🎬 Video Lesson Available
Watch the full 7-slide video lesson for Chemical Equations with AI teacher narration and visual explanations.
01The Recipe of Reactions: Introduction to Chemical Equations
“Welcome, future doctors! Imagine a chemical equation like a kitchen recipe. You start with raw ingredients called reactants and end with a delicious dish called products. In chemistry, we write this balance as a universal code, ensuring mass is never lost in the cooking process!”
Think of a chemical equation as the ultimate recipe for the universe. Just as you wouldn't bake a cake without knowing the ratio of flour to eggs, you cannot master NEET Chemistry without understanding how equations represent the transformation of matter. A chemical equation is a symbolic representation of a chemical change, where starting materials (reactants) are converted into new substances (products). This isn't just notation; it is a quantitative map describing what happens at the molecular level when bonds break and new ones form.
In NEET, viewing equations as recipes demystifies complex reactions. When we write $H_2 + O_2 \rightarrow H_2O$, we are describing a specific event where hydrogen and oxygen collide to form water. However, a raw recipe is rarely enough. To satisfy the Law of [Conservation of Mass](/neet/chemistry/laws-of-chemical-combination-class-11-neet), these recipes must be perfectly balanced. This ensures every atom you start with is accounted for in the final product. No atom is lost to the void, and none are magically created. This concept serves as the bridge to the Mole Concept and Stoichiometry.
Understanding this 'universal code' is vital for speed and accuracy. Chemical notation allows students to understand processes without ambiguity. If you can visualize the 'ingredients' being rearranged into the 'dish,' you are halfway to solving difficult problems. This mental picture is the foundation for all quantitative analysis in Chemistry.
Quick Revision Points
- Reactants: Substances existing before the reaction, written on the left.
- Products: New substances formed, written on the right.
- Yield: The amount of product produced; determined by starting reactants.
- Conservation: Total mass of reactants equals total mass of products in a closed system.
- Notation: Plus signs (+) separate multiple species, and the arrow ($\rightarrow$) shows the direction of change.
NEET Exam Angle
- Identifying the physical state and reaction type is the first step in numericals.
- Questions test the link between macroscopic mass and microscopic atom counts.
- Clarity on 'yield' helps in problems involving percentage purity and theoretical output.
| Component | Role in the Equation | Practical Analogy |
|---|---|---|
| Reactants | Starting materials | Ingredients (Flour, Sugar) |
| Products | Ending materials | Finished Dish (Cake) |
| Arrow ($\rightarrow$) | Transformation | Baking in the oven |
| Coefficients | Relative quantities | Number of cups/spoons |
02Chemical Shorthand: The Universal Language of Chemistry Grammar
“A chemical equation is simply a shorthand for a reaction. Reactants on the left, products on the right, linked by an arrow. Think of the arrow as a 'becomes' sign. It is the fundamental grammar of our chemical language, vital for solving your NEET problems quickly!”
If chemistry is a language, then chemical equations are its sentences. To communicate effectively, scientists use a shorthand notation that condenses complex molecular interactions into a single, readable line. The central feature of this grammar is the chemical arrow. Unlike an equals sign in mathematics, which suggests a static relationship, the arrow in chemistry signifies a dynamic transformation. It points from the past (reactants) to the future (products), effectively saying 'becomes' or 'yields.' Mastery of this notation is essential for the NEET exam, where every second saved in interpreting an equation can be used for complex calculations.
A standard chemical equation also includes information about the physical environment of the reaction. We use specific abbreviations in parentheses: (s) for solids, (l) for liquids, (g) for gases, and (aq) for aqueous solutions—substances dissolved in water. These descriptors are not just fluff; they are critical for understanding enthalpy changes and gas laws in later units like Thermodynamics and States of Matter. For instance, the reaction of solid calcium carbonate with aqueous hydrochloric acid looks very different on paper once you add these state symbols, providing a clearer picture of the reaction's behavior, such as the evolution of carbon dioxide gas.
Using this shorthand effectively accelerates your problem-solving speed. Instead of writing 'two molecules of hydrogen gas react with one molecule of oxygen gas to produce two molecules of liquid water,' we simply write $2H_2(g) + O_2(g) \rightarrow 2H_2O(l)$. This efficiency is what allows competitive exam candidates to scan a question and immediately identify the stoichiometric relationships required. It bridges the gap between Unit 1 and more advanced topics like Chemical Bonding, where the representation of molecular structures becomes even more nuanced.
Quick Revision Points
- The Arrow: Represents the direction of the reaction; can be single (irreversible) or double (reversible/equilibrium).
- State Symbols: (s), (l), (g), and (aq) denote the physical state of each species.
- Standard Format: Reactants appear on the left; products appear on the right.
- Conditions: Temperature, pressure, or catalysts are often written above or below the arrow.
NEET Exam Angle
- Pay close attention to the (aq) symbol in ionic equations; it dictates how substances dissociate in solution.
- Questions may ask you to identify the correct balanced equation including state symbols for a given descriptive sentence.
- Understanding shorthand is crucial for interpreting Unit 4 molecular representations and Unit 8 redox reactions.
| Symbol | Meaning | Example Context |
|---|---|---|
| (s) | Solid | Precipitates in a reaction |
| (l) | Liquid | Pure water or molten salts |
| (g) | Gas | Effervescence or gas evolution |
| (aq) | Aqueous | Salts dissolved in water |
| $\Delta$ | Heat | Thermal decomposition reactions |
03The Golden Rule: Law of Conservation of Mass in Equations

“The Law of Conservation of Mass is our golden rule: matter is neither created nor destroyed! Atoms must be identical on both sides. If you start with two hydrogen atoms, you must end with two. Always check your atom count—it’s the secret to balancing any equation.”
The Law of Conservation of Mass is the bedrock of chemistry. Formulated through Dalton’s Atomic Theory, this law states that atoms are neither created nor destroyed during a chemical reaction—they are simply rearranged. Imagine Lego bricks: you can build a car, dismantle it, and build a house, but the total number of bricks remains constant. In a closed system, the mass of the reactants will exactly match the mass of the products. This is why we balance equations: to reflect physical reality.
For NEET aspirants, this law is a practical tool. Every time you face a stoichiometry problem, your first step should be verifying the atom count. If you start with four hydrogen atoms in the reactants, you must have four in the products. If numbers don't match, subsequent calculations for moles, mass, or volume will be incorrect. This 'Golden Rule' provides a fail-safe check before moving into complex numerical analysis.
Understanding mass balance is crucial when dealing with combustion or gas-evolution reactions where products might seem to 'disappear.' In reality, the mass escapes as gas. If the reaction occurred in a sealed container, the total mass would remain constant. This prevents the common mistake of assuming mass is lost when a gas is produced. Atoms are the currency of reactions, and the Law of Conservation of Mass ensures the 'balance sheet' always zeros out.
Quick Revision Points
- Dalton’s Influence: Atoms are indivisible and indestructible in chemical processes.
- Equality Requirement: Atoms of each element must be identical on both sides.
- Total Mass: Mass of Reactants = Mass of Products.
- Closed System: Conservation is most visible when no matter enters or leaves the system.
NEET Exam Angle
- A common trick in NEET is providing unbalanced equations to see if students balance them before calculation.
- This law justifies the balancing steps taught in Unit 1 and Unit 8 (Redox).
- Conceptual questions may ask about the law's validity in chemical vs. nuclear reactions.
| Element | Atoms in Reactants | Atoms in Products | Balanced? |
|---|---|---|---|
| Hydrogen | 4 | 4 | Yes |
| Oxygen | 2 | 2 | Yes |
| Carbon | 1 | 1 | Yes |
| Nitrogen | 2 | 2 | Yes |
04The Balancing Act: Coefficients, Subscripts, and Equation Logic
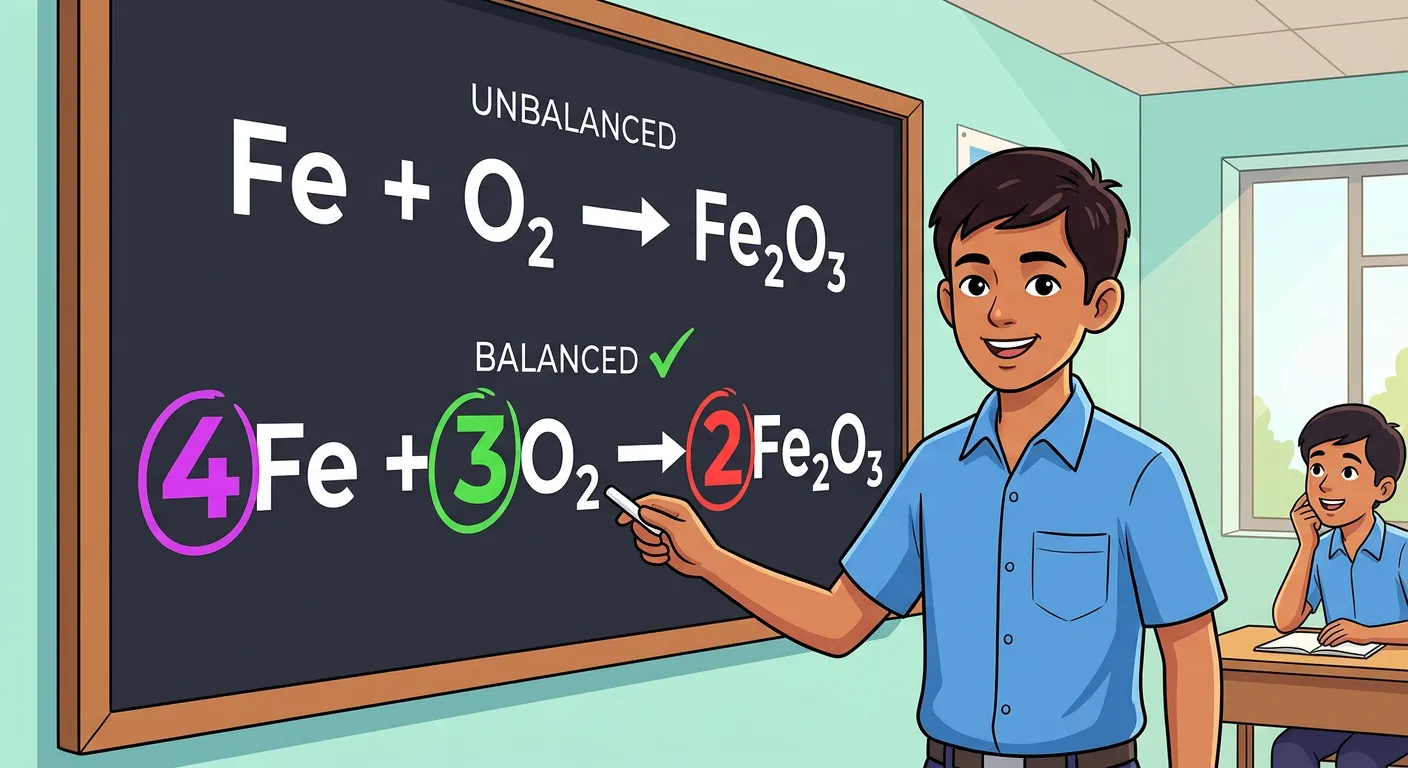
“Balancing equations is like solving a puzzle. We use coefficients—the big numbers placed in front of formulas—to match atom counts. Never change the subscripts! Keep the formula identity intact; just adjust the quantities to ensure the Law of Conservation of Mass is respected.”
Balancing a chemical equation requires specific rules to ensure you don't 'cheat' the chemistry. The most important rule is the distinction between coefficients and subscripts. A subscript (the small number, like the '2' in $H_2O$) defines the substance's identity. Changing it changes the molecule entirely—turning water ($H_2O$) into hydrogen peroxide ($H_2O_2$). Conversely, a coefficient (the large number in front, like '2' in $2H_2O$) tells you how many units exist. To balance an equation, you only adjust coefficients.
When tackling reactions, use the inspection method. Start by balancing elements that appear in only one reactant and one product. Leave hydrogen and oxygen for last, as they often appear in multiple places and balance themselves once heavier atoms are set. For example, in the combustion of propane ($C_3H_8$), balance carbons first, then hydrogens, and finally oxygens. This systematic approach prevents the frustrating loop of changing one number only to disrupt another.
Visualizing this at the molecular level is key. If the equation says $2H_2 + O_2 \rightarrow 2H_2O$, imagine two pairs of hydrogen and one pair of oxygen atoms colliding to form two water molecules. If you change subscripts, you are inventing molecules that don't exist in that reaction. Stick to coefficients; they are your only legal tool for achieving the mass balance required by law.
Quick Revision Points
- Coefficients: Numbers in front; they multiply the entire formula.
- Subscripts: Small numbers within the formula; they define the specific substance.
- Balancing Goal: Ensure atom counts for each element are identical on both sides.
- Priority Rule: Balance atoms appearing in multiple species last (usually H and O).
NEET Exam Angle
- NEET often presents skeletal equations that require balancing before mole calculations begin.
- Changing subscripts is a common distractor in MCQ options—never select them!
- This logic is a prerequisite for advanced methods like the 'Oxidation Number Method' in Unit 8.
| Feature | Coefficient | Subscript |
|---|---|---|
| Location | In front of the formula | Within the formula |
| Indicates | Number of moles/units | Number of atoms in a unit |
| Can change? | Yes | NO! |
| Multiplies | The whole compound | Only the specific element |
05Stoichiometry: The Quantitative Backbone of Chemical Math

“Stoichiometry is just the math of chemistry! It tells us exactly how much reactant is needed to yield a specific amount of product. Think of it as calculating the exact amount of batter needed for twenty pancakes. It’s the backbone of all quantitative NEET questions.”
Once an equation is balanced, it becomes a powerful predictive tool through the science of stoichiometry. Derived from the Greek words 'stoicheion' (element) and 'metron' (measure), stoichiometry is the study of the quantitative relationships between reactants and products. It allows us to calculate exactly how much of a reactant is needed to produce a desired amount of product. Think of it as the 'math of the recipe.' If a recipe says 2 cups of flour make 12 cookies, stoichiometry tells us that 4 cups will make 24.
The secret weapon in stoichiometry is the 'Mole Ratio.' The coefficients in a balanced equation represent the ratio of moles of each substance involved. For the reaction $N_2 + 3H_2 \rightarrow 2NH_3$, the mole ratio of $N_2$ to $H_2$ is $1:3$. This ratio acts as a conversion factor. If you have 5 moles of nitrogen, you immediately know you need 15 moles of hydrogen. This logic extends to mass-mass, mole-mole, and even mass-volume relationships, making it the backbone of almost all numerical problems you will face in NEET Chemistry.
In practical terms, stoichiometry helps us determine the theoretical yield—the maximum amount of product that could be formed from a given amount of reactant. Just like calculating exactly how much pancake batter you need for a specific number of guests, stoichiometric calculations ensure efficiency in the lab and accuracy in your exam answers. Whether you are dealing with grams, liters, or molecules, the mole remains your central hub for all conversions.
Quick Revision Points
- Stoichiometry: Measures the quantitative relationship between substances in a reaction.
- Mole Ratio: Derived from coefficients; used to convert between different substances.
- Mass-Mass Calculations: Converting mass of A $\rightarrow$ moles of A $\rightarrow$ moles of B $\rightarrow$ mass of B.
- Theoretical Yield: The calculated maximum product based on stoichiometry.
NEET Exam Angle
- Most NEET stoichiometry questions are 'multi-step,' requiring you to convert mass to moles before using the mole ratio.
- Familiarity with the 'Mole Map' (mass-moles-volume) is essential for solving these quickly under exam pressure.
- Stoichiometry is often integrated with the Ideal Gas Law ($PV=nRT$) for reactions involving gases at non-STP conditions.
| From | To | Operation |
|---|---|---|
| Mass (g) | Moles | Divide by Molar Mass |
| Moles | Mass (g) | Multiply by Molar Mass |
| Moles A | Moles B | Use Mole Ratio from balanced equation |
| Moles (gas) | Volume (STP) | Multiply by 22.4 L/mol |
06The Limiting Reagent: Navigating Reaction Bottlenecks
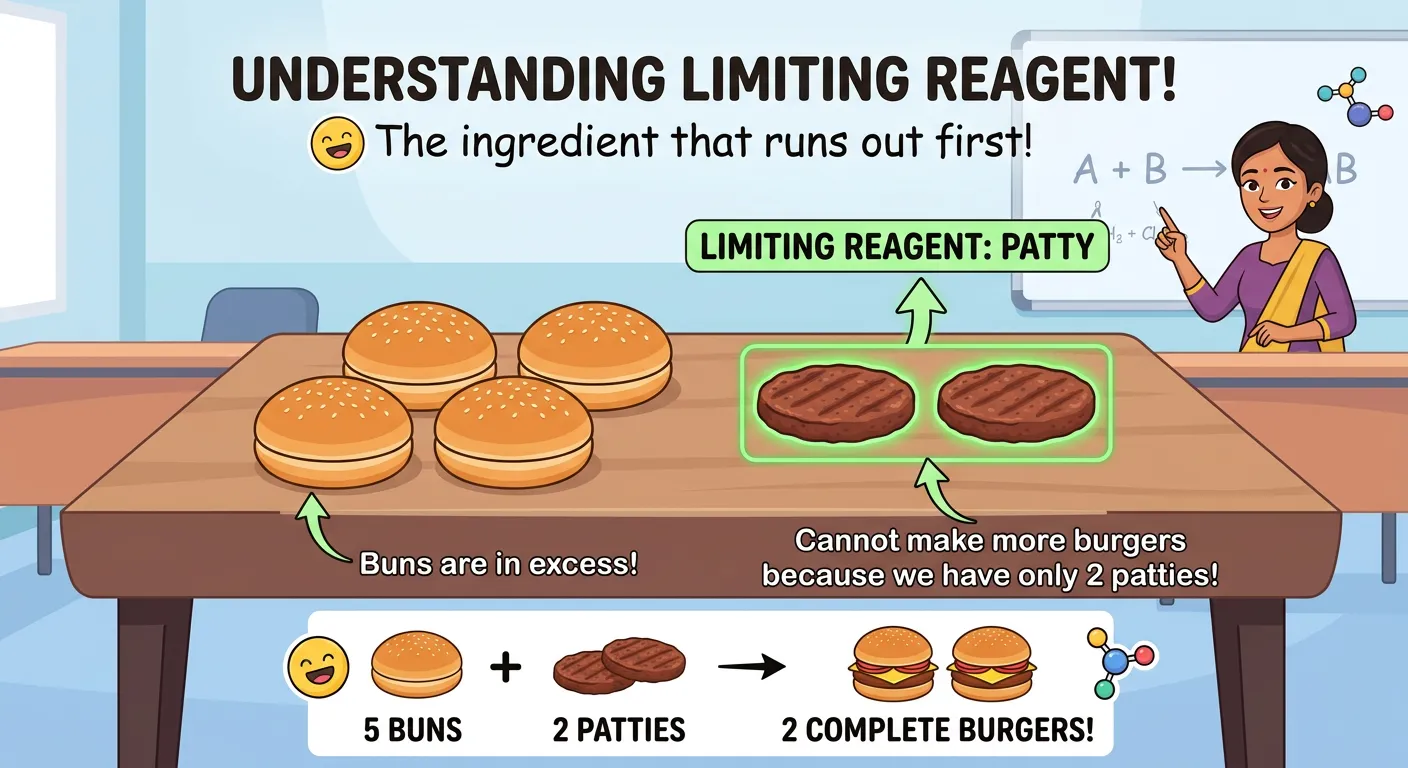
“The limiting reagent is the 'ingredient' that runs out first, stopping the reaction. Like having five buns but only two patties—you can only make two burgers! Identify the limiting reagent first, and the rest of your stoichiometric calculation becomes a piece of cake.”
In the real world, and in many NEET problems, reactants are rarely provided in the exact stoichiometric proportions. Usually, one reactant will run out before the others. This substance is called the Limiting Reagent (LR). It is the 'bottleneck' of the reaction because it limits the amount of product that can form. The reactants left over after the LR is consumed are called Excess Reagents. Understanding this concept is crucial for solving high-yield NEET numericals where multiple reactant quantities are given.
Consider the 'Burger Analogy.' If you have 10 burger patties but only 8 buns, you can only make 8 complete burgers. Even though you have 'excess' patties, the buns 'limit' your production. In chemistry, if you have 2 moles of $H_2$ and 2 moles of $O_2$ to form water ($2H_2 + O_2 \rightarrow 2H_2O$), the hydrogen will run out first. Even though the molar amounts are equal, the equation requires two parts hydrogen for every one part oxygen. Thus, hydrogen is the limiting reagent, and the amount of water produced is dictated solely by the amount of hydrogen available.
To identify the LR mathematically, calculate the number of moles of each reactant and divide by its respective stoichiometric coefficient. The substance with the smallest resulting value is your limiting reagent. Once the LR is identified, all subsequent calculations for product yield or amount of excess reactant used must be based on the LR. Never use the excess reagent for product yield calculations; it will lead to an overestimation and a wrong answer on your OMR sheet.
Quick Revision Points
- Limiting Reagent (LR): The reactant completely consumed in a reaction; it determines the maximum product yield.
- Excess Reagent: The reactant that remains after the reaction stops.
- Identification Rule: Smallest (Moles / Coefficient) ratio = Limiting Reagent.
- Significance: The LR dictates the theoretical yield of the reaction.
NEET Exam Angle
- Questions often give masses of two reactants and ask for the mass of the product. Identifying the LR is always the mandatory first step.
- Problems may also ask 'how much excess reagent remains?' which requires subtracting the amount consumed from the initial amount.
- Limiting reagent concepts are frequently paired with organic chemistry multi-step syntheses in Class 12 topics.
| Characteristic | Limiting Reagent | Excess Reagent |
|---|---|---|
| Consumption | Completely used up | Partially used up |
| Determines Yield? | Yes | No |
| Remaining after Rxn | Zero | Initial amount - Consumed amount |
| Moles/Coeff Ratio | Lower | Higher |
07NEET Mastery Strategy: Implementing Stoichiometric Success
“Great work! You’ve mastered the language of chemical equations. Remember: balance the atoms, follow the mole ratios, and identify the limiting reagent. Practice these steps, and you’ll crush the stoichiometry section in your NEET exam. Keep learning, keep growing, and see you next time!”
Mastering stoichiometry for NEET requires a fast, reliable workflow. To solve quantitative problems, follow this protocol: Balance, Convert, Ratio. First, always ensure the equation is balanced. Second, convert given quantities (mass, STP volume, or particles) into moles. Third, apply the mole ratio from the balanced equation to find moles of the unknown, then convert to required units. This systematic approach minimizes silly mistakes and saves time during the exam.
Common 'traps' include gas volumes and reactant purity. At STP, 1 mole of an ideal gas occupies 22.4 Liters. If a question mentions '11.2 L of $O_2$ at STP,' read it as '0.5 moles.' However, be wary: if conditions are not STP, use the Ideal Gas Equation. Another factor is 'percentage purity.' If you have 100g of 80% pure $CaCO_3$, only 80g reacts. Always adjust for purity before starting your stoichiometric ratios to avoid inflated results.
Finally, always perform a 'sanity check.' Does the product mass seem reasonable? Did you account for the limiting reagent? Stoichiometry is the quantitative grammar of chemistry. By mastering these steps, you gain the ability to predict reaction outcomes with mathematical precision. Practice these protocols until they become second nature, turning daunting puzzles into manageable, logical paths to success. Consistency in unit conversion and careful identification of the LR will ensure you excel in this high-scoring section.
Quick Revision Points
- Workflow: 1. Balance Equation, 2. Convert to Moles, 3. Apply Mole Ratio.
- STP Shortcuts: 1 mol = 22.4 L for gases at Standard Temp and Pressure.
- Purity Factor: Calculate the 'pure' mass before starting ratios.
- Consistency: Maintain unit consistency to avoid decimal errors.
NEET Exam Angle
- Integrated questions combine stoichiometry with concentration terms like Molarity.
- Recent papers (2022-2024) favor 'Assertion-Reason' questions on Limiting Reagents.
- Mastery here is the foundation for Class 12 Electrochemistry and Kinetics.
| Calculation Type | Critical Step | Common Trap |
|---|---|---|
| Mass-Mass | Molar Mass conversion | Using wrong atomic weights |
| Mass-Volume | 22.4 L at STP | Forgetting to check STP conditions |
| Limiting Reagent | Moles/Coefficient | Comparing moles without dividing |
| Percent Yield | (Actual/Theoretical)x100 | Using Actual yield in ratios |
Recommended Reading
Explore related Biology topics to build deeper chapter connections for NEET.
- Dalton's Atomic Theory · Topic 1.2
- Laws of Chemical Combination · Topic 1.4
- Mole Concept · Topic 1.6
- Molar Mass · Topic 1.7
- Nature of Electromagnetic Radiation · Topic 2.1
- Photoelectric Effect · Topic 2.2
- Jump to Key Terms (Quick Revision)
- Review Common NEET Mistakes
- Read Topic FAQs
- Check PYQ Pattern Notes
- Practice NEET MCQs
- Solve NEET PYQs
📚 Key Terms
⚠️ Common NEET Mistakes
- 1Changing chemical subscripts instead of coefficients when balancing an equation.
- 2Forgetting to balance the equation before performing stoichiometric calculations.
- 3Using the mass or moles of an excess reagent to calculate the product yield instead of the limiting reagent.
- 4Assuming all gas volume problems are at STP without verifying the temperature and pressure conditions.
- 5Not converting mass to moles before applying the mole ratio (trying to use mass ratios directly).
- 6Neglecting to account for the percentage purity of a reactant in numerical problems.
📝 NEET PYQ Pattern
Stoichiometry and Limiting Reagent questions appear consistently in NEET, often requiring 2-3 step calculations. Recent papers (2021-2024) frequently combine these concepts with the Mole Concept and gas laws (STP conditions) to test integrated quantitative understanding.
❓ Frequently Asked Questions
Why can't we change the subscripts while balancing a chemical equation?
Subscripts define the chemical identity and molecular structure of a substance. Changing a subscript changes the substance itself (e.g., $H_2O$ is water, but $H_2O_2$ is hydrogen peroxide). Balancing only involves changing coefficients, which represent the quantity of the substance, not its nature.
What is the first step in solving a stoichiometry problem involving multiple reactants?
The first step is always to identify the limiting reagent. You do this by converting all reactant quantities to moles and then dividing each by its stoichiometric coefficient from the balanced equation. The reactant with the smallest value is the limiting reagent.
How do you identify the limiting reagent when given the masses of all reactants?
1. Convert masses to moles for each reactant. 2. Divide the moles of each reactant by its coefficient in the balanced equation. 3. The reactant with the lowest ratio is the limiting reagent.
Does the Law of Conservation of Mass apply to nuclear reactions mentioned in NEET?
In strict chemical reactions, yes. However, in nuclear reactions, mass can be converted into energy ($E=mc^2$). For NEET Chemistry Unit 1, we assume the Law of Conservation of Mass holds true because the mass-energy conversion is negligible in ordinary chemical changes.
What is the difference between a stoichiometric coefficient and a mole ratio?
A stoichiometric coefficient is the number in front of a formula in a balanced equation (e.g., the '2' in $2H_2$). A mole ratio is the ratio of coefficients between two different substances in that equation (e.g., $2:1$ for $H_2:O_2$ in water formation), used as a conversion factor.
How does the limiting reagent affect the calculation of the percentage yield?
The limiting reagent determines the 'theoretical yield' (the maximum possible product). The percentage yield is then calculated by dividing the actual (experimental) yield by this theoretical yield and multiplying by 100.
Written By
NEET Content Strategist & Biology Expert
Sangita Kumari is a NEET educator and content strategist with over 6 years of experience teaching Biology, Chemistry, and Physics to Class 11 and 12 aspirants. She helps bridge the gap between traditional NCERT preparation and modern AI-powered learning. Her content is trusted by thousands of NEET aspirants across India.