🎬 Video Lesson Available
Watch the full 7-slide video lesson for Quantum Mechanical Model with AI teacher narration and visual explanations.
01The Paradigm Shift: From Planetary Orbits to Quantum Mechanics
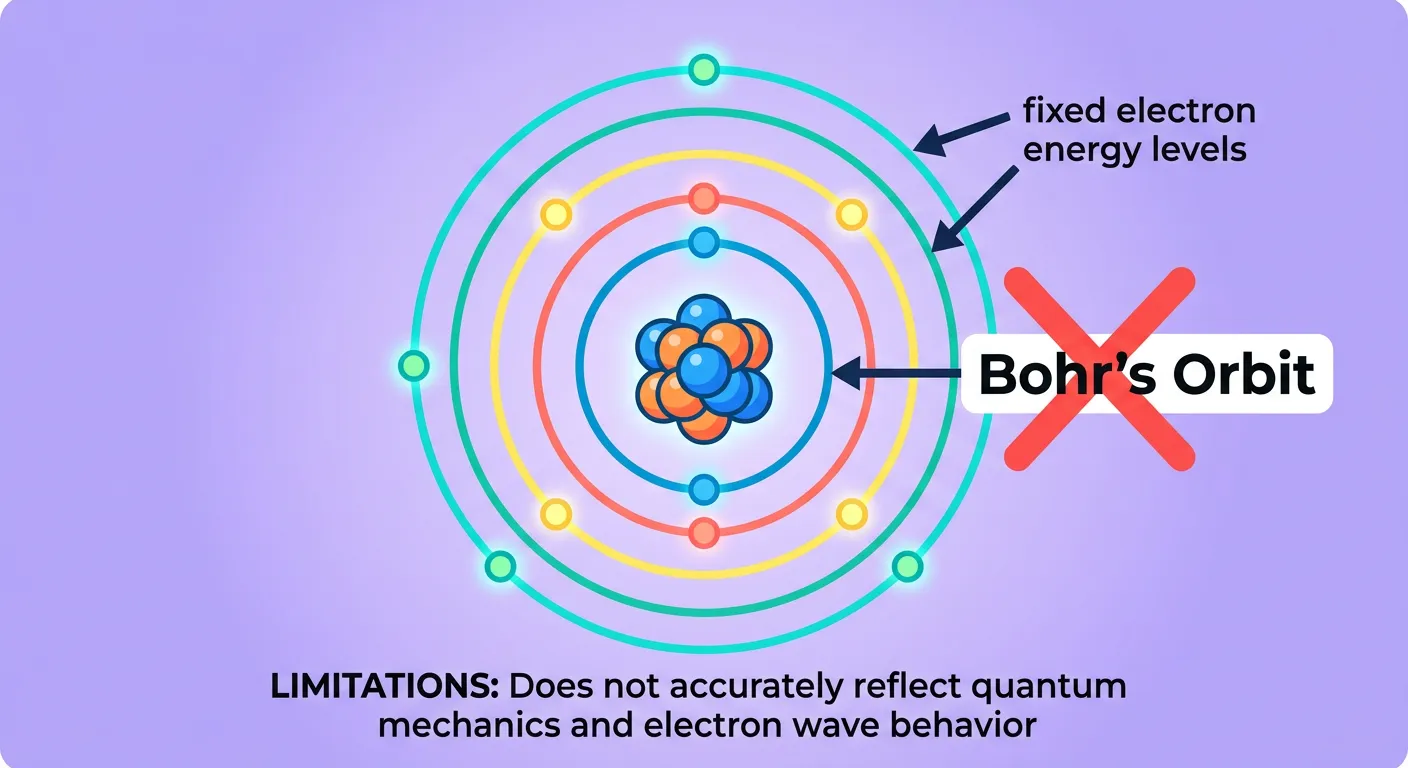
“Welcome, NEET aspirants! For years, we thought electrons orbited the nucleus like planets around the Sun. But physics is full of surprises! That fixed orbit model failed for complex atoms. Today, we step into the mysterious, non-deterministic world of the Quantum Mechanical Model. Let's dive in!”
For a long time, students and scientists alike were comfortable with Bohr’s model of the atom. It was easy to visualize: a central nucleus with electrons revolving in neat, circular tracks like planets around the sun. However, as we peered deeper into the subatomic world, this 'planetary' model began to crack. Bohr’s theory could perfectly explain the hydrogen spectrum, but it fell flat when faced with multi-electron atoms. It couldn't account for the splitting of spectral lines in magnetic fields (the Zeeman effect) or electric fields (the Stark effect). Most importantly, Bohr’s model treated electrons as simple particles moving in definite paths, completely ignoring the wave-like behavior of matter discovered by de Broglie and the inherent uncertainty proposed by Heisenberg.
Enter the Quantum Mechanical Model—a theoretical framework that replaced deterministic orbits with a sophisticated understanding of probability. In classical mechanics, if you know the starting position and velocity of a particle, you can predict its entire future path. But in the quantum world, this is fundamentally impossible for an electron. We had to move away from the idea of fixed 'orbits' and embrace 'orbitals'—three-dimensional regions where the probability of finding an electron is high. This shift represents the birth of modern chemistry, where we stop asking 'Exactly where is the electron?' and start asking 'Where is the electron most likely to be?'
| Feature | Classical Mechanics (Bohr) | Quantum Mechanics (Schrödinger) |
|---|---|---|
| Electron Path | Fixed, circular orbits (2D) | Three-dimensional probability regions (3D) |
| Certainty | Position and momentum known exactly | Position and momentum cannot be known simultaneously |
| Nature | Particle-only behavior | Wave-particle duality |
| Complexity | Failed for multi-electron systems | Applicable to all elements |
Quick Revision Points
- Bohr's model failed to explain the spectra of multi-electron atoms and the Zeeman/Stark effects.
- Classical mechanics is deterministic; quantum mechanics is probabilistic.
- The shift involves moving from 2D orbits to 3D orbitals.
- The Heisenberg Uncertainty Principle makes the concept of definite trajectories obsolete.
NEET Exam Angle
- Questions often ask why Bohr’s model was discarded; remember the Stark and Zeeman effects.
- Understand that classical mechanics works for macroscopic objects but fails at the subatomic level.
- Be prepared for Assertion-Reasoning questions regarding the deterministic vs. non-deterministic nature of these models.
02The Electron Cloud Concept: Visualizing Probability in Motion

“Forget strict paths! Heisenberg and Schrödinger changed the game. Instead of a trajectory, we talk about the 'Electron Cloud'. Think of it like a ceiling fan spinning—you can't point to one spot, but you know where the blades spend most of their time. It's all about probability!”
To understand the Quantum Mechanical Model, you must first unlearn the word 'trajectory.' In the macroscopic world, a cricket ball follows a clear path. In the atomic world, the Heisenberg Uncertainty Principle tells us that we cannot simultaneously know the exact position and momentum of an electron. Therefore, the very idea of a path or a 'trajectory' is meaningless. Instead, we describe the electron as an 'Electron Cloud.' This doesn't mean the electron is smeared out like jelly; rather, it means that at any given moment, the electron could be at various points, and we represent this statistically.
A great way to visualize this is the ceiling fan analogy. When a fan is off, you can see the three distinct blades. When you turn it on at high speed, you no longer see the blades in one spot. Instead, you see a blurred 'cloud' or a disk. You can't say exactly where a blade is at a specific millisecond, but you know the region of space where it spends its time. Similarly, in an atom, the electron moves so rapidly and its wave nature is so prominent that we visualize its position as a time-averaged density of probability. Where the cloud is dense, the chance of finding the electron is high; where the cloud is thin, the chance is low.
This 'pathless' motion is a fundamental feature of the quantum mechanical model of the atom. It changes how we think about atomic size and shape. We no longer look for a hard boundary like the edge of a marble; we look for the boundary of the electron cloud. This concept of electron density is crucial because it dictates how atoms interact, bond, and repel each other in chemical reactions.
Quick Revision Points
- The Heisenberg Uncertainty Principle forbids the existence of definite trajectories for electrons.
- An 'Electron Cloud' represents the probability distribution of an electron in 3D space.
- High electron density corresponds to a high probability of finding the electron.
- The ceiling fan analogy helps conceptualize time-averaged position versus instantaneous location.
NEET Exam Angle
- NEET questions often focus on why 'orbits' were replaced by 'electron clouds.'
- Identify the role of the Uncertainty Principle in defining the 'cloud' model.
- Understand that electron density is a statistical concept, not a physical 'smearing' of the particle.
03Wave-Particle Duality: The Vibrating String Behavior of Electrons
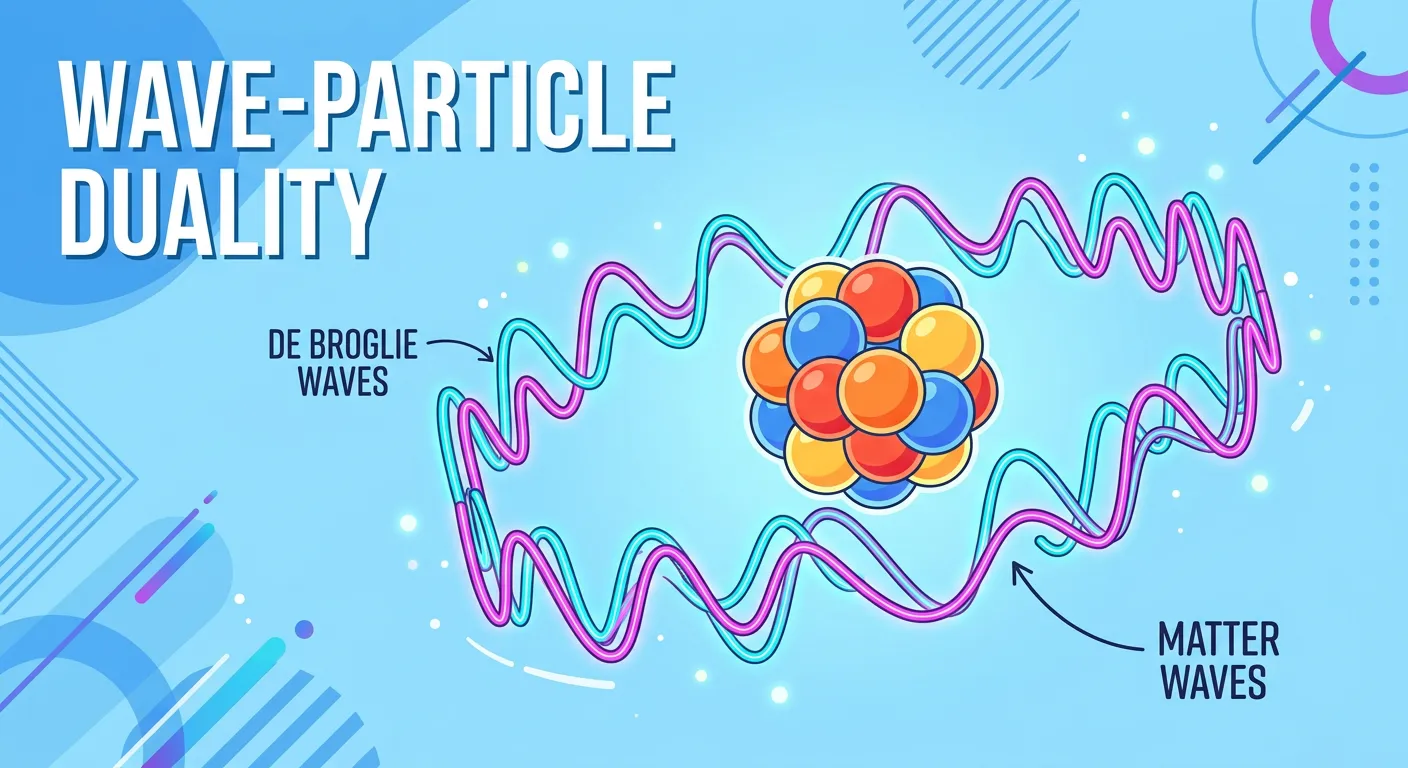
“Louis de Broglie suggested that electrons have a dual nature—they are particles and waves! This means they don't move in straight lines; they behave like vibrating strings. This wave-like behavior is why we can't pinpoint their exact location. Nature loves a bit of mystery, right?”
The cornerstone of the quantum mechanical model is the dual nature of matter. Proposed by Louis de Broglie, this principle states that just as light behaves as both a wave and a particle, so does the electron. In the confines of an atom, an electron doesn't just act like a tiny ball; it acts like a stationary or 'standing wave' around the nucleus. Imagine a guitar string plucked at both ends—it vibrates in specific patterns called harmonics. It cannot vibrate at just any frequency; it must fit the length of the string perfectly. Electrons in an atom are very similar.
Because the electron is confined by the electrostatic pull of the nucleus, its wave function must 'fit' around the nucleus in a way that doesn't cancel itself out. This leads to the quantization of energy. Only certain wavelengths—and therefore only certain energy levels—are allowed. If an electron were just a particle, it could theoretically have any energy. But because it is a wave, its energy is restricted to specific 'standing wave' states. This explains why electrons occupy discrete energy levels without spiraling into the nucleus.
This wave nature is also why we cannot pinpoint the electron's coordinates. Waves are inherently spread out over a region of space. When you try to locate a wave, you can't say it's at one single mathematical point. By embracing the wave-particle duality, the quantum mechanical model provides a mathematical reason for the stability of atoms and the existence of the periodic table.
Quick Revision Points
- De Broglie's equation ($λ = h/mv$) links the particle property (momentum) to the wave property (wavelength).
- Electrons behave as standing waves confined by the nucleus.
- Quantization of energy is a direct result of the wave-like restrictions on electron motion.
- Wave-particle duality prevents the pinpointing of exact electron coordinates.
NEET Exam Angle
- Numerical problems on $λ = h/mv$ are common; ensure you use SI units (kg for mass).
- Understand the conceptual link between 'standing waves' and 'quantized energy levels.'
- Be ready for questions asking why the wave nature of electrons is negligible for macroscopic objects but vital for atoms.
04The Schrödinger Wave Equation: The Mathematical Foundation of Chemistry

“Schrödinger gave us a master equation! He introduced the 'Wave Function', represented by the Greek letter psi. It describes the energy and position of electrons. Solving this for complex atoms is tough, but it's the foundation of all modern quantum chemistry and NEET-level success!”
In 1926, Erwin Schrödinger took the wave-particle concept and developed the fundamental equation of quantum mechanics. This equation is to chemistry what Newton’s laws are to classical physics. The Schrödinger Wave Equation, often written as $\hat{H}\psi = E\psi$, describes the behavior of an electron in an atom. Here, $\psi$ (the Greek letter psi) is the 'Wave Function.' It is a mathematical function that contains all the information we can possibly know about an electron in a specific energy state.
The symbol $\hat{H}$ is the Hamiltonian operator, a set of mathematical instructions that, when applied to the wave function, yields the total energy ($E$) of the system. When this equation is solved for an atom, it doesn't give us one single answer. Instead, it gives us a set of permissible wave functions (orbitals) and their corresponding energy levels. Any energy state that doesn't solve the equation is 'non-permissible.' This is how the model naturally explains why electrons can only exist in certain energy states.
Solving this equation for the hydrogen atom was a triumph, as it perfectly matched experimental data. For multi-electron atoms, the equation becomes incredibly complex because of the repulsions between electrons, but the principles remain the same. The solutions to the Schrödinger equation lead directly to the four quantum numbers, which serve as the 'postal address' for every electron in an atom. This mathematical rigor is what makes the quantum mechanical model the absolute gold standard for modern chemistry.
Quick Revision Points
- The Schrödinger equation ($\hat{H}\psi = E\psi$) is the basis of the quantum mechanical model.
- $\psi$ is the wave function; it represents the amplitude of the electron wave.
- $\hat{H}$ is the Hamiltonian operator representing total energy (Kinetic + Potential).
- Solving the equation provides 'eigenvalues' (permissible energy levels) and 'eigenfunctions' (orbitals).
NEET Exam Angle
- You don't need to solve the equation for NEET, but you must know what $\psi$, $\hat{H}$, and $E$ represent.
- Remember that the equation treats the electron as a wave in a 3D potential field.
- Focus on the fact that the Schrödinger equation leads to the discovery of quantum numbers.
05Probability Density: Deciphering the |ψ|² Hotspots
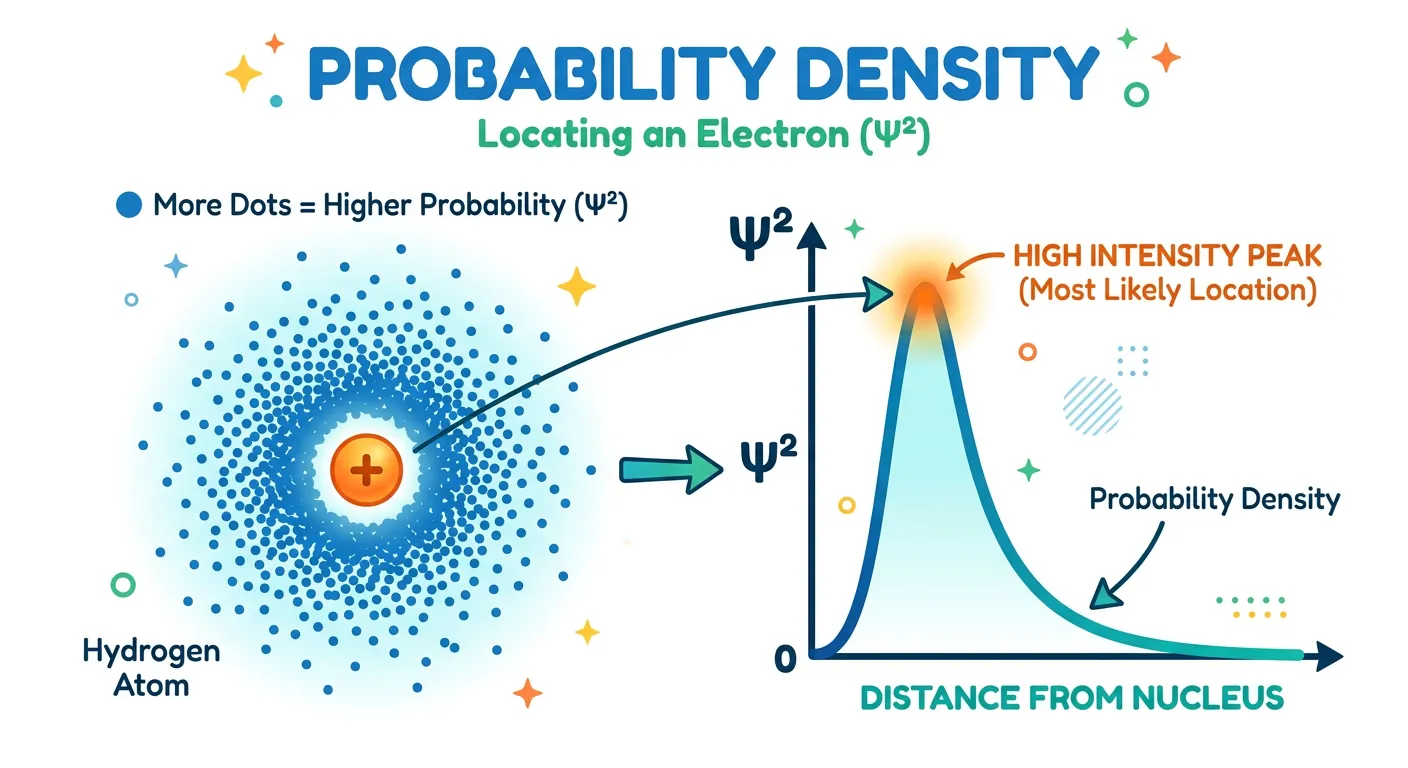
“If psi is the wave function, then psi-squared tells us the probability density. It tells us where an electron is most likely to be found. It’s not a path; it's a map of 'hotspots' where you have the best chance of catching that elusive electron!”
While the wave function $\psi$ is mathematically essential, it doesn't have a direct physical meaning because it can have positive or negative values, and even complex numbers. However, Max Born provided the key to unlocking its physical significance. He proposed that the square of the absolute value of the wave function, represented as $|\psi|^2$, represents the 'Probability Density.' This is a purely positive value that tells us the probability of finding an electron in a tiny volume around a specific point.
Think of $|\psi|^2$ as a 'heat map' or a map of 'hotspots.' In regions where $|\psi|^2$ is high, you have a high statistical chance of detecting an electron. In regions where it is zero, you will never find an electron; these zero-probability regions are called 'nodes.' This concept is a total departure from the Bohr model. Instead of saying the electron is at a distance of 0.529 Å, we say that at 0.529 Å, the probability density is at its maximum for a hydrogen atom in its ground state.
Graphical representations of these probabilities are vital for your NEET preparation. We often look at Radial Probability Distribution curves, which plot the total probability of finding an electron at a certain distance ($r$) from the nucleus. These graphs show us that while the electron spends most of its time near the nucleus, there is a small but non-zero chance of it being quite far away. This 'no-boundary' nature of the atom is why we use 90% probability surfaces to define orbital shapes.
| Term | Mathematical Symbol | Physical Meaning |
|---|---|---|
| Wave Function | $\psi$ | Amplitude of the electron wave; can be (+) or (-) |
| Probability Density | $ | \psi |
| Node | $\psi = 0$ | A region where the probability of finding an electron is zero |
| Max Probability | Peak of $ | \psi |
Quick Revision Points
- $\psi$ has no physical meaning, but $|\psi|^2$ is the probability density.
- $|\psi|^2$ is always positive and represents the statistical likelihood of finding an electron.
- Nodes are regions of zero probability density.
- Radial probability distribution functions help us visualize atomic size and 'hotspots.'
NEET Exam Angle
- A very common NEET question asks for the physical significance of $\psi$ vs. $|\psi|^2$.
- Be prepared to identify the number of nodes in a radial probability graph.
- Remember: Max probability for 1s hydrogen is at the Bohr radius, but the electron isn't confined to that exact spot.
06The 3D Anatomy of Orbitals: s, p, d, and f Hangout Zones
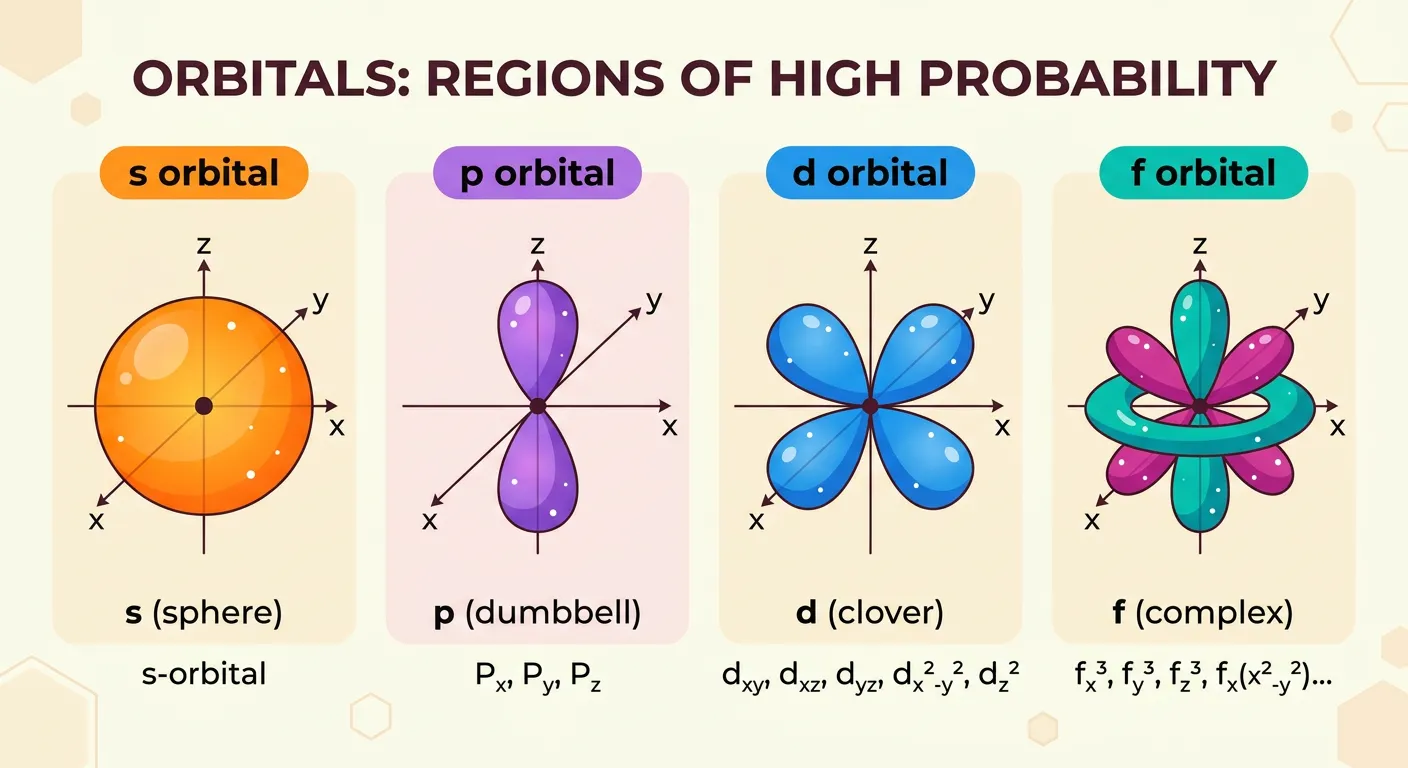
“So, what’s an orbital? It's just a 3D space where the probability of finding an electron is maximum—usually 90 percent! We have s, p, d, and f orbitals with unique shapes. Think of them as the electron's 'favorite hangout zones' around the nucleus.”
Since an electron cloud theoretically extends to infinity, how do we draw an atom? Chemists use 'Boundary Surface Diagrams.' By convention, we draw a shape that encloses the region where there is a 90% to 95% probability of finding the electron. This 3D space is what we call an 'Atomic Orbital.' It’s not a physical container, but a 'favorite hangout zone' for the electron. Each orbital has a unique shape determined by its angular momentum quantum number ($l$).
The 's' orbitals are simple spheres. No matter which direction you move from the nucleus, the probability remains the same at a given distance. The 'p' orbitals are dumbbell-shaped, oriented along the x, y, and z axes, meaning the electron prefers to hang out in two lobes on opposite sides of the nucleus. The 'd' orbitals are mostly double-dumbbells (cloverleaf shapes), and 'f' orbitals are even more complex. These shapes aren't just for show; they determine how atoms overlap to form chemical bonds.
It is crucial to distinguish between an 'orbit' (Bohr's 2D path) and an 'orbital' (Schrödinger's 3D region). Orbitals can hold a maximum of two electrons with opposite spins, and their specific geometry is the reason why molecules like water are bent and methane is a tetrahedron. When you study the Quantum Mechanical Model, you aren't just studying atoms—you are studying the geometric blueprint of the entire universe.
| Orbital Type | Shape | L-value | Degeneracy (No. of Orbitals) |
|---|---|---|---|
| s | Spherical | 0 | 1 |
| p | Dumbbell | 1 | 3 |
| d | Double-Dumbbell | 2 | 5 |
| f | Complex/Diffuse | 3 | 7 |
Quick Revision Points
- An orbital is a 3D region with ~90% probability of finding an electron.
- s-orbitals are spherical; p-orbitals are dumbbell-shaped; d-orbitals are cloverleaf-shaped.
- The shape of an orbital is defined by the angular momentum quantum number ($l$).
- Boundary surface diagrams represent the shape of these probability regions.
NEET Exam Angle
- Know the shapes and the number of nodes (radial and angular) for each orbital type.
- Practice identifying which orbitals have 'directional' properties (p, d, f) vs. 'non-directional' (s).
- Questions often link orbital shapes to the 'magnetic quantum number' ($m_l$) which defines orientation.
07Mastering the Quantum Model for NEET Success

“And that’s the Quantum Mechanical Model! It’s not about fixed paths, but about waves, probabilities, and those famous 'hangout zones' called orbitals. Mastering these concepts is your golden ticket to nailing those tricky periodic property questions in NEET. Keep dreaming, keep learning, and ace that exam!”
As we conclude our journey into the Quantum Mechanical Model, let's summarize the 'big picture.' We have moved from a world of certainty and orbits to a world of probability and orbitals. This model tells us that an atom is mostly empty space, with a tiny nucleus surrounded by complex, vibrating waves of electron density. The energy of an electron is quantized because it behaves like a standing wave, and its position is a statistical 'cloud' rather than a pinpoint location.
This model is the foundation for everything that follows in NEET Chemistry. Without it, we cannot understand the Periodic Table, the nature of chemical bonding, or why certain elements are more reactive than others. For example, the way p-orbitals overlap explains the strength of a double bond in organic chemistry. The way d-orbitals split in energy explains the vibrant colors of coordination compounds. Mastering the features of the quantum mechanical model isn't just about scoring marks in one chapter; it’s about building the logic for the entire subject.
To ace NEET questions on this topic, focus on conceptual clarity. Don't just memorize the Schrödinger equation—understand what it represents. Practice visualizing the graphs of $\psi$ and $|\psi|^2$ versus distance ($r$). When you see an 'Assertion-Reason' question, ask yourself: 'Does this statement assume the electron is a particle with a fixed path?' if so, it’s likely referring to the old Bohr model, not the modern Quantum model.
| Concept | Key Takeaway for NEET |
|---|---|
| Quantization | Emerges naturally from wave behavior in the Schrödinger equation. |
| Orbits vs Orbitals | Orbits are 2D paths (wrong); Orbitals are 3D regions (right). |
| Quantum Numbers | The address of the electron, derived from the wave function. |
| Electron Density | High density = high $ |
Quick Revision Points
- The model is based on wave-particle duality and the Uncertainty Principle.
- It replaces the concept of trajectories with probability distributions.
- Wave functions ($\psi$) contain all physical information about the electron.
- Orbital shapes and energies are crucial for understanding chemical properties.
NEET Exam Angle
- Focus on 'Correct/Incorrect Statement' type questions regarding model features.
- Expect questions relating the quantum mechanical model to periodic trends.
- Use the 'Checklist of Concepts': Waves, Probabilities, Orbitals, and Quantum Numbers.
Recommended Reading
Explore related Biology topics to build deeper chapter connections for NEET.
- Nature of Electromagnetic Radiation · Topic 2.1
- Atomic Orbitals · Topic 2.13
- Significance of Quantum Numbers · Topic 2.16
- Photoelectric Effect · Topic 2.2
- Bohr Model · Topic 2.4
- de Broglie's Relationship · Topic 2.9
- Jump to Key Terms (Quick Revision)
- Review Common NEET Mistakes
- Read Topic FAQs
- Check PYQ Pattern Notes
- Practice NEET MCQs
- Solve NEET PYQs
📚 Key Terms
⚠️ Common NEET Mistakes
- 1Confusing 'Orbit' with 'Orbital': Remember that orbits are 2D circles (Bohr) and orbitals are 3D clouds (Quantum).
- 2Thinking ψ has physical meaning: ψ is just a mathematical amplitude; only |ψ|² represents probability.
- 3Assuming an electron cloud is a 'physical mist': It is a statistical representation of the electron's position over time, not a physical substance.
- 4Neglecting units in de Broglie calculations: Always convert mass to kilograms and wavelength to meters for the formula λ = h/mv.
- 5Thinking probability is zero outside the orbital boundary: The 90% boundary is arbitrary; the probability is non-zero even very far from the nucleus.
📝 NEET PYQ Pattern
NEET questions from 2018–2024 frequently test the conceptual difference between ψ and |ψ|² and the definition of an orbital. There is a consistent trend of asking about the features of the quantum mechanical model in 'Correct/Incorrect Statement' type questions. Students should also be comfortable identifying radial nodes from graphs of |ψ|² vs r.
❓ Frequently Asked Questions
What is the physical significance of the wave function (ψ) in the quantum model?
By itself, the wave function (ψ) has no direct physical meaning and can even be a complex number. It represents the amplitude of the electron wave. However, its square (|ψ|²) is physically significant as it represents the probability density of finding an electron at a specific point.
Why did the Bohr model fail to explain the spectra of multi-electron atoms?
Bohr's model only considered the attraction between the nucleus and a single electron. In multi-electron atoms, electron-electron repulsions occur, which Bohr's model couldn't account for. It also failed to explain the splitting of spectral lines in magnetic (Zeeman effect) and electric fields (Stark effect).
What is the difference between an orbit and an orbital in terms of electron motion?
An orbit (Bohr model) is a well-defined circular 2D path where an electron moves with a fixed velocity and position. An orbital (Quantum model) is a 3D region of space where the probability of finding an electron is maximum (~90%), acknowledging the electron's wave nature and uncertainty.
How does the concept of probability density (|ψ|²) eliminate the need for fixed trajectories?
Since the Heisenberg Uncertainty Principle states we cannot know the exact position and momentum simultaneously, we cannot define a trajectory. Probability density allows us to describe where an electron is likely to be over time, replacing the 'path' with a 'cloud'.
What defines the boundary of an orbital where probability is 90%?
Because the probability of finding an electron only becomes zero at infinity, orbitals don't have hard edges. Scientists use 'boundary surface diagrams' to draw a shape that encloses the region where the probability of finding the electron is 90-95%.
How does the quantum mechanical model account for the wave nature of electrons proposed by de Broglie?
The model uses the Schrödinger wave equation to treat electrons as 3D standing waves. This wave behavior leads to quantized energy levels, as only certain wavelengths can 'fit' around the nucleus without destructive interference.
Written By
NEET Content Strategist & Biology Expert
Sangita Kumari is a NEET educator and content strategist with over 6 years of experience teaching Biology, Chemistry, and Physics to Class 11 and 12 aspirants. She helps bridge the gap between traditional NCERT preparation and modern AI-powered learning. Her content is trusted by thousands of NEET aspirants across India.