🎬 Video Lesson Available
Watch the full 7-slide video lesson for de Broglie's Relationship with AI teacher narration and visual explanations.
01Beyond Classical Mechanics: Introducing the Dual Nature of Matter
“Welcome! Imagine a cricket ball flying across a stadium. It moves in a straight line, right? But what about an electron? In the quantum world, things get weird. Louis de Broglie dared to ask: if light acts like a particle, can matter act like a wave?”
For centuries, classical physics, governed by Newtonian mechanics, provided a comfortable and predictable view of the universe. In this world, a cricket ball thrown by a bowler follows a distinct, measurable trajectory. It has a definite position and a definite momentum at every instant. However, as scientists began peering into the subatomic realm at the start of the 20th century, these rules started to break down. The discovery of the photoelectric effect by Albert Einstein demonstrated that light, which was long thought to be purely a wave, also behaves like a stream of particles called photons. This was the first major hint that nature might be more complex than our eyes suggest.
Louis de Broglie, a young French physicist, looked at this 'wave-particle duality' of light and made a daring intellectual leap. He suggested that if light could have a dual nature, perhaps matter—things we traditionally think of as solid particles—could also possess wave-like properties. This wasn't just a philosophical idea; it was a fundamental challenge to how we perceive the building blocks of the universe. For NEET aspirants, understanding this shift is crucial because it marks the transition from the Bohr model, which still treated electrons as little planets, to the modern quantum mechanical model where the electron is described as a wave-function.
Imagine the universe as a grand symphony where every moving object has its own rhythm. While a cricket ball’s 'rhythm' is too fast and small to detect, an electron’s wave nature is so significant that it determines the very structure of the atom. This realization didn't just add a new chapter to chemistry; it rewrote the entire book. De Broglie’s hypothesis provided the 'missing link' that explained why electrons are restricted to certain energy levels and how they behave when they aren't being 'particles.'
Quick Revision Points
- Classical mechanics fails to explain the behavior of microscopic particles like electrons.
- The photoelectric effect proved light has particle properties (photons).
- Louis de Broglie hypothesized that all moving matter has an associated wave nature.
- This concept is the foundation of the dual nature of matter.
- It shifts the focus from fixed trajectories to wave-like distributions.
NEET Exam Angle
- Questions often test the conceptual origins: why was Bohr's model insufficient? Answer: It ignored the wave nature.
- Understand the symmetry: Light (Wave → Particle) and Matter (Particle → Wave).
- Remember that de Broglie’s work was the theoretical catalyst for Heisenberg and Schrödinger.
02The Hypothesis of Matter Waves: Louis de Broglie’s Revolutionary Proposal
“De Broglie proposed the dual nature of matter. He suggested that every moving object, whether a dust particle or an electron, has a wave character associated with it. This is the 'Matter Wave' or 'de Broglie Wave' concept—the secret rhythm of the universe!”
In 1924, Louis de Broglie proposed that every moving material particle—whether it's a grain of sand, a planet, or an electron—has an associated wave. He termed these 'matter waves' or 'de Broglie waves.' It is important to distinguish these from the waves you encounter in everyday life. Unlike mechanical waves (like sound), they don't need a medium to travel through. Unlike electromagnetic waves (like light), they aren't generated by oscillating charges and don't consist of electric and magnetic fields. Matter waves are a fundamental property of matter in motion.
The genius of de Broglie lay in his assertion that this wave nature is universal. However, he also noted that the wave character is inversely proportional to the mass of the object. For macroscopic objects, the wavelength is so incredibly small (on the order of $10^{-35}$ meters) that no instrument on Earth can detect it. But for microscopic particles like electrons, the mass is so tiny that the wavelength becomes comparable to atomic dimensions. This makes the wave nature of electrons not just detectable, but the dominant factor in their behavior.
For a student preparing for NEET, it is vital to visualize the electron not as a 'dot' circling a nucleus, but as a 'cloud' or a standing wave surrounding it. This wave character explains phenomena like electron diffraction, which is used in electron microscopes to see things far smaller than what visible light can capture. The de Broglie hypothesis essentially demolished the idea of the electron as a simple billiard ball and introduced the concept of the 'probability wave' that defines where an electron might be found.
Quick Revision Points
- Matter waves are associated with any material particle in motion.
- They are distinct from electromagnetic waves and mechanical waves.
- The wave character is most prominent in the microscopic domain (electrons, protons).
- For large objects, the wave nature exists but is practically 'negligible.'
- The hypothesis was experimentally confirmed later by the Davisson-Germer experiment.
NEET Exam Angle
- A common MCQ trap: 'Are matter waves electromagnetic?' The answer is NO.
- Pay attention to the 'microscopic' vs 'macroscopic' distinction; wave nature is significant only for subatomic particles.
- Conceptual questions often link de Broglie waves to the wave-particle duality of the electron.
03The de Broglie Equation: Mathematical Relationship of Wavelength and Momentum
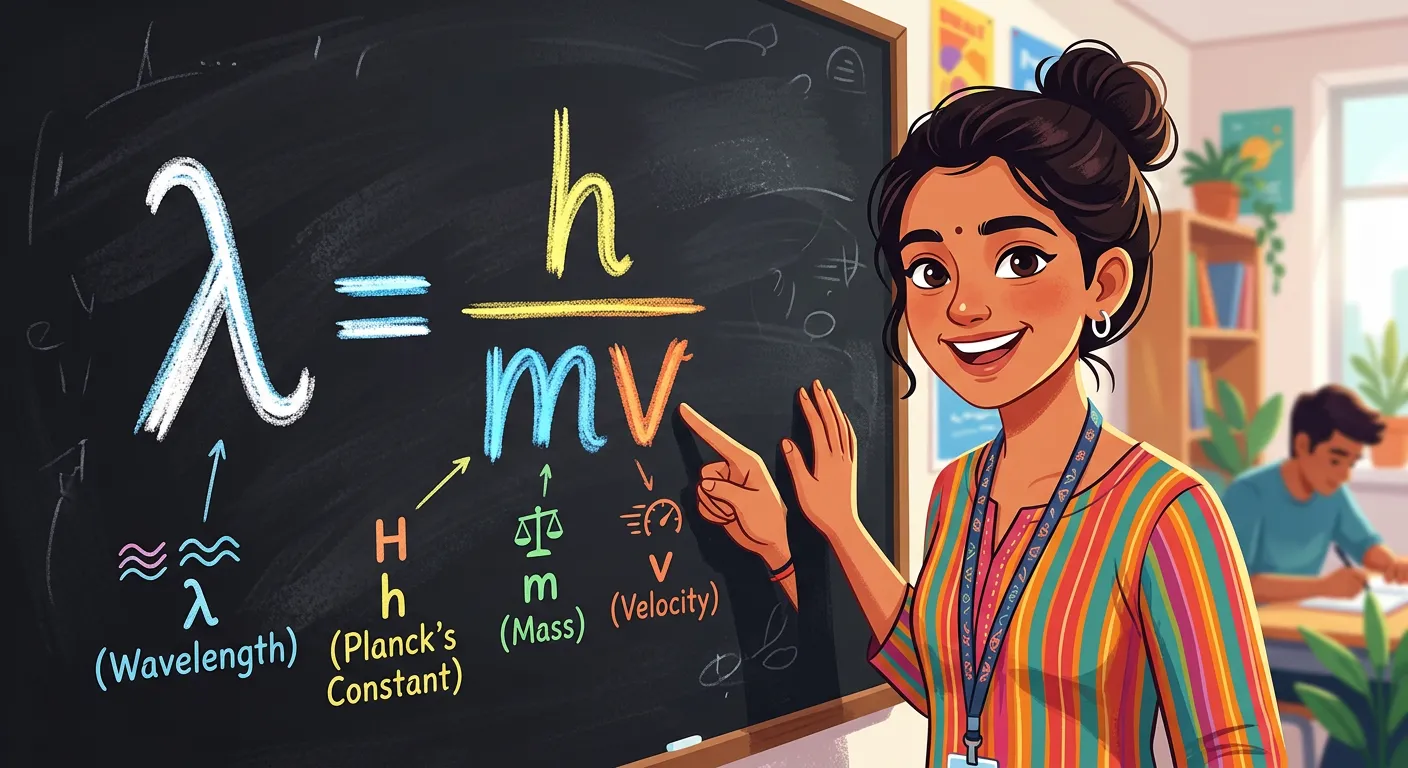
“Here is the magic formula: Lambda equals h divided by momentum. Lambda is the wavelength, h is Planck’s constant, and m times v is momentum. It tells us that wavelength and mass are inversely related. The smaller the mass, the bigger the wave character!”
To turn his hypothesis into a rigorous scientific law, de Broglie combined the two most famous energy equations of the time. First, from Planck’s quantum theory, $E = h\nu$ (where $h$ is Planck’s constant and $\nu$ is frequency). Second, from Einstein’s mass-energy equivalence, $E = mc^2$. By equating these and substituting the relationship between frequency, wavelength, and the speed of light ($\nu = c/\lambda$), he derived the relationship: $\lambda = h/mc$. For a particle moving with a velocity $v$ (rather than the speed of light $c$), this becomes the celebrated de Broglie equation: $\lambda = h/mv$.
In this equation, $\lambda$ represents the de Broglie wavelength, $h$ is Planck’s constant ($6.626 \times 10^{-34} J\cdot s$), $m$ is the mass of the particle, and $v$ is its velocity. Since the product of mass and velocity ($m \times v$) is momentum ($p$), the equation is often written as $\lambda = h/p$. This simple formula tells a profound story: wavelength and momentum are inversely related. If you increase the mass or the speed of an object, its wavelength shrinks. If you decrease them, the wavelength grows.
Understanding the units is critical for numerical accuracy in NEET. Planck’s constant $h$ has the units of Joule-seconds ($kg \cdot m^2 \cdot s^{-1}$), mass $m$ must be in kilograms ($kg$), and velocity $v$ in meters per second ($m/s$). This ensures that the wavelength $\lambda$ comes out in meters. Often, you will need to convert these meters into Angstroms ($\text{\AA}$) or nanometers ($nm$) to match the options in the exam.
| Variable | Symbol | SI Unit | Significance |
|---|---|---|---|
| Wavelength | $\lambda$ | meters ($m$) | Measure of wave character |
| Planck's Constant | $h$ | $J \cdot s$ | The fundamental quantum of action |
| Mass | $m$ | $kg$ | Determines the scale of the wave |
| Velocity | $v$ | $m/s$ | Affects the 'tightness' of the wave |
| Momentum | $p$ | $kg \cdot m/s$ | Inversely proportional to wavelength |
Quick Revision Points
- The de Broglie equation: $\lambda = h/mv$ or $\lambda = h/p$.
- It links a wave property (wavelength) to a particle property (momentum).
- Planck's constant ($h$) is the proportionality constant.
- Larger momentum results in a shorter, less detectable wavelength.
- Smaller momentum (like a slow electron) results in a larger, significant wavelength.
NEET Exam Angle
- Calculation of $\lambda$ for an electron is a high-yield question. Use $m = 9.1 \times 10^{-31} kg$.
- Always check your units! If mass is given in grams, convert to $kg$ immediately.
- The relationship $\lambda \propto 1/p$ is frequently used in ratio-based questions (e.g., if velocity doubles, wavelength halves).
04Microscopic vs. Macroscopic: Why We Don't See 'Waving' Humans
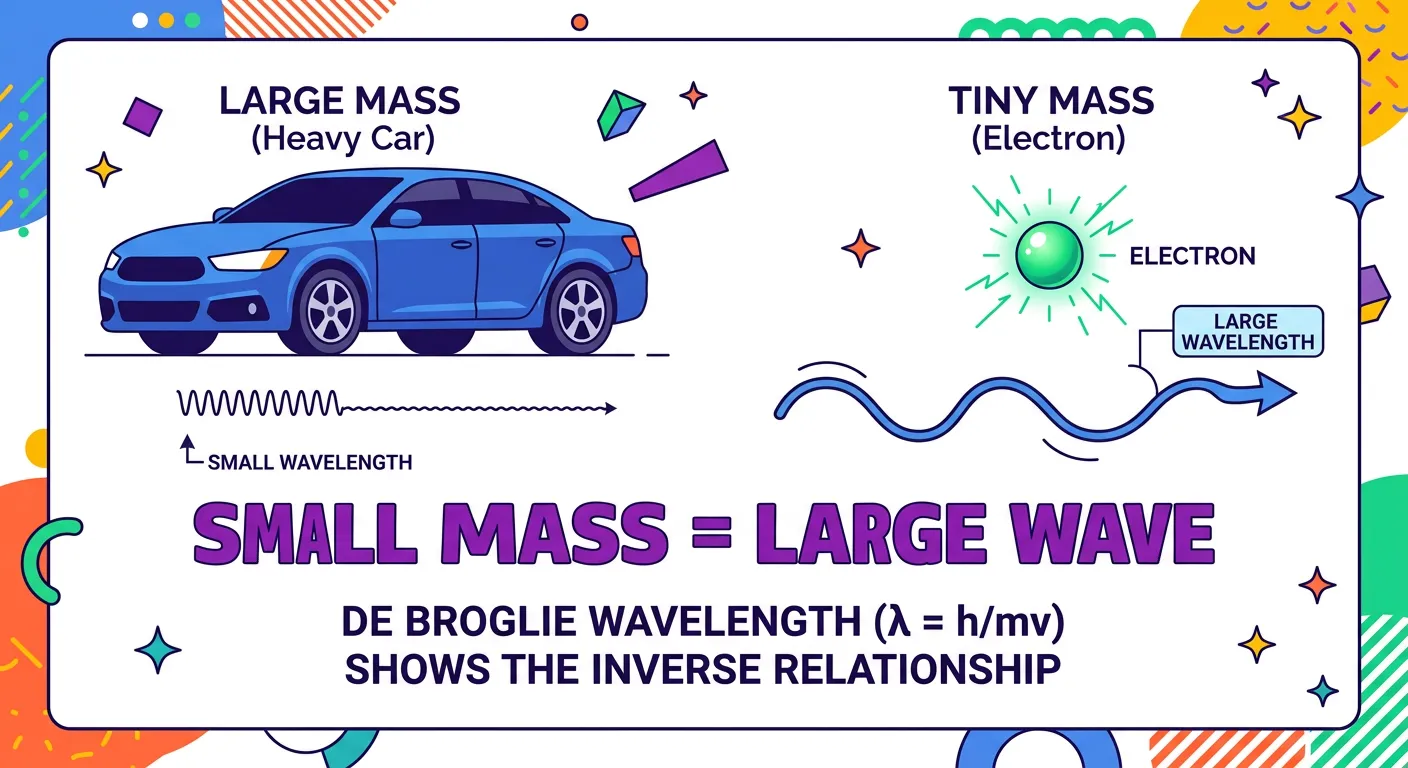
“Why don't we see people waving around as they walk? Because for a massive object, the wavelength is so incredibly tiny that it’s undetectable. But for a microscopic electron? That wave character is massive and defines how it behaves in an atom.”
A common question from curious students is: "If I have a wave nature, why don't I diffract when I walk through a doorway?" The answer lies in the sheer magnitude of the mass. Let’s compare a macroscopic object, like a 100g ball, with a microscopic object, like an electron. If both move at $100 m/s$, the ball’s wavelength is roughly $10^{-34}$ meters. For context, the diameter of an atomic nucleus is about $10^{-15}$ meters. The ball’s wavelength is billions of times smaller than a nucleus! It is physically impossible to measure or observe.
However, for an electron moving at the same speed, its tiny mass ($9.11 \times 10^{-31} kg$) results in a wavelength of about $7.27 \times 10^{-6}$ meters. This falls into the observable range and is large enough to interact with the spacing between atoms in a crystal. This is why we can see diffraction patterns for electrons but not for cricket balls. In the world of NEET Chemistry, this distinction is why we only apply quantum mechanics to subatomic particles. Classical mechanics is simply an approximation that works when the wavelength is too small to matter.
This practical application of de Broglie’s theory led to the invention of the Electron Microscope. While traditional optical microscopes are limited by the wavelength of visible light (about $400-700 nm$), electron microscopes use electron waves with much shorter wavelengths. Because the resolution is inversely proportional to wavelength, electron microscopes can magnify objects millions of times, allowing us to see individual atoms and viruses that are invisible to light.
| Object | Mass ($kg$) | Velocity ($m/s$) | de Broglie $\lambda$ ($m$) | Detectability |
|---|---|---|---|---|
| Cricket Ball | $0.15$ | $30$ | $\approx 1.4 \times 10^{-34}$ | Non-detectable |
| Dust Particle | $10^{-9}$ | $0.01$ | $\approx 6.6 \times 10^{-23}$ | Non-detectable |
| Electron | $9.1 \times 10^{-31}$ | $2 \times 10^{6}$ | $\approx 3.6 \times 10^{-10}$ | Highly Detectable |
Quick Revision Points
- Wave nature is universal but mass-dependent.
- Large mass $\rightarrow$ extremely small $\lambda$ (classical behavior).
- Small mass $\rightarrow$ significant $\lambda$ (quantum behavior).
- Resolution of a microscope depends on the wavelength of the probe used.
- Diffraction experiments (like Davisson-Germer) prove the wave nature of electrons.
NEET Exam Angle
- Expect questions asking why the wave nature of a car is not observed. Focus on the 'negligible wavelength' due to large mass.
- Theoretical questions on the Electron Microscope: it utilizes the wave nature of electrons to achieve high resolution.
- Understand that for macroscopic objects, the particle nature overrides the wave nature.
05Validating Bohr’s Model: Standing Waves and Quantized Orbits

“This discovery explained Bohr’s theory! De Broglie showed that an electron orbits the nucleus only where it forms a complete 'standing wave'. If it doesn't fit the circumference, the wave cancels out. This is why orbits are quantized and stable!”
One of the greatest triumphs of de Broglie’s relationship was providing a theoretical basis for Bohr’s second postulate. Bohr had stated that an electron can only revolve in certain fixed orbits where its angular momentum is an integral multiple of $h/2\pi$ ($mvr = nh/2\pi$). While Bohr had the right formula, he couldn't explain why this was the case. De Broglie solved this by treating the electron as a circular standing wave.
For an electron wave to be stable in a circular orbit, the wave must 'close' perfectly on itself. This only happens if the circumference of the orbit ($2\pi r$) is equal to an integer number of wavelengths ($n\lambda$). If the circumference is not an integer multiple, the wave will interfere with itself destructively and cancel out. Therefore, only specific orbits are 'allowed' because only they support a stable standing wave. Mathematically, if $2\pi r = n\lambda$, and we substitute de Broglie’s $\lambda = h/mv$, we get $2\pi r = n(h/mv)$. Rearranging this gives $mvr = nh/2\pi$, which is exactly Bohr’s postulate!
This realization changed our view of atomic stability. The electron doesn't just 'sit' in an orbit; it exists as a resonant wave pattern. If the wave doesn't fit the 'ring,' the electron cannot exist there. This is why energy levels are quantized—they are the only physical spaces where the electron wave can survive without destroying itself through interference.
| Condition | Wave Interaction | Result |
|---|---|---|
| $2\pi r = n\lambda$ | Constructive (In phase) | Stable Orbit (Standing Wave) |
| $2\pi r \neq n\lambda$ | Destructive (Out of phase) | Unstable / Disallowed Energy Level |
| $n = 1, 2, 3...$ | Quantization | Principal Quantum Number ($n$) |
Quick Revision Points
- De Broglie justified Bohr’s quantization of angular momentum.
- Orbits are stable only when the circumference is an integral multiple of $\lambda$.
- The formula is $2\pi r = n\lambda$.
- This explains why electrons occupy fixed, discrete energy levels.
- A 'standing wave' is a wave that remains in a constant position.
NEET Exam Angle
- A favorite derivation-based question: 'Which principle explains $mvr = nh/2\pi$?' Answer: de Broglie’s matter waves.
- Numericals may ask for the number of waves in a particular orbit ($n$ is the number of waves).
- Conceptual questions on constructive vs. destructive interference in the context of atomic orbits.
06NEET Numerical Hacks: Wavelength, Kinetic Energy, and Potential

“NEET aspirants, listen up! Questions often ask you to calculate the wavelength given kinetic energy. Remember: momentum squared equals two times mass times energy. Use this substitution to quickly solve those tricky numericals. It is a favorite topic for examiners every year!”
In NEET, you will rarely get a simple 'plug and play' question using $\lambda = h/mv$. Instead, examiners love to link wavelength with Kinetic Energy ($K.E.$) or accelerating potential ($V$). Since $K.E. = \frac{1}{2}mv^2$, we can express momentum as $p = \sqrt{2m(K.E.)}$. Substituting this into the de Broglie equation gives us a very important shortcut: $\lambda = \frac{h}{\sqrt{2mE}}$, where $E$ is the kinetic energy.
Furthermore, if a charged particle (like an electron or proton) is accelerated through a potential difference of $V$ volts, its kinetic energy is $qV$ (where $q$ is the charge). This gives us the final comprehensive formula: $\lambda = \frac{h}{\sqrt{2mqV}}$. For an electron specifically, if you plug in the constants for $h$, $m$, and $q$, you get a very useful 'speed formula': $\lambda (in \text{\AA}) \approx \sqrt{\frac{150}{V}}$ or $\frac{12.27}{\sqrt{V}}$. Using this can save you precious minutes during the exam.
Another common pattern involves ratios. You might be asked to find the ratio of wavelengths for a proton and an alpha particle moving with the same kinetic energy. In such cases, remember that $\lambda \propto \frac{1}{\sqrt{m}}$. Since an alpha particle is 4 times heavier than a proton, its wavelength will be $\frac{1}{\sqrt{4}} = \frac{1}{2}$ that of the proton. Mastering these proportionalities is the key to cracking the Chemistry section of NEET.
| Scenario | Formula to Use | Dependency |
|---|---|---|
| Given Velocity | $\lambda = h/mv$ | $\lambda \propto 1/v$ |
| Given Kinetic Energy | $\lambda = h/\sqrt{2mE}$ | $\lambda \propto 1/\sqrt{m}, 1/\sqrt{E}$ |
| Given Potential ($V$) | $\lambda = h/\sqrt{2mqV}$ | $\lambda \propto 1/\sqrt{V}$ |
| Electron shortcut | $\lambda = 12.27 / \sqrt{V} \text{\AA}$ | For fast calculations |
Quick Revision Points
- $\lambda = h/\sqrt{2mE}$ connects wavelength to energy.
- For charged particles, $E = qV$, so $\lambda = h/\sqrt{2mqV}$.
- For an electron: $\lambda = \frac{12.27}{\sqrt{V}} \text{\AA}$ is a life-saver.
- Wavelength is inversely proportional to the square root of mass and energy.
- Protons, deuterons, and alpha particles are common comparison subjects.
NEET Exam Angle
- Ratio questions: If $KE_1 = KE_2$, then $\lambda_1/\lambda_2 = \sqrt{m_2/m_1}$.
- If a particle is accelerated by a potential $V$, and $V$ is quadrupled, the wavelength becomes half.
- Always be careful with charge ($q$) when comparing protons ($+e$) and alpha particles ($+2e$).
07Synthesis: The de Broglie Legacy in Quantum Mechanics
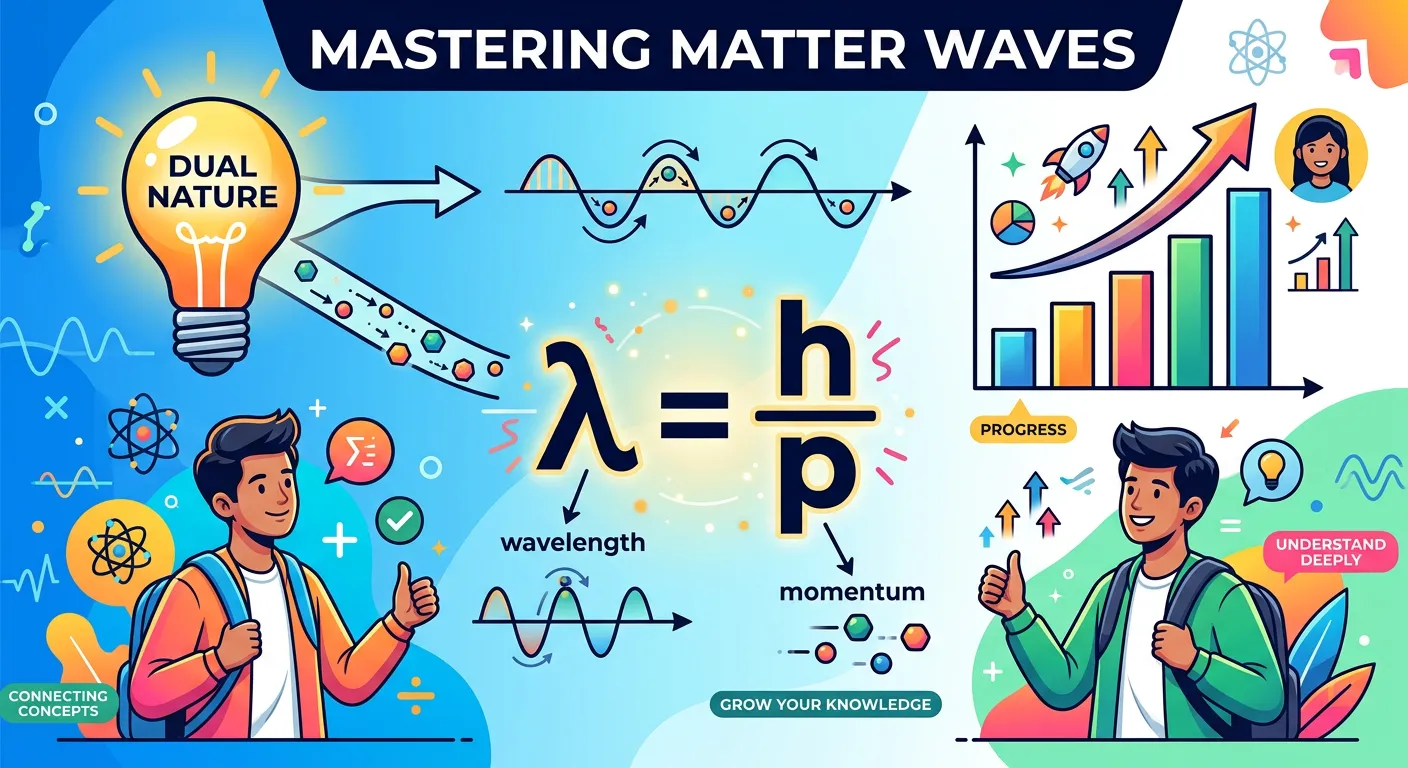
“So, matter is both a particle and a wave. From the smallest electron to the biggest planet, everything has a frequency. You’ve just decoded the foundation of quantum mechanics! Keep this logic clear, and you will ace any question on de Broglie in the NEET exam.”
The de Broglie relationship is not just another formula to memorize; it is the cornerstone of modern chemistry. By proving that matter has a wave nature, de Broglie opened the door for Werner Heisenberg’s Uncertainty Principle and Erwin Schrödinger’s Wave Equation. Without the concept of matter waves, we would still be stuck with the 'solar system' model of the atom, unable to explain chemical bonding, the periodic table's structure, or the behavior of semiconductors.
As you conclude this topic, remember the central takeaway: the universe is not made of either particles or waves, but entities that exhibit properties of both depending on how we measure them. This duality is the essence of quantum mechanics. For an electron in an atom, its 'wave-ness' is what allows it to exist in clouds of probability (orbitals) rather than rigid paths. This transition from 'certainty' in classical mechanics to 'probability' in quantum mechanics is the most significant shift in scientific history.
For your NEET preparation, ensure you have a firm grasp of the inverse relationships: mass vs. wavelength and velocity vs. wavelength. Practice the conversion between energy, potential, and wavelength until it becomes second nature. This topic bridge Units 2 (Atomic Structure) with Class 12 Physics (Dual Nature of Matter), making it a high-value area for scoring. By mastering de Broglie's relationship, you aren't just learning a chemistry topic; you're learning the fundamental language of the universe.
Quick Revision Points
- Matter-wave duality is a fundamental pillar of modern science.
- It led directly to the development of quantum mechanical models and orbitals.
- Wave nature is a consequence of motion; at rest, wavelength is undefined.
- De Broglie's work reconciled the particle and wave views of the universe.
- Understanding this topic simplifies many concepts in Class 12 Physics.
NEET Exam Angle
- Final check: Do you know the value of $h$? Can you calculate $\sqrt{150}$? Do you remember $m_e$?
- Be prepared for 'assertion-reason' questions regarding the stability of Bohr orbits.
- Review the experimental proof (Davisson-Germer) as it is occasionally asked in the context of 'who proved it?'
Recommended Reading
Explore related Biology topics to build deeper chapter connections for NEET.
- Nature of Electromagnetic Radiation · Topic 2.1
- Quantum Mechanical Model · Topic 2.12
- Atomic Orbitals · Topic 2.13
- Significance of Quantum Numbers · Topic 2.16
- Photoelectric Effect · Topic 2.2
- Bohr Model · Topic 2.4
- Jump to Key Terms (Quick Revision)
- Review Common NEET Mistakes
- Read Topic FAQs
- Check PYQ Pattern Notes
- Practice NEET MCQs
- Solve NEET PYQs
📚 Key Terms
⚠️ Common NEET Mistakes
- 1Forgetting to convert mass from grams to kilograms in numerical problems.
- 2Confusing matter waves with electromagnetic waves (matter waves do not consist of electric/magnetic fields).
- 3Using the speed of light (c) instead of the particle's actual velocity (v) in the formula λ = h/mv.
- 4Applying de Broglie's formula to stationary particles (wavelength is only defined for moving particles).
- 5Mistaking the relationship with Kinetic Energy as λ ∝ 1/E instead of the correct λ ∝ 1/√E.
📝 NEET PYQ Pattern
In NEET 2018–2024, de Broglie's relationship consistently appears as numerical problems requiring the calculation of wavelength from Kinetic Energy or accelerating potential. There is a frequent pattern of comparing the ratio of wavelengths for different particles (like protons, deuterons, and alpha particles) moving with the same velocity or energy.
❓ Frequently Asked Questions
Why is the wave nature of a moving car not visible in daily life?
A car has a very large mass compared to subatomic particles. Since wavelength (λ) is inversely proportional to mass (λ = h/mv), the wavelength of a moving car is extremely small (around 10⁻³⁸ m), which is far below the limits of detection and doesn't affect its macroscopic movement.
What is the mathematical relationship between de Broglie wavelength and Kinetic Energy?
The relationship is λ = h / √(2mE), where h is Planck's constant, m is the mass of the particle, and E is its kinetic energy. This shows that wavelength is inversely proportional to the square root of the kinetic energy.
How does the de Broglie hypothesis support Bohr’s quantization of angular momentum?
De Broglie proposed that an electron orbit is stable only if it forms a standing wave, which requires the circumference (2πr) to be an integral multiple of the wavelength (nλ). Substituting λ = h/mv into 2πr = nλ yields mvr = nh/2π, which is Bohr's postulate.
Does de Broglie's relationship apply only to electrons or to all material particles?
It applies to all material particles in motion, regardless of their size. However, the wave properties are only significant and measurable for microscopic particles like electrons, protons, and neutrons due to their small mass.
How does the wavelength change if the velocity of an electron is doubled?
Since wavelength is inversely proportional to velocity (λ ∝ 1/v), if the velocity is doubled, the de Broglie wavelength will be halved (become λ/2).
What are the SI units for all variables in the de Broglie equation λ = h/mv?
The SI units are: Wavelength (λ) in meters (m), Planck's constant (h) in Joule-seconds (J·s or kg·m²/s), mass (m) in kilograms (kg), and velocity (v) in meters per second (m/s).
Written By
NEET Content Strategist & Biology Expert
Sangita Kumari is a NEET educator and content strategist with over 6 years of experience teaching Biology, Chemistry, and Physics to Class 11 and 12 aspirants. She helps bridge the gap between traditional NCERT preparation and modern AI-powered learning. Her content is trusted by thousands of NEET aspirants across India.