🎬 Video Lesson Available
Watch the full 7-slide video lesson for Mole Concept with AI teacher narration and visual explanations.
01Understanding the Mole: Chemistry’s Universal Counting Unit

“Welcome, NEET aspirants! Imagine you are buying a dozen eggs or a gross of pencils. In chemistry, we deal with tiny atoms, so we use a special 'baker's dozen' called a Mole. It is our magical unit to count atoms, molecules, and ions in one go!”
Welcome to one of the most fundamental pillars of your NEET chemistry preparation. In our everyday lives, we use collective terms to simplify counting. If you go to a bakery, you ask for a 'dozen' eggs; if you are in a stationery shop, you might buy a 'gross' of pencils. In chemistry, however, we deal with entities so unimaginably small—atoms, ions, and molecules—that counting them by the dozen would be like trying to empty the ocean with a teaspoon. This is where the Mole concept enters the stage as the SI base unit for the amount of substance.
The mole serves as the vital bridge between the microscopic world of individual atoms and the macroscopic world of grams and liters that we handle in the laboratory. By definition, a mole is the amount of substance that contains as many elementary entities as there are atoms in exactly 12 grams of the Carbon-12 isotope. This provides a standardized reference point that allows chemists across the globe to communicate quantitative data with absolute precision. Without the mole, calculating the exact amount of a reactant needed to produce a specific amount of medicine or industrial chemical would be impossible.
Historically, the mole was developed because chemical reactions occur based on the number of particles, not necessarily their mass. For instance, one atom of sodium reacts with one atom of chlorine, but their masses are vastly different. To reflect this reality in the lab, we needed a unit that counts by number while allowing us to weigh by mass. Understanding this conceptual shift from 'how much does it weigh' to 'how many particles are there' is your first step toward mastering stoichiometry for the NEET exam.
Quick Revision Points
- The mole is the 7th SI base unit, denoted by the symbol 'mol'.
- It measures the 'amount of substance', not the mass or volume directly.
- 1 mole is defined based on the number of atoms in 0.012 kg of C-12.
- It provides a link between microscopic particles and macroscopic laboratory measurements.
- The term 'mole' was introduced by Wilhelm Ostwald in 1896.
NEET Exam Angle
- Questions often test your understanding of the mole as a fundamental unit versus a derived unit.
- You must distinguish between '1 mole of atoms' and '1 mole of molecules' (e.g., 1 mole of O2 vs. 1 mole of O).
- Always look for the 'Carbon-12' reference in conceptual MCQ options.
| Unit | Represents | Used For |
|---|---|---|
| Dozen | 12 items | Macroscopic objects |
| Gross | 144 items | Bulk stationery/goods |
| Mole | 6.022 × 10^23 | Atoms, ions, electrons |
02Avogadro’s Number: The Magnitude of 6.022 × 10^23

“Meet Avogadro's Number: 6.022 times 10 to the power of 23. It is a massive number! Just like a 'dozen' represents 12 items, a 'mole' represents this giant count. Whether it is atoms or molecules, this constant is your ultimate key to unlocking stoichiometry.”
To truly appreciate the mole concept, we must grasp the sheer scale of Avogadro’s Number (NA). This constant, valued at approximately 6.022 × 10^23, represents the number of particles in one mole of any substance. To give you some perspective, if you had a mole of marbles, they would cover the entire surface of the Earth to a depth of several miles! In chemistry, however, atoms are so small that this enormous number of particles fits easily within a few grams of matter.
In the NEET syllabus, Avogadro's number is not just a constant to memorize; it is a versatile tool. You will be asked to apply it across different chemical entities. For example, one mole of water (H2O) contains 6.022 × 10^23 molecules, but it contains three times that many atoms (two moles of Hydrogen atoms and one mole of Oxygen atoms). Being able to dissect a molecule into its constituent atoms and then applying NA is a high-yield skill for the exam.
Furthermore, this constant applies to subatomic particles as well. We often talk about 'a mole of electrons' in electrochemistry (which is associated with Faraday's constant). The magnitude of NA ensures that even the most minute chemical changes involve a count of particles that is statistically significant and measurable. When you see a stoichiometry problem, your first instinct should be to ask: "How many Avogadro units are we dealing with here?"
Quick Revision Points
- Avogadro’s Constant (NA) = 6.02214076 × 10^23 mol⁻¹.
- It is the proportionality factor that relates the molar mass of a substance to the mass of a single sample.
- NA is named after Amedeo Avogadro, though he did not determine the value himself.
- This number is the same for all substances, whether they are elements, compounds, or ions.
NEET Exam Angle
- Practice calculating the number of ions in ionic compounds (e.g., number of Cl- ions in 1 mole of CaCl2).
- NEET frequently asks for the 'total number of electrons' in a given weight of a compound—this requires multiplying moles by electrons per molecule, then by NA.
- Watch out for units; NA is expressed 'per mole'.
| Substance | 1 Mole contains... | Total Atoms/Particles |
|---|---|---|
| Helium (He) | 1 mole of He atoms | 6.022 × 10^23 |
| Oxygen (O2) | 1 mole of O2 molecules | 1.204 × 10^24 |
| Water (H2O) | 1 mole of H2O molecules | 1.806 × 10^24 |
03Molar Mass: Linking Atomic Scale to the Laboratory Balance
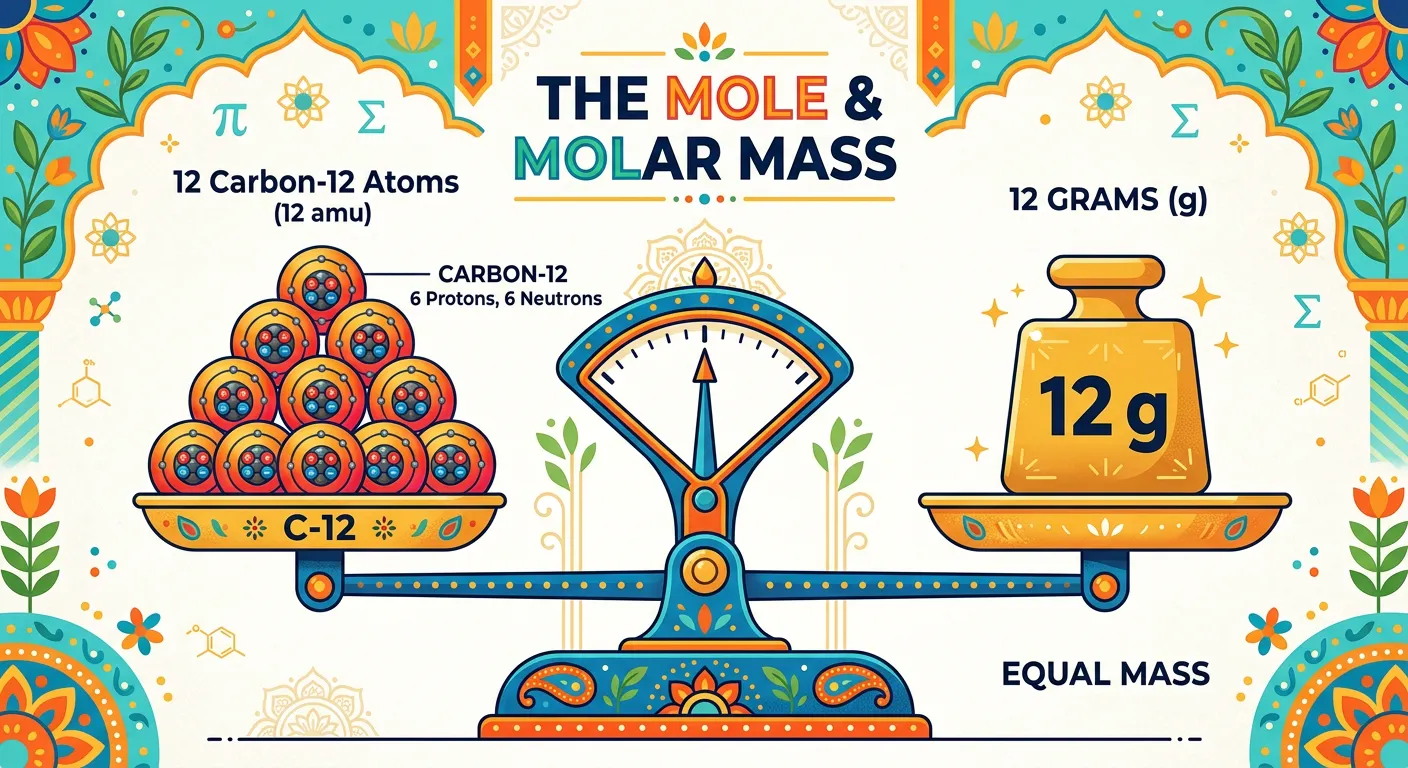
“Think of Molar Mass as the 'weight of the dozen'. For Carbon, 1 mole of atoms weighs exactly 12 grams. It is the bridge between the microscopic world of atoms and the macroscopic world of the weighing balance we use in our labs.”
Molar mass is the physical property defined as the mass of one mole of a substance. It is usually expressed in grams per mole (g/mol). This concept is the 'secret sauce' of chemistry because it numerically equals the atomic or molecular mass of a substance but changes the units from atomic mass units (amu) to grams. While one atom of Carbon weighs 12 amu (an invisible quantity), one mole of Carbon weighs 12 grams (something you can hold in your hand).
To calculate the molar mass of a compound (Gram Molecular Mass or GMM), you simply sum the atomic masses of all the constituent atoms as listed on the periodic table. For instance, for Glucose (C6H12O6), you calculate: (6 × 12) + (12 × 1) + (6 × 16) = 180 g/mol. This value tells us that if we weigh 180 grams of glucose on a lab balance, we are holding exactly 6.022 × 10^23 molecules of glucose. This link is vital for performing experiments where you need to react specific ratios of substances.
Carbon-12 remains the gold standard for these definitions. Because the amu is defined as 1/12th the mass of a C-12 atom, the molar mass of any element in g/mol is numerically identical to its average atomic mass in amu. This elegant symmetry allows students to use periodic table values interchangeably for single-atom calculations and bulk-substance calculations, provided they respect the units.
Quick Revision Points
- Gram Atomic Mass (GAM) is the mass of 1 mole of atoms of an element.
- Gram Molecular Mass (GMM) is the mass of 1 mole of molecules of a compound.
- Molar mass = Mass of substance (g) / Number of moles (mol).
- Carbon-12 is used as the reference isotope for defining these masses.
NEET Exam Angle
- NEET often gives atomic masses in the question paper, but you should memorize the first 20 elements plus common ones like Fe, Cu, Ag, and Ba.
- Distinguish between 'molecular mass' (mass of one molecule in amu) and 'molar mass' (mass of 6.022 × 10^23 molecules in grams).
- Calculation speed is key: practice summing masses for common molecules like H2SO4 (98), NaOH (40), and CaCO3 (100).
04The Mole Bridge: Navigating Mass-Mole-Particle Interconversions
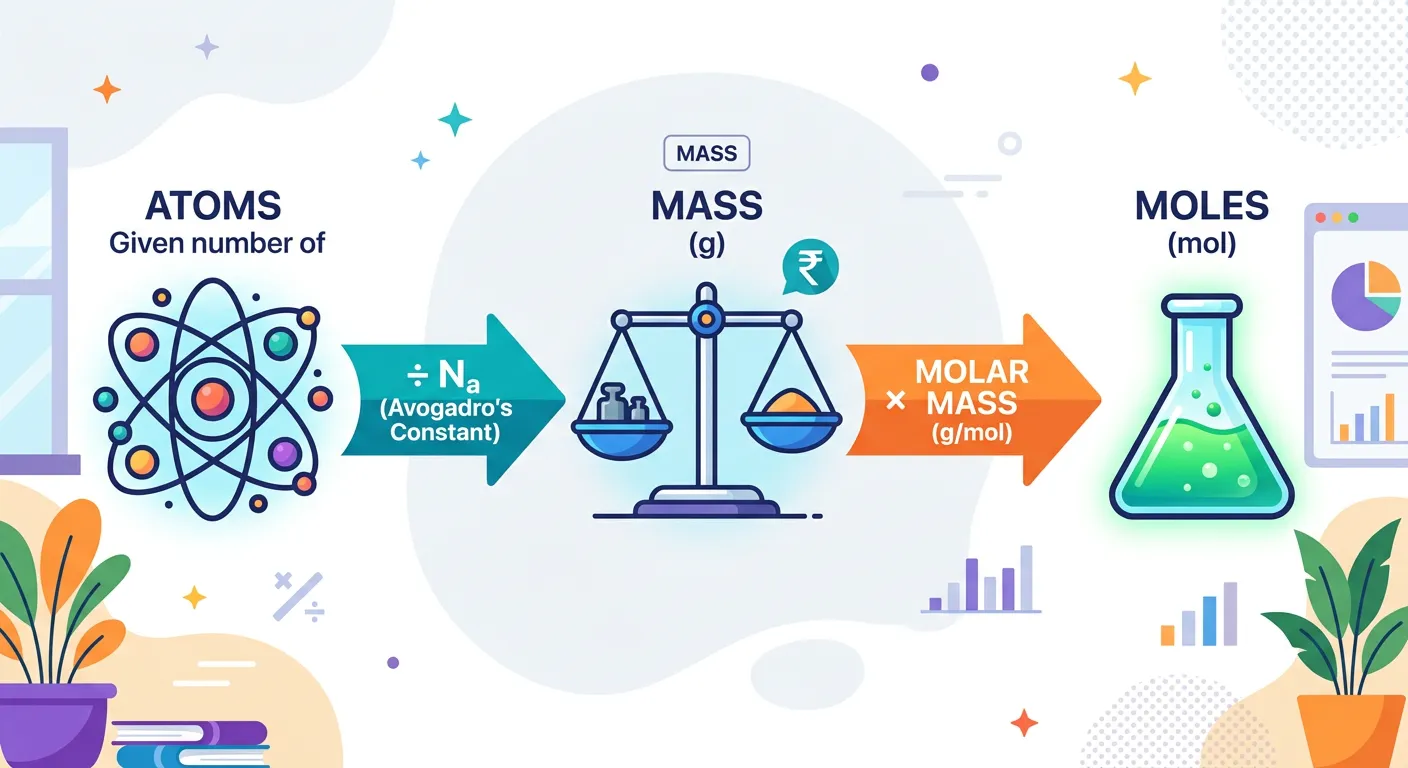
“To ace your NEET exam, master the Mole bridge! If you have the mass, divide by Molar Mass to get moles. If you have the number of particles, divide by Avogadro's constant. It is just like converting currency—keep your units straight, and you will never go wrong!”
Mastering the 'Mole Bridge' is the single most effective way to improve your score in NEET Chemistry. Almost every numerical problem in this unit requires you to convert between three main pillars: Mass, Moles, and Number of Particles. The mole sits at the center of this map. To go from Mass (m) to Moles (n), you divide by the Molar Mass (M). To go from Moles to the Number of Particles (N), you multiply by Avogadro's Constant (NA).
One of the biggest hurdles for students is 'dimensional analysis' or simply keeping track of units. If you are given 54 grams of water and asked for the number of molecules, don't try to jump directly from grams to molecules. Always stop at the 'Mole Station' first! Step 1: 54g H2O / 18g/mol = 3 moles. Step 2: 3 moles × 6.022 × 10^23 = 1.806 × 10^24 molecules. This step-by-step approach prevents silly mistakes and ensures that your units cancel out correctly, leaving you with the desired answer.
Another common pitfall involves the distinction between atoms and molecules. For a diatomic gas like Nitrogen (N2), one mole of the gas is one mole of molecules, but it contains two moles of atoms. If a NEET question asks for the 'number of atoms in 14g of Nitrogen gas', you must calculate the moles of N2 first, then account for the two atoms per molecule. Ignoring the atomicity of the molecule is a 'trap' often set by exam paper setters to filter out careless candidates.
Quick Revision Points
- Formula 1: n = given mass (w) / molar mass (M).
- Formula 2: n = given number of particles (N) / Avogadro’s Number (NA).
- Always convert to moles first before solving for other quantities.
- Dimensional analysis: Unit cancellation ensures the final answer is in the correct units.
NEET Exam Angle
- High-frequency question: Comparing the number of atoms in different masses of various elements (e.g., 1g Au vs 1g Na).
- Pro Tip: If the masses are equal, the substance with the lowest molar mass has the highest number of moles/atoms.
- Look out for 'total number of ions' in electrolytes; remember to multiply by the Van't Hoff factor (i).
| Conversion Path | Operation | Formula |
|---|---|---|
| Mass → Moles | Divide by Molar Mass | n = m / M |
| Moles → Mass | Multiply by Molar Mass | m = n × M |
| Moles → Particles | Multiply by NA | N = n × NA |
| Particles → Moles | Divide by NA | n = N / NA |
05Molarity and Concentration: Measuring Moles in Solution

“What about liquids? We use Molarity. It is simply moles of solute dissolved in one liter of solution. Imagine adding two spoons of sugar to a cup of tea; Molarity tells us how 'strong' or concentrated that solution is. Simple, right?”
In medical and pharmaceutical contexts, most chemistry happens in solutions. Whether it is a saline drip or a chemical reagent, we need a way to express how many moles of a substance are present in a given volume of liquid. This is the essence of Concentration. Molarity (M) is the most frequently used concentration term in NEET. It is defined as the number of moles of solute dissolved in one liter (or one cubic decimeter) of solution.
The physical meaning of Molarity is simple: it tells us how 'crowded' the solute particles are. A 2M (2 Molar) solution of HCl is more concentrated and aggressive than a 0.1M solution because it contains twenty times more moles of acid in the same volume. When solving problems, remember the golden rule: Volume must be in Liters. If you are given milliliters (mL), divide by 1000 before proceeding with your Molarity formula.
A crucial conceptual point for NEET is the effect of temperature. Since Molarity depends on the volume of the solution, and volume changes with temperature (due to expansion or contraction), Molarity is temperature-dependent. This is why in some high-precision experiments, we use Molality (moles per kg of solvent), which is mass-based and stays constant regardless of the weather. Keep this distinction in mind for 'Assertion-Reason' type questions.
Quick Revision Points
- Molarity (M) = Moles of solute / Volume of solution in Liters.
- Unit: mol/L or Molar (M).
- Dilution Formula: M1V1 = M2V2 (Used when adding water to change concentration).
- Mixing Formula: M_res = (M1V1 + M2V2) / (V1 + V2).
NEET Exam Angle
- Expect questions linking Molarity to Density and Mass Percentage.
- Remember: M = (mass % × 10 × density) / Molar Mass of solute. This shortcut saves minutes!
- Watch for 'millimoles' (mmol); 1 mole = 1000 mmol. Using mmol can often simplify calculations involving small volumes.
- Molarity is temperature-dependent, while Molality and Mole Fraction are not.
06Molar Volume of Gases: The 22.4 Liter Golden Rule
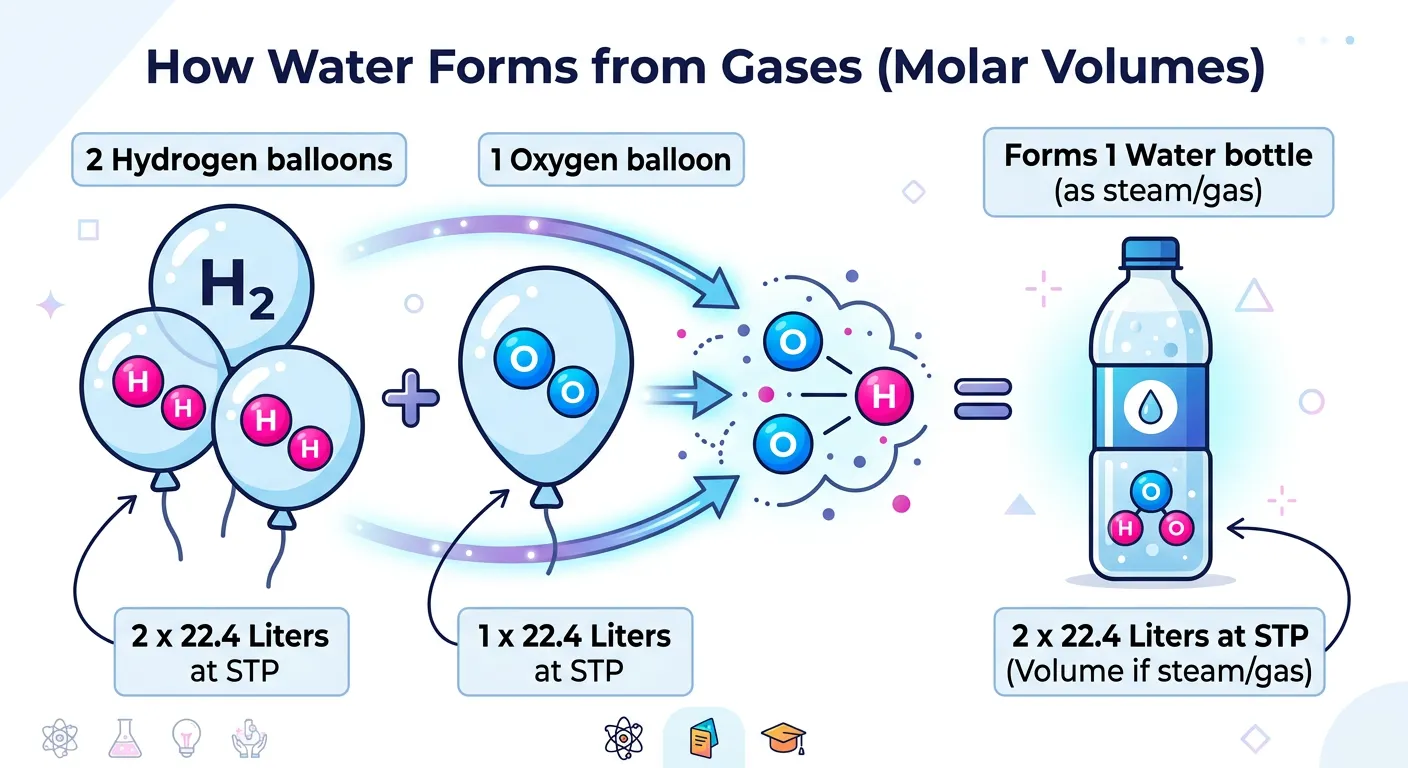
“Gases love the Mole concept too! At Standard Temperature and Pressure, exactly one mole of any ideal gas occupies 22.4 liters. It is a fixed, golden rule that makes calculating gas volumes in reactions a piece of cake for your upcoming chemistry paper.”
When dealing with gases, we have a unique advantage. According to Avogadro’s Hypothesis, equal volumes of all gases at the same temperature and pressure contain the same number of molecules. This led to the discovery of Molar Volume. At Standard Temperature and Pressure (STP), exactly one mole of any ideal gas occupies a volume of approximately 22.4 liters. This is a 'magic number' that simplifies gas stoichiometry significantly.
It is important to be precise about what 'STP' means. In older conventions still often used in NEET, STP is 0°C (273.15 K) and 1 atm pressure. Under these conditions, the volume is 22.414 L. The newer IUPAC standard uses 1 bar pressure instead of 1 atm, which slightly changes the volume to 22.7 L. However, for most NEET numericals, unless specified otherwise, the 22.4 L value is your go-to constant for calculation.
This rule applies only to gases, and only under ideal behavior. Do not mistakenly apply 22.4L to liquids like water or solids like glucose! If you have a mixture of gases, the rule still holds; one mole of the total mixture will occupy 22.4L at STP. This is incredibly useful for calculating the results of combustion reactions where the reactants and products are gaseous.
Quick Revision Points
- 1 mole of any ideal gas at STP = 22.4 L.
- Moles of gas (n) = Given Volume at STP / 22.4 L.
- This relationship stems from the Ideal Gas Equation (PV = nRT).
- Molar volume is independent of the identity of the gas (O2, H2, CO2 all occupy the same volume per mole).
NEET Exam Angle
- Frequent trap: Giving the volume of a liquid (like H2O at 25°C) and expecting you to divide by 22.4. Only use this for gases!
- If conditions are not at STP, use PV = nRT or the combined gas law to find the volume first.
- Be aware of the density of gases; Density = Molar Mass / Molar Volume.
| Condition | Temperature | Pressure | Molar Volume |
|---|---|---|---|
| STP (Old/NEET) | 273.15 K | 1 atm | 22.4 L |
| STP (IUPAC) | 273.15 K | 1 bar | 22.7 L |
| NTP | 293.15 K | 1 atm | ~24.0 L |
07Mastering Stoichiometry: Strategy for Competitive Success

“You have cracked the code! From atoms to grams, and liters to moles, you now have the tools to solve any stoichiometry problem. Keep practicing these conversions, stay curious, and you are well on your way to that dream medical seat. Keep shining, future doctors!”
Now that you have the individual pieces of the puzzle—mass, moles, particles, and volume—it is time to integrate them into the master skill of Stoichiometry. Stoichiometry is the study of the quantitative relationships between the amounts of reactants used and products formed in a chemical reaction. A balanced chemical equation is essentially a recipe: the coefficients (numbers in front of formulas) represent the 'mole ratio' of the participants.
In the NEET exam, you won't just get direct mole questions. You will get multi-step problems. For example: "How many grams of CO2 are produced by burning 16g of CH4?" Step 1: Balance the equation (CH4 + 2O2 → CO2 + 2H2O). Step 2: Convert mass to moles (16g CH4 = 1 mole). Step 3: Use the mole ratio (1 mole CH4 gives 1 mole CO2). Step 4: Convert moles back to the desired unit (1 mole CO2 = 44g). Learning to flow through this sequence without hesitation is what separates top rankers from the rest.
Time management is critical. During the exam, don't write out every single word of the formula. Use mental shortcuts. If you see '100g of CaCO3', immediately think '1 mole'. If you see '22.4L of O2 at STP', think '1 mole'. Developing this 'chemical intuition' allows you to solve stoichiometry problems in 30-45 seconds, giving you more time for complex Physics or Biology questions. Keep practicing, and the mole will become your strongest ally in the NEET paper.
Quick Revision Points
- Stoichiometry relies on the Law of Conservation of Mass.
- Coefficients in a balanced equation represent moles, not grams.
- Limiting Reagent: The reactant that is completely consumed first and limits the amount of product formed.
- Always check if the given equation is balanced before starting calculations.
NEET Exam Angle
- Limiting reagent problems are a NEET favorite. Divide given moles by the stoichiometric coefficient; the smallest value identifies the limiting reagent.
- Percentage yield and purity calculations are frequently combined with mole concept questions.
- The 'total number of atoms' in a specific mass of a reactant is a recurring theme in PYQs (Previous Year Questions).
- Remember that the 'mole ratio' is the only way to compare different substances in a reaction.
Recommended Reading
Explore related Biology topics to build deeper chapter connections for NEET.
- Chemical Equations · Topic 1.10
- Dalton's Atomic Theory · Topic 1.2
- Laws of Chemical Combination · Topic 1.4
- Molar Mass · Topic 1.7
- Nature of Electromagnetic Radiation · Topic 2.1
- Photoelectric Effect · Topic 2.2
- Jump to Key Terms (Quick Revision)
- Review Common NEET Mistakes
- Read Topic FAQs
- Check PYQ Pattern Notes
- Practice NEET MCQs
- Solve NEET PYQs
📚 Key Terms
⚠️ Common NEET Mistakes
- 1Using 22.4L for liquids or solids in volume-to-mole conversions.
- 2Forgetting to convert Celsius to Kelvin (K = °C + 273) in gas calculations.
- 3Confusing 'mole of atoms' with 'mole of molecules' for diatomic gases like O2 or N2.
- 4Not checking if a chemical equation is balanced before performing stoichiometric calculations.
- 5Using mass of solvent instead of volume of solution when calculating Molarity.
- 6Ignoring the units of atomic mass units (amu) versus grams (g) when discussing single atoms vs bulk matter.
📝 NEET PYQ Pattern
Between 2018–2024, NEET has consistently featured 1–2 direct questions on the Mole Concept, specifically focusing on the number of atoms in different masses and Molarity calculations. A recurring trend involves calculating the total number of electrons or ions in a given mole of a substance, requiring a two-step conversion process.
❓ Frequently Asked Questions
How do I calculate the number of atoms in a given mass of a compound?
First, convert the given mass to moles by dividing by the molar mass of the compound. Then, multiply the number of moles by Avogadro's number (6.022 × 10^23) to find the number of molecules. Finally, multiply by the number of atoms present in a single molecule (the atomicity) to get the total number of atoms.
Why is 22.4L used only for gases at STP and not for liquids?
The value 22.4L is derived from the Ideal Gas Law (PV=nRT), which assumes that the particles have negligible volume and no intermolecular forces. This is approximately true for gases. In liquids, particles are closely packed and have strong interactions, so their volume depends heavily on their specific density and molecular structure, not a universal gas constant.
What is the difference between molarity and molality in NEET problems?
Molarity (M) is moles of solute per liter of solution and is temperature-dependent because volume changes with temperature. Molality (m) is moles of solute per kilogram of solvent and is temperature-independent. NEET often asks about this distinction in conceptual questions regarding concentration stability.
Does Avogadro's number change if the isotope of Carbon is changed?
No, Avogadro's number is a fixed fundamental constant. However, the definition of the mole is specifically based on Carbon-12. If the reference standard were changed (e.g., to Oxygen-16), the numerical value of NA would shift slightly to maintain the relationship between mass and particle count, but for all practical calculations in the current system, NA is constant.
How do I quickly convert volume to moles when the gas is not at STP?
If the gas is not at STP, you must use the Ideal Gas Equation: n = PV / RT. Ensure that pressure (P) is in atm, volume (V) is in liters, and temperature (T) is in Kelvin (Celsius + 273.15). Use the gas constant R = 0.0821 L⋅atm/(K⋅mol).
Why is the mole considered a counting unit rather than just a weight?
A mole is a counting unit because chemical reactions occur between discrete particles (atoms/molecules) in fixed whole-number ratios. Weight varies because different atoms have different masses (e.g., 1 mole of H weighs 1g while 1 mole of C weighs 12g), but both contain the exact same number of reactive particles.
Written By
NEET Content Strategist & Biology Expert
Sangita Kumari is a NEET educator and content strategist with over 6 years of experience teaching Biology, Chemistry, and Physics to Class 11 and 12 aspirants. She helps bridge the gap between traditional NCERT preparation and modern AI-powered learning. Her content is trusted by thousands of NEET aspirants across India.