🎬 Video Lesson Available
Watch the full 7-slide video lesson for Laws of Chemical Combination with AI teacher narration and visual explanations.
01Antoine Lavoisier and the Law of Conservation of Mass

“Welcome, future doctors! Chemistry isn't just formulas; it's a balance sheet. Antoine Lavoisier proved that matter is neither created nor destroyed. Think of your kitchen: if you combine 10 grams of flour and 2 grams of sugar, you must get exactly 12 grams of dough. Mass is conserved!”
In the late 18th century, Antoine Lavoisier revolutionized chemistry by introducing the concept of quantitative measurement. Before him, chemistry was often mystical and vague; Lavoisier turned it into a rigorous science of the 'balance sheet.' He proposed that in any physical or chemical change, the total mass of the products remains exactly equal to the total mass of the reactants. This means matter cannot be created or destroyed out of nothing; it simply changes form. For a NEET aspirant, this is the fundamental reason why we must balance every chemical equation we write.
To visualize this, imagine you are baking in your kitchen. If you take 500 grams of flour, 100 grams of butter, and 200 grams of sugar, the resulting dough must weigh exactly 800 grams. If it weighs less, some 'matter' likely escaped as steam; if it weighs more, you probably added an ingredient you forgot to record. In the laboratory, Lavoisier demonstrated this by burning phosphorus and sulfur in lead containers, proving that the mass gained by the phosphorus was exactly equal to the mass lost by the surrounding air. This law forms the bedrock of stoichiometry, allowing us to predict exactly how much product will form from a specific amount of starting material.
However, as you progress into Modern Physics, you will encounter a slight exception: nuclear reactions. In these high-energy processes, a tiny amount of mass is converted into energy according to Einstein’s equation, $E=mc^2$. For standard chemical reactions involving electrons and bonds, this mass change is so infinitesimal that it is ignored, but for NEET, remember that the law is strictly applicable only to non-nuclear chemical processes.
Quick Revision Points
- Father of Chemistry: Antoine Lavoisier established mass conservation in 1789.
- Mass Balance: Total mass of reactants = Total mass of products.
- Physical vs. Chemical: The law holds true for both melting ice (physical) and burning magnesium (chemical).
- Foundation: This law is the logical basis for the 'Principle of Atom Conservation' (POAC) used in complex numericals.
NEET Exam Angle
- Numerical Check: Always ensure the sum of weights on the left side equals the right side in a closed system.
- Limitation: In nuclear reactions, mass-energy equivalence ($E=mc^2$) applies, making this law appear to 'fail.'
- Experimental Verification: Landolt's experiment using H-shaped tubes is often cited as classic proof for this law.
| Reaction Component | Mass Before (g) | Mass After (g) | Status |
|---|---|---|---|
| Reactants ($A + B$) | 10.50 | - | Starting Point |
| Products ($C + D$) | - | 10.50 | Conserved |
| Open Container | 10.50 | 9.20 | Mass lost as gas |
02Law of Definite Proportions: The Consistency of Chemical Composition
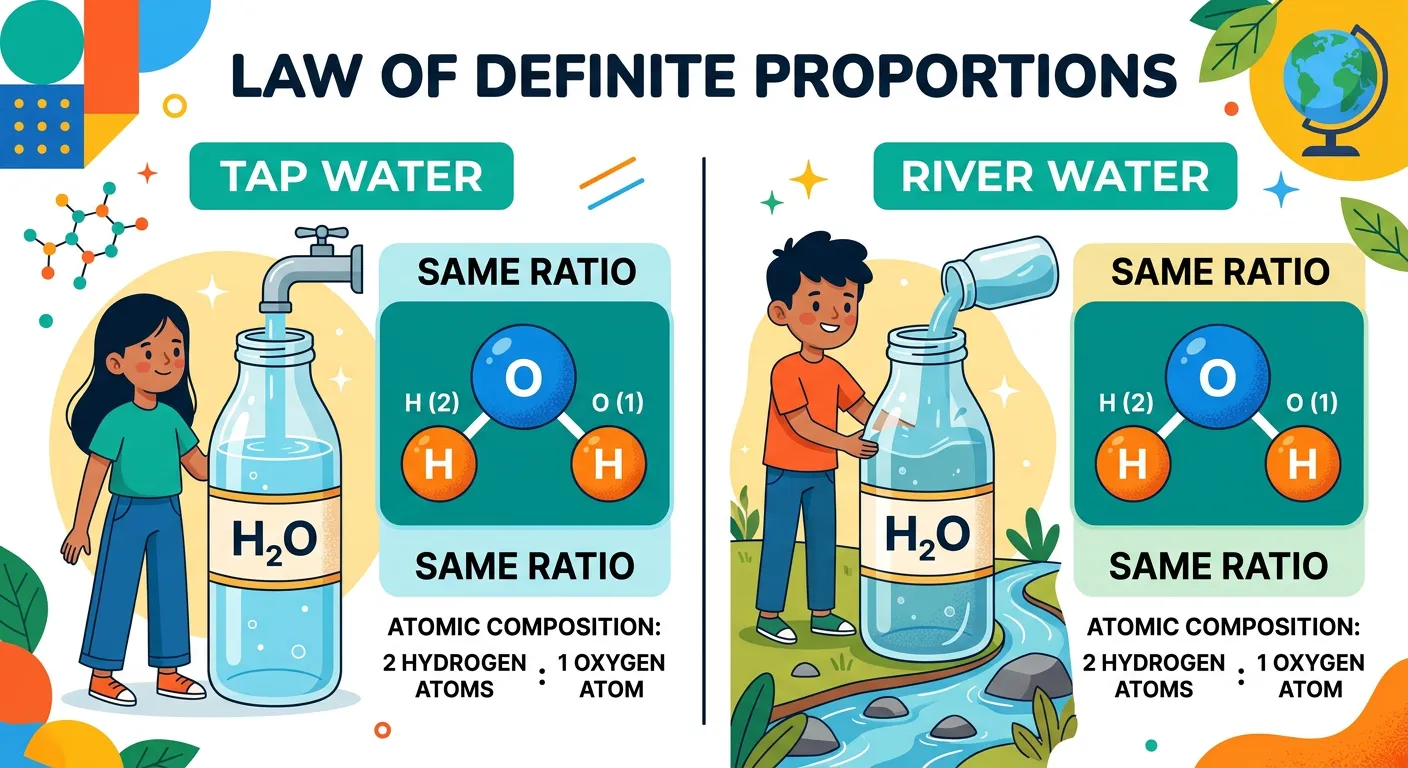
“Ever wonder if water from the Ganga is different from water in a test tube? Proust’s Law of Definite Proportions says no! A chemical compound always contains elements in a fixed ratio by mass. Whether it’s rain or a lab reaction, H2O is always chemically identical.”
Joseph Proust, a French chemist, observed that a chemical compound always contains the same elements combined together in the same proportion by mass. This is often called the Law of Constant Composition. Whether you obtain water from the holy river Ganga, from the glaciers of the Alps, or synthesize it in a high-tech laboratory in New York, the ratio of the mass of Hydrogen to the mass of Oxygen will always be 1:8. This fixed nature is what defines a 'pure' chemical compound and distinguishes it from a mixture, where components can be added in any ratio.
This law is essential for analytical chemistry. If you analyze a sample of Cupric Carbonate ($CuCO_3$), whether it is naturally occurring malachite or prepared synthetically, you will find it contains exactly 51.35% copper, 9.74% carbon, and 38.91% oxygen by weight. If the percentages differ, the substance is either impure or an entirely different compound. For students, this simplifies calculations: once you know the empirical formula of a substance, its mass percentage remains a constant, regardless of the scale of the reaction.
There are minor exceptions to this law known as 'non-stoichiometric compounds' or Berthollides (like $Fe_{0.95}O$), where the ratio of atoms is not a simple whole number due to crystal defects. However, for the scope of NEET and general Class 11 Chemistry, we treat compounds as following Proust’s law strictly. This law allows chemists to identify substances by their elemental analysis—a precursor to modern mass spectrometry.
Quick Revision Points
- Proponent: Joseph Proust (1799).
- Fixed Ratios: A compound's elemental mass ratio is independent of its source or method of preparation.
- Pure Substances: This law applies only to pure chemical compounds, not mixtures.
- Calculation Tool: Used to determine the percentage composition of elements in a given sample.
NEET Exam Angle
- Sample Comparison: If two samples of a compound from different sources show the same mass percentage of elements, they follow this law.
- Distinction: Don't confuse this with the Law of Multiple Proportions (which deals with two different compounds).
- Isotope Alert: The law slightly shifts if different isotopes are used (e.g., $D_2O$ vs $H_2O$ have different mass ratios), which is a common trick question.
| Source of Water | Mass of H (g) | Mass of O (g) | Ratio (H:O) |
|---|---|---|---|
| Tap Water | 2.0 | 16.0 | 1:8 |
| Rain Water | 1.0 | 8.0 | 1:8 |
| Lab Synthesized | 0.5 | 4.0 | 1:8 |
03Dalton’s Law of Multiple Proportions and Elemental Ratios
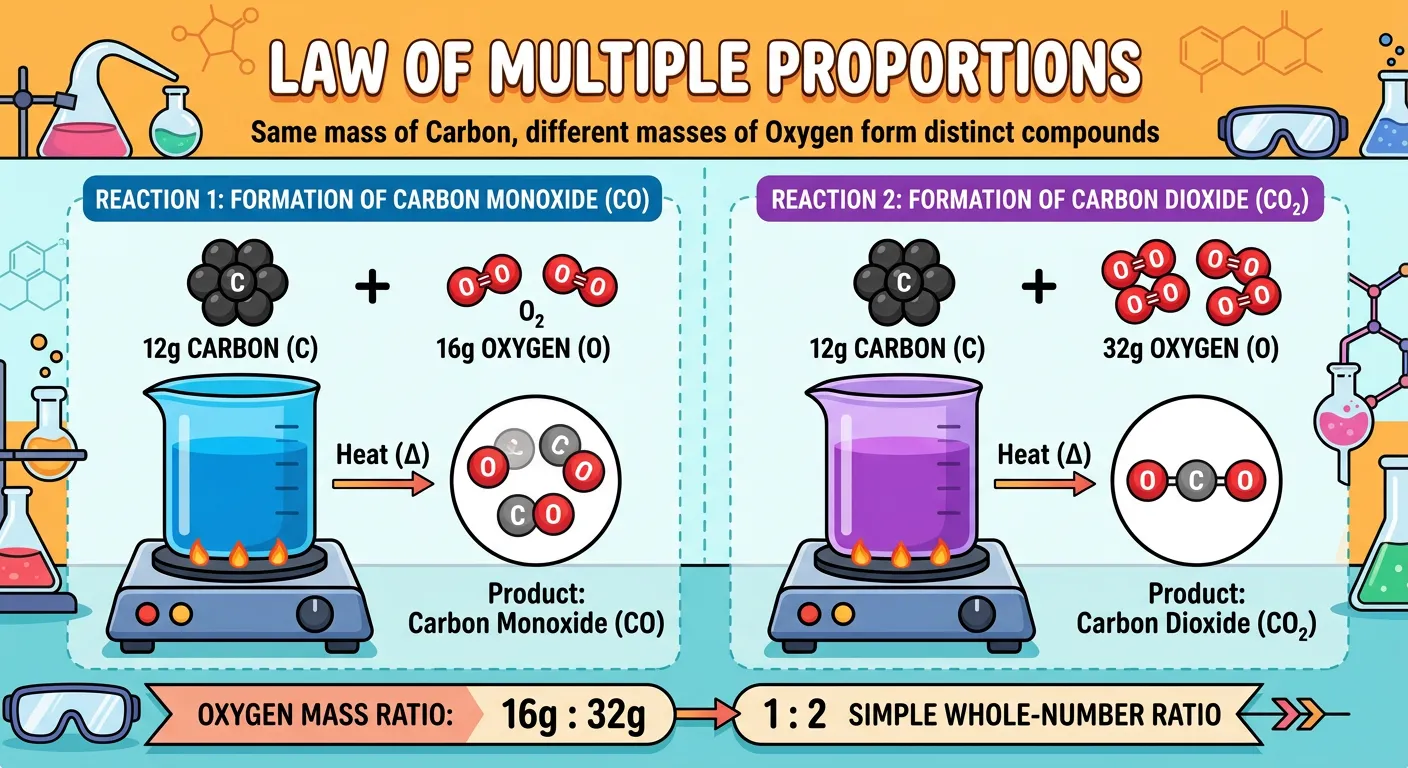
“John Dalton noticed something cool: elements can combine in different ratios to form different compounds. If 12 grams of Carbon reacts with 16 grams of Oxygen, you get CO. But with 32 grams, you get CO2. The Oxygen masses are in a simple whole-number ratio: 1:2!”
John Dalton expanded our understanding of chemical combinations by looking at elements that can form more than one compound. The Law of Multiple Proportions states that if two elements combine to form more than one compound, the masses of one element that combine with a fixed mass of the other element are in a ratio of small whole numbers. This was a groundbreaking realization because it hinted at the existence of discrete 'units' of matter—atoms.
Consider Carbon and Oxygen. They can form Carbon Monoxide ($CO$) or Carbon Dioxide ($CO_2$). In $CO$, 12g of Carbon reacts with 16g of Oxygen. In $CO_2$, 12g of Carbon reacts with 32g of Oxygen. If we fix the mass of Carbon at 12g, the ratio of Oxygen masses is 16:32, which simplifies to a perfect 1:2. This whole-number ratio is the 'fingerprint' of the Law of Multiple Proportions. It proves that atoms don't combine in random fragments; they combine in whole units.
Another classic example frequently tested in NEET involves the oxides of Nitrogen. Nitrogen and Oxygen can combine to form $N_2O, NO, N_2O_3, NO_2,$ and $N_2O_5$. By calculating the mass of oxygen that combines with a fixed mass (say 28g) of Nitrogen across all these compounds, you will find the oxygen masses follow a simple sequence: 16g, 32g, 48g, 64g, and 80g. The ratio is 1:2:3:4:5. When you see a numerical asking to identify a law based on two different compounds of the same elements, this is your go-to answer.
Quick Revision Points
- Developer: John Dalton (1803).
- Multiple Compounds: Applies when two elements form a series of different substances (like $SO_2$ and $SO_3$).
- Fixed Mass Method: Keep one element's mass constant to find the simple ratio of the second element.
- Whole Numbers: The ratios are always simple integers like 1:2, 2:3, or 1:3.
NEET Exam Angle
- Identification: If a question gives you data for two different oxides or chlorides of the same metal, it is testing this law.
- Hydrocarbon Example: Methane ($CH_4$) and Ethane ($C_2H_6$) are classic organic examples where Carbon-Hydrogen ratios follow this law.
- Data Interpretation: Practice converting percentages to mass to find the ratio of the variable element.
| Compound | Mass of N (fixed) | Mass of O | Ratio of O |
|---|---|---|---|
| $N_2O$ | 28g | 16g | 1 |
| $N_2O_3$ | 28g | 48g | 3 |
| $N_2O_5$ | 28g | 80g | 5 |
04Gay Lussac’s Law of Gaseous Volumes: The Choreography of Gases
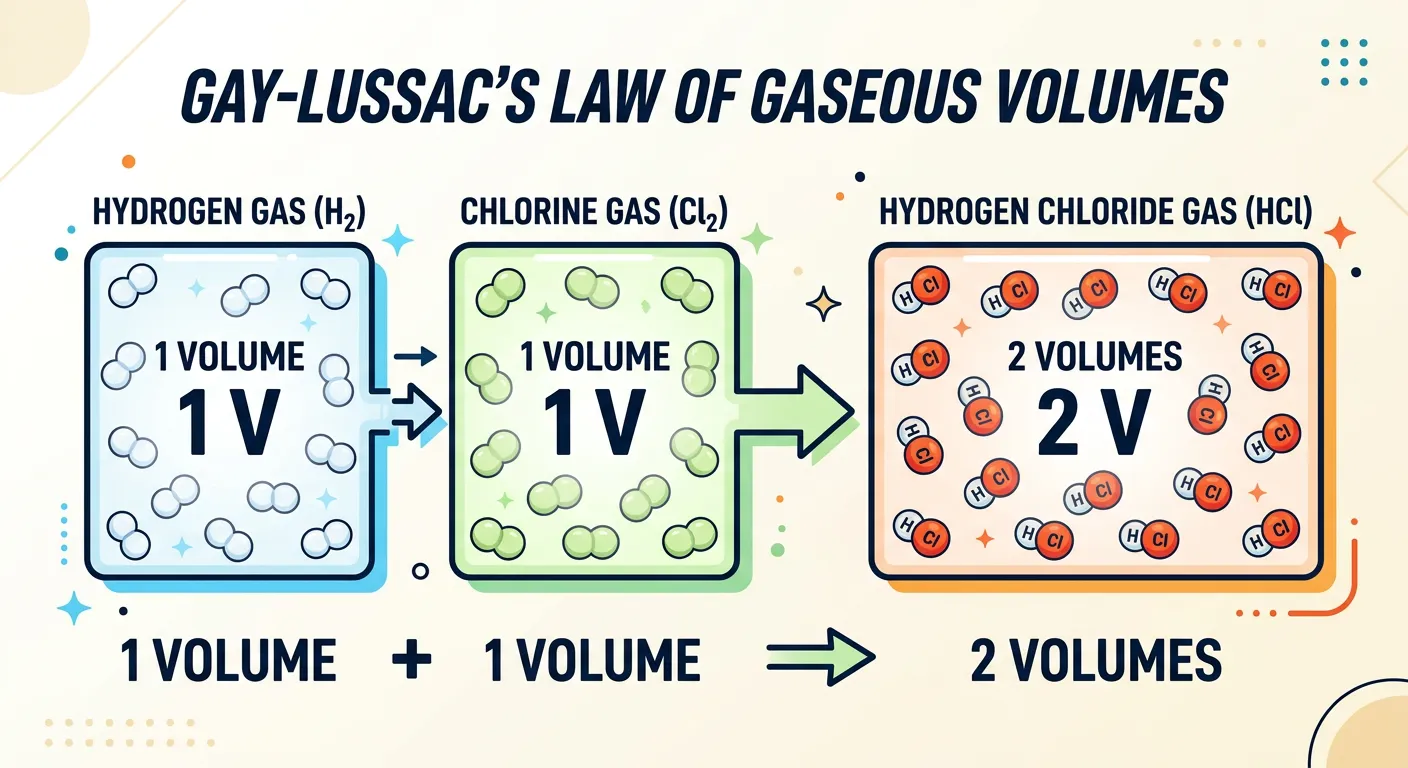
“When gases react, they do so in volumes that bear a simple ratio to one another, provided temperature and pressure stay constant. If you mix 100ml of Hydrogen and 100ml of Chlorine, you get 200ml of Hydrogen Chloride. It’s like a perfectly choreographed dance of molecules!”
While previous laws focused on mass, Joseph Louis Gay-Lussac turned his attention to the behavior of gases. He observed that when gases react together to produce other gases, their volumes bear a simple whole-number ratio to one another, provided the temperature and pressure remain constant. This was a crucial discovery because it suggested that gas volume is a direct reflection of the number of reacting particles, moving chemistry away from just weighing solids to measuring the 'space' gases occupy.
For example, consider the synthesis of ammonia: 1 volume of Nitrogen gas reacts with 3 volumes of Hydrogen gas to yield 2 volumes of Ammonia gas ($1:3:2$). If you have 100 ml of Nitrogen, you would need exactly 300 ml of Hydrogen, and you would produce exactly 200 ml of Ammonia. This simplicity is only possible because gases are mostly empty space, and the identity of the gas doesn't change the 'volume-per-particle' significantly under standard conditions.
It is important to note that Gay-Lussac’s Law only applies to gaseous reactants and products. If a reaction produces a liquid or a solid (like the formation of liquid water from hydrogen and oxygen gases), the volume of the liquid is not part of the simple whole-number ratio. This law laid the groundwork for Avogadro to later explain why these volumes behave so predictably, eventually leading to the concept of the mole.
Quick Revision Points
- Gaseous Focus: Strictly applicable to substances in the gaseous state.
- Condition: Temperature and Pressure must be kept constant throughout the measurement.
- Volume Ratio: Reactants and products relate via simple integers (e.g., 2:1:2 for water vapor synthesis).
- Non-Gaseous Exception: If a product is a liquid, its volume is ignored in the ratio calculation.
NEET Exam Angle
- Stoichiometry Short-cut: Use volume ratios directly in place of mole ratios for gas-phase reactions to save time.
- State of Matter: Always check if the reaction specifies '(g)' or '(l)'. If water is formed as a liquid, do not include its volume in the ratio.
- Combined Gas Law: Often paired with $PV=nRT$ questions to find final volumes after a reaction.
| Reaction | Volume Ratio (Reactants : Product) | Condition |
|---|---|---|
| $H_2(g) + Cl_2(g) \rightarrow 2HCl(g)$ | 1 : 1 : 2 | Const. T & P |
| $2H_2(g) + O_2(g) \rightarrow 2H_2O(g)$ | 2 : 1 : 2 | Water as Steam |
| $N_2(g) + 3H_2(g) \rightarrow 2NH_3(g)$ | 1 : 3 : 2 | Haber Process |
05Avogadro’s Law: Equal Volumes and the Molecular Guest List
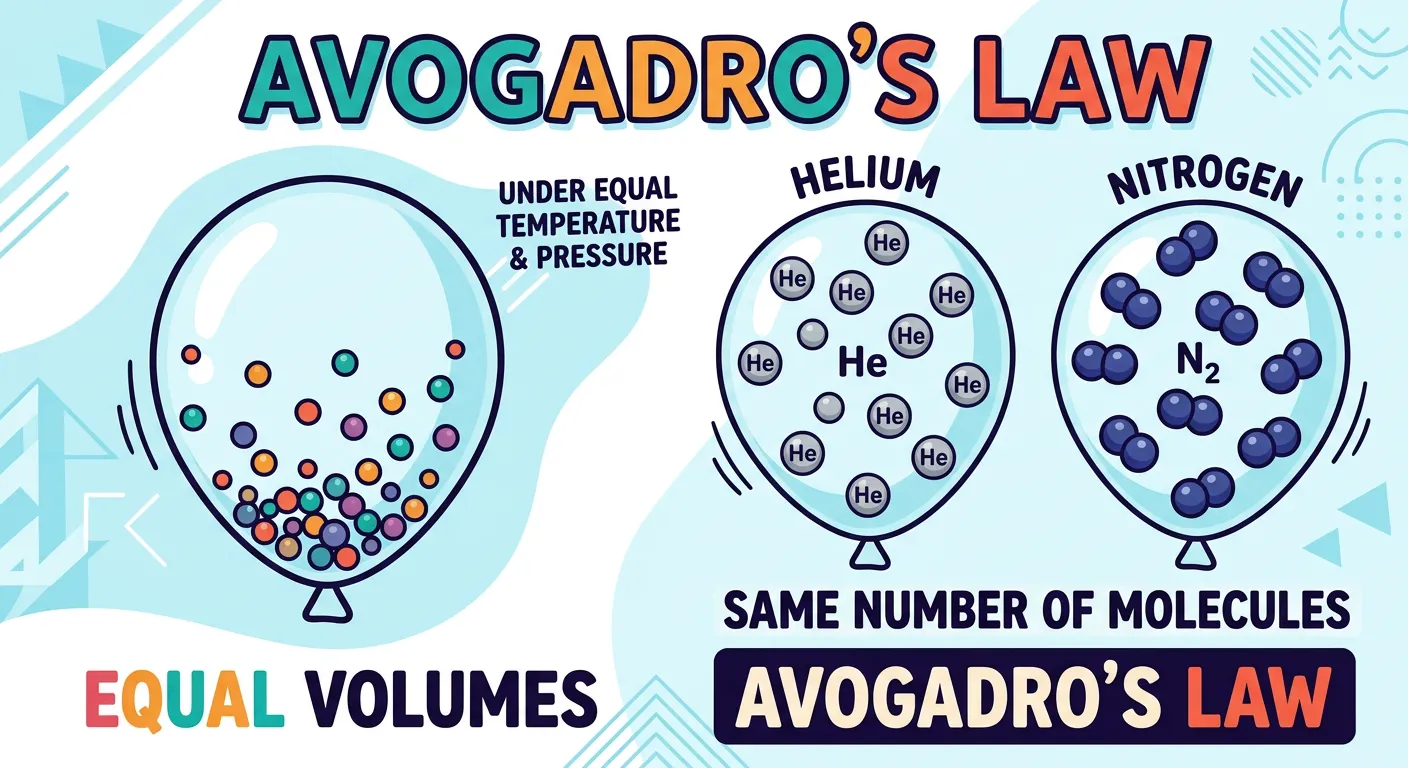
“Avogadro was a genius! He proposed that equal volumes of all gases at the same temperature and pressure contain an equal number of molecules. It doesn’t matter if the gas is heavy or light; it’s all about the space they occupy. Think of it as a uniform guest list!”
In 1811, Amedeo Avogadro provided the missing link between Gay-Lussac’s volumes and Dalton’s atoms. He proposed that equal volumes of all gases, at the same temperature and pressure, contain an equal number of molecules. This was a radical idea because it distinguished between atoms and molecules for the first time. It explained why 1 volume of Hydrogen and 1 volume of Chlorine produced 2 volumes of Hydrogen Chloride—because each 'volume' contained the same number of units, and those units (molecules) were splitting and recombining.
Avogadro’s Law implies that the volume of a gas ($V$) is directly proportional to the number of moles ($n$) of the gas ($V \propto n$). This is why we can say that 1 mole of any ideal gas at Standard Temperature and Pressure (STP) occupies exactly 22.4 liters. Whether the gas is light like Hydrogen or heavy like Carbon Dioxide, the space it occupies is determined by the number of molecules, not the size or mass of the individual molecules themselves. Think of it like a theater: if the seats are spaced equally, 100 small children (Hydrogen) take up the same number of seats as 100 large adults (CO2).
This law is the foundation for the Mole Concept. It allows chemists to 'count' atoms by measuring volumes. Without Avogadro’s insight, we wouldn't be able to determine the atomicity of gases (like why Oxygen is $O_2$ and not $O$). For NEET, understanding this law is essential for solving any problem involving the Ideal Gas Equation or gas stoichiometry.
Quick Revision Points
- Principle: $V \propto n$ (at constant T and P).
- Molecule vs Atom: Avogadro was the first to suggest that gases like Hydrogen and Oxygen exist as diatomic molecules ($H_2, O_2$).
- Molar Volume: At STP, 1 mole of any gas = 22.4 L (or 22.7 L under modern IUPAC STP).
- Universal Nature: The law is independent of the chemical identity or 'heaviness' of the gas.
NEET Exam Angle
- Atomicity Problems: Use this law to deduce if a gas is monoatomic, diatomic, or triatomic based on volume data.
- STP Conversions: Memorize 22.4 L for calculations, but be aware of pressure units (1 atm vs 1 bar).
- Number of Particles: If volumes are equal, the ratio of molecules is 1:1, regardless of the molar masses.
| Gas Type | Volume at STP | Moles | Number of Molecules |
|---|---|---|---|
| Hydrogen ($H_2$) | 22.4 L | 1.0 | $6.022 \times 10^{23}$ |
| Oxygen ($O_2$) | 22.4 L | 1.0 | $6.022 \times 10^{23}$ |
| Methane ($CH_4$) | 22.4 L | 1.0 | $6.022 \times 10^{23}$ |
06Dalton’s Atomic Theory: The Indivisible Building Blocks

“Dalton gave us the foundation: atoms are the ultimate, indivisible building blocks of matter. All atoms of a specific element are identical in mass and properties. When elements combine, they do so in simple whole-number ratios to form compounds. It’s the Lego set of the universe!”
John Dalton’s Atomic Theory, published in 1808, was the first successful attempt to explain the laws of chemical combination using a theoretical model. He proposed that matter consists of indivisible atoms, and that all atoms of a given element are identical in mass and properties. When these atoms combine in fixed, whole-number ratios, they form compounds. This provided a logical 'why' behind Lavoisier’s mass conservation and Proust’s definite proportions.
According to Dalton, a chemical reaction is simply a rearrangement of atoms. Atoms are not created or destroyed in a reaction; they just switch partners. This perfectly explains the Law of Conservation of Mass. Furthermore, because atoms are indivisible units, they must combine in whole numbers (like 1:1 or 2:3), which explains the Law of Definite Proportions and Multiple Proportions. Dalton’s theory acted as the 'Lego instructions' for the universe, showing that complex matter is just a specific arrangement of simple, indestructible blocks.
While Dalton's theory was revolutionary, modern science has refined it. We now know that atoms can be divided into subatomic particles (protons, neutrons, electrons), and that isotopes exist (atoms of the same element with different masses). Despite these modifications, the core of Dalton’s theory remains the starting point for all of chemistry. For a NEET student, understanding where Dalton was right—and where he was slightly off—is a common area for conceptual questions.
Quick Revision Points
- Indivisibility: Dalton viewed atoms as the smallest units of matter (later disproved by subatomic particles).
- Identical Atoms: He believed all atoms of an element are identical (later disproved by isotopes).
- Compound Formation: Compounds are formed when atoms of different elements combine in fixed ratios.
- Rearrangement: Chemical reactions involve the reorganization of atoms, not their creation or destruction.
NEET Exam Angle
- Limitations: Be ready to identify 'Isotopes' as the reason why the 'identical mass' postulate failed.
- Subatomic Particles: The discovery of electrons and protons disproved the 'indivisible' nature of the atom.
- Theory vs Law: Dalton’s theory explains the laws of chemical combination; it is not a law itself.
- Isobars: The existence of isobars (different elements with same mass) also contradicts Dalton's original postulates.
07Synthesizing the Five Pillars for NEET Stoichiometry

“You’ve just mastered the five pillars of chemical combination! These laws are the bedrock of NEET chemistry. Understand them, visualize the ratios, and you’ll find stoichiometry becomes a piece of cake. Keep practicing, stay curious, and let’s ace this exam together. See you in the next lecture!”
The five laws of chemical combination are more than just historical facts; they are the logic gates through which every chemical reaction must pass. As a NEET aspirant, your goal is to move from theoretical understanding to pattern recognition. When you see a problem involving masses, think Lavoisier, Proust, or Dalton. When you see volumes and gases, immediately switch your brain to Gay-Lussac and Avogadro. These laws are the 'rules of the game' for the entire stoichiometry unit.
Most high-yield questions from this topic between 2018 and 2024 require you to identify which law is being demonstrated by a set of experimental data. For instance, if a question says '10g of A reacts with 5g of B to give 15g of C,' it’s a direct application of the Law of Conservation of Mass. If it shows two different compounds of the same elements, it's Multiple Proportions. Mastering these distinctions allows you to solve conceptual MCQs in seconds, leaving more time for the complex numericals in the later chapters of Physical Chemistry.
Remember, chemistry is a cumulative subject. The ratios you learn here will reappear when you calculate empirical formulas, find limiting reagents, and work with the Ideal Gas Law. Keep these five pillars solid in your mind, and the rest of the syllabus will have a firm foundation to stand on. Stay focused on the 'simple whole number' aspect of these laws—it is the universe's way of keeping things orderly and predictable.
Quick Revision Points
- Conservation of Mass: Look for total mass balance in closed systems.
- Definite Proportions: Look for constant percentage composition across different samples.
- Multiple Proportions: Look for fixed mass of one element vs. simple ratio of another.
- Gay-Lussac/Avogadro: Use these for volume-volume stoichiometry in gas reactions.
NEET Exam Angle
- Identify the Law: Common MCQ pattern—match experimental data to the correct law.
- Isotope Pitfalls: Always remember that isotopes are the 'exception' to Dalton’s mass-related postulates.
- State Symbols: In Gay-Lussac problems, ignore the volumes of solids (s) and liquids (l).
- Calculated Ratios: Practice dividing by the smallest value to find the simple whole-number ratio quickly.
| Law | Key Variable | Major Scientist | Typical NEET Question |
|---|---|---|---|
| Conservation of Mass | Mass | Lavoisier | Find missing product mass |
| Multiple Proportions | Ratio | Dalton | Compare two different oxides |
| Gaseous Volumes | Volume | Gay-Lussac | Volume of gas produced |
Recommended Reading
Explore related Biology topics to build deeper chapter connections for NEET.
- Chemical Equations · Topic 1.10
- Dalton's Atomic Theory · Topic 1.2
- Mole Concept · Topic 1.6
- Molar Mass · Topic 1.7
- Nature of Electromagnetic Radiation · Topic 2.1
- Photoelectric Effect · Topic 2.2
- Jump to Key Terms (Quick Revision)
- Review Common NEET Mistakes
- Read Topic FAQs
- Check PYQ Pattern Notes
- Practice NEET MCQs
- Solve NEET PYQs
📚 Key Terms
⚠️ Common NEET Mistakes
- 1Applying Gay-Lussac's Law to liquids or solids instead of only gases.
- 2Confusing 'Definite Proportions' (one compound, constant ratio) with 'Multiple Proportions' (different compounds, simple ratio).
- 3Forgetting that the Law of Conservation of Mass only applies to the total mass of a *closed* system where gas cannot escape.
- 4Assuming Dalton's theory is still 100% accurate; remember isotopes and subatomic particles as modern exceptions.
- 5Calculating volume ratios without checking if temperature and pressure are constant.
- 6Using mass ratios in Gay-Lussac's Law instead of volume ratios.
📝 NEET PYQ Pattern
Analysis of NEET papers from 2018–2024 shows a focus on numerical problems where students must identify the law from experimental data. Questions frequently test the Law of Multiple Proportions using oxides of Nitrogen or Carbon. There is also a recurring theme of theoretical questions regarding the failures of Dalton's Atomic Theory (isotopes) and volume-volume stoichiometry applications of Gay Lussac's Law.
❓ Frequently Asked Questions
Does the Law of Conservation of Mass hold true for nuclear reactions?
No, it does not. In nuclear reactions, mass is converted into energy according to Einstein's mass-energy equivalence equation ($E=mc^2$). Therefore, mass and energy are conserved together as a single entity, rather than mass alone.
How does the Law of Definite Proportions differ from the Law of Multiple Proportions?
The Law of Definite Proportions deals with a single compound (e.g., $H_2O$ always has an $H:O$ mass ratio of 1:8). The Law of Multiple Proportions deals with two or more *different* compounds formed by the same elements (e.g., in $CO$ and $CO_2$, the masses of oxygen that react with 12g of carbon are in a 1:2 ratio).
Why are isotopes considered an exception to Dalton's Atomic Theory?
Dalton's theory stated that all atoms of a specific element are identical in mass. Isotopes are atoms of the same element (same atomic number) but with different numbers of neutrons, resulting in different atomic masses, which contradicts Dalton's postulate.
Under what specific conditions does Gay Lussac’s Law of Gaseous Volumes apply?
It applies only when all reactants and products are in the gaseous state and the temperature and pressure are kept constant throughout the measurement of volumes.
Can you explain the Law of Multiple Proportions with an example of Nitrogen oxides?
In Nitrogen oxides like $N_2O$ and $N_2O_3$, the masses of oxygen that combine with a fixed mass of nitrogen (e.g., 28g) are 16g and 48g respectively. These masses of oxygen (16:48) are in a simple whole-number ratio of 1:3.
How did Avogadro’s Law help in determining the atomicity of gases?
By showing that equal volumes contain equal molecules, Avogadro explained why 1 volume of $H_2$ and 1 volume of $Cl_2$ produce 2 volumes of $HCl$. This was only possible if $H_2$ and $Cl_2$ were diatomic ($H_2$, $Cl_2$), allowing each molecule to split and provide atoms for two $HCl$ molecules.
Written By
NEET Content Strategist & Biology Expert
Sangita Kumari is a NEET educator and content strategist with over 6 years of experience teaching Biology, Chemistry, and Physics to Class 11 and 12 aspirants. She helps bridge the gap between traditional NCERT preparation and modern AI-powered learning. Her content is trusted by thousands of NEET aspirants across India.