-1 = 2) and 0 angular nodes, totaling 2 nodes. A 2p orbital has 0 radial nodes (2-1-1 = 0) and 1 angular node, totaling 1 node.
Why is the s-orbital the only one that is non-directional?
The s-orbital is non-directional because it is spherically symmetrical. The probability of finding an electron depends only on the distance from the nucleus, not on the angle or direction.
Which d-orbital does not have the standard four-lobe clover-leaf shape?
The dz² orbital is the unique d-orbital. It has two lobes pointing along the z-axis and a doughnut-shaped 'collar' or ring of electron density in the xy-plane.
What is the difference between a radial node and an angular node?
A radial node is a spherical surface where the probability is zero, calculated as (n-l-1). An angular node is a flat or conical surface (nodal plane) where probability is zero, equal to the value of l.
How does the size of a 4p orbital compare to a 2p orbital?
A 4p orbital is significantly larger and has a higher energy than a 2p orbital because its principal quantum number (n) is higher, meaning the electron is likely to be found further from the nucleus.
Written By
NEET Content Strategist & Biology Expert
Sangita Kumari is a NEET educator and content strategist with over 6 years of experience teaching Biology, Chemistry, and Physics to Class 11 and 12 aspirants. She helps bridge the gap between traditional NCERT preparation and modern AI-powered learning. Her content is trusted by thousands of NEET aspirants across India.