🎬 Video Lesson Available
Watch the full 7-slide video lesson for Molar Mass with AI teacher narration and visual explanations.
01The Mole Concept: Transitioning from Atomic Scales to the Chemist's Dozen
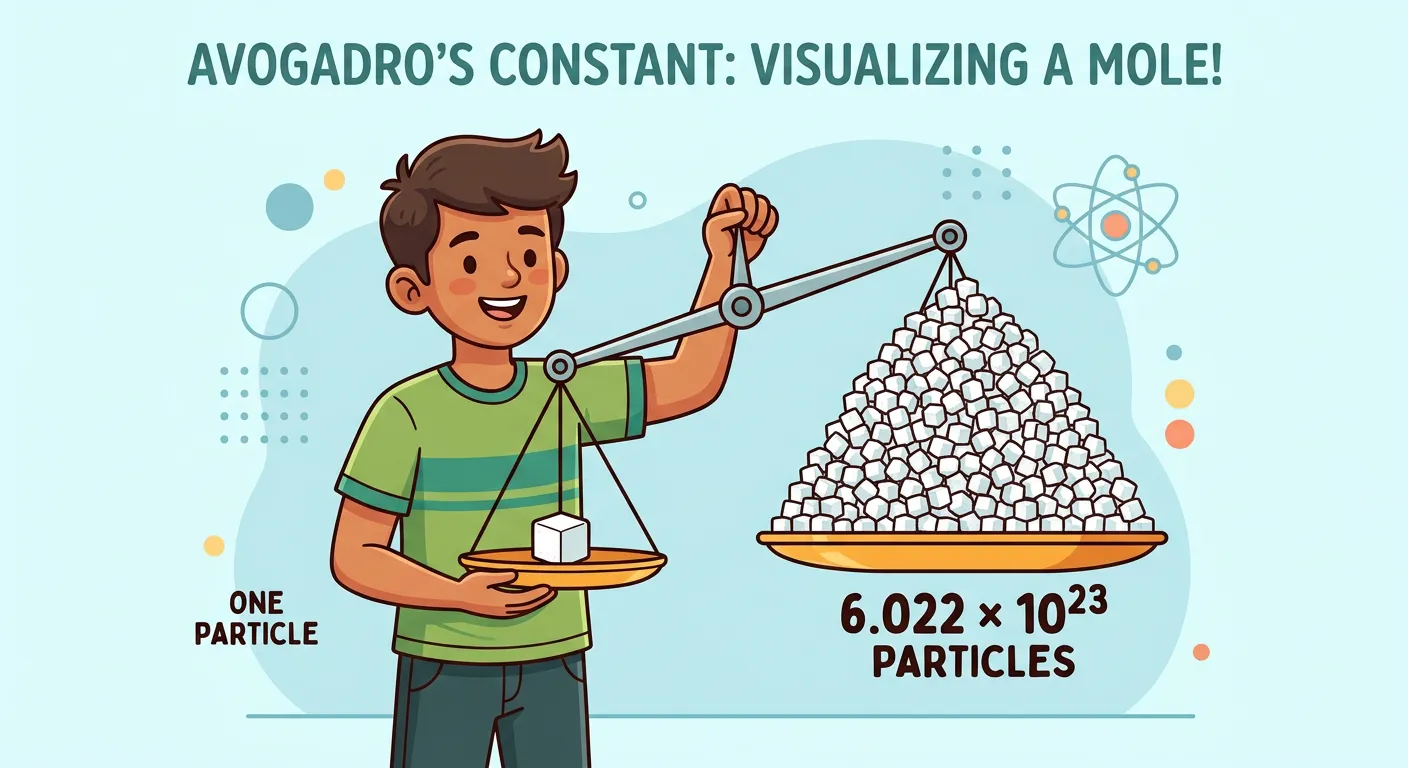
“Welcome, NEET warriors! Imagine you are at a sweet shop. Buying one sugar cube is easy, but how do we weigh an impossible number of atoms? Meet the Mole—the chemist's dozen! Just like a dozen is 12, a mole is simply a giant count of particles.”
In the world of chemistry, we often find ourselves caught between two scales: the microscopic world of atoms and the macroscopic world of the laboratory. Imagine trying to count every individual grain of sand on a beach—it is practically impossible. Similarly, atoms are so incredibly small that counting them one by one is out of the question. A single drop of water contains more atoms than there are stars in the observable universe. To bridge this gap, chemists use a fundamental unit called the 'mole.' Think of the mole as a specialized counting tool, much like how a baker uses a 'dozen' to represent twelve eggs or a 'gross' to represent 144 items.
The mole is defined as the amount of substance that contains as many elementary entities (atoms, molecules, or ions) as there are atoms in exactly 12 grams of the Carbon-12 isotope. This specific number is known as Avogadro's Number, approximately $6.022 \times 10^{23}$. For a NEET aspirant, mastering the mole is the first step toward understanding how we can weigh something on a balance and translate that mass into a specific number of reacting particles. This transition from counting to weighing is the backbone of stoichiometry, which governs every chemical reaction you will study in Class 11 and 12.
Why do we need such a massive number? Because chemical reactions happen between particles, not just masses. If you want to react Hydrogen with Oxygen to form water, you need exactly two atoms of Hydrogen for every one atom of Oxygen. Since we cannot pick up individual atoms with tweezers, we use the mole to ensure we have the correct ratio of particles by weighing them in grams. This concept transforms the invisible atomic world into something tangible and measurable in the lab.
Quick Revision Points
- The Mole is the SI unit for the amount of substance (symbol: mol).
- One mole always contains $6.022 \times 10^{23}$ particles, regardless of the substance.
- Avogadro's number ($N_A$) is the proportionality factor that relates the number of constituent particles in a sample with the amount of substance.
- The 'dozen' analogy helps visualize the mole as a quantity of 'count' rather than a measure of weight.
- Understanding the mole is essential for solving stoichiometry and concentration problems.
NEET Exam Angle
- Standalone questions on the definition of a mole are rare, but the concept is a prerequisite for 80% of physical chemistry.
- Always remember that the mole refers to the number of particles, not the mass itself.
- NEET often tests the relationship between the number of particles and STP (Standard Temperature and Pressure) conditions in gas-related problems.
02The Carbon-12 Standard: Bridging Atomic Mass Units and Grams

“Why exactly that number? Because it's the bridge between the tiny atomic world and our grams! One mole of Carbon-12 atoms weighs exactly 12 grams. This special mass for one mole of any substance is what we call its Molar Mass.”
To understand molar mass, we must first look at the international standard that anchors all chemical measurements: Carbon-12. Before the modern era, chemists struggled with different reference points for atomic mass. Eventually, the scientific community agreed upon Carbon-12 ($^{12}C$) as the universal standard. This isotope was chosen because of its stability and abundance. By definition, one atom of Carbon-12 is assigned a mass of exactly 12 atomic mass units (u). This creates a direct, elegant link between the microscopic and macroscopic worlds.
When we talk about 'Molar Mass,' we are specifically referring to the mass of one mole of a substance. For Carbon-12, the molar mass is exactly 12 grams per mole. This is not a coincidence; the mole was defined specifically so that the numerical value of the atomic mass in 'u' would be identical to the molar mass in 'g/mol'. This clever calibration allows chemists to read the mass of an atom from the periodic table and immediately know how many grams constitute one mole of those atoms.
Molar mass is defined as the mass of one mole of a substance in grams. It is expressed in units of g/mol (grams per mole). For example, if the atomic mass of Nitrogen is 14.007 u, its molar mass is 14.007 g/mol. This means if you weigh out 14.007 grams of Nitrogen, you are holding exactly $6.022 \times 10^{23}$ Nitrogen atoms in your hand. For NEET students, this realization is 'the bridge'—it allows you to convert the theoretical masses found in the Periodic Table into practical, weighable quantities for laboratory experiments and numerical problems.
Quick Revision Points
- Carbon-12 is the reference standard for both atomic mass and the mole.
- Molar mass is the mass of one mole ($N_A$ particles) of a substance.
- The SI unit for molar mass is kg/mol, but in chemistry, g/mol is almost universally used.
- The definition ensures that the mass of 1 mole of $^{12}C$ is exactly 12 grams.
- This standard allows for a seamless transition between 'u' (microscopic) and 'g' (macroscopic).
NEET Exam Angle
- Understand that the 'u' (unified mass) and 'g/mol' are numerically equal but represent different scales.
- Questions may ask why Carbon-12 is used (stability, exactly 12 units by definition).
- Remember that molar mass is an intensive property—it does not change based on the amount of substance you have; it is a characteristic of the substance itself.
03The NEET Shortcut: Converting Atomic Mass (u) to Molar Mass (g/mol)
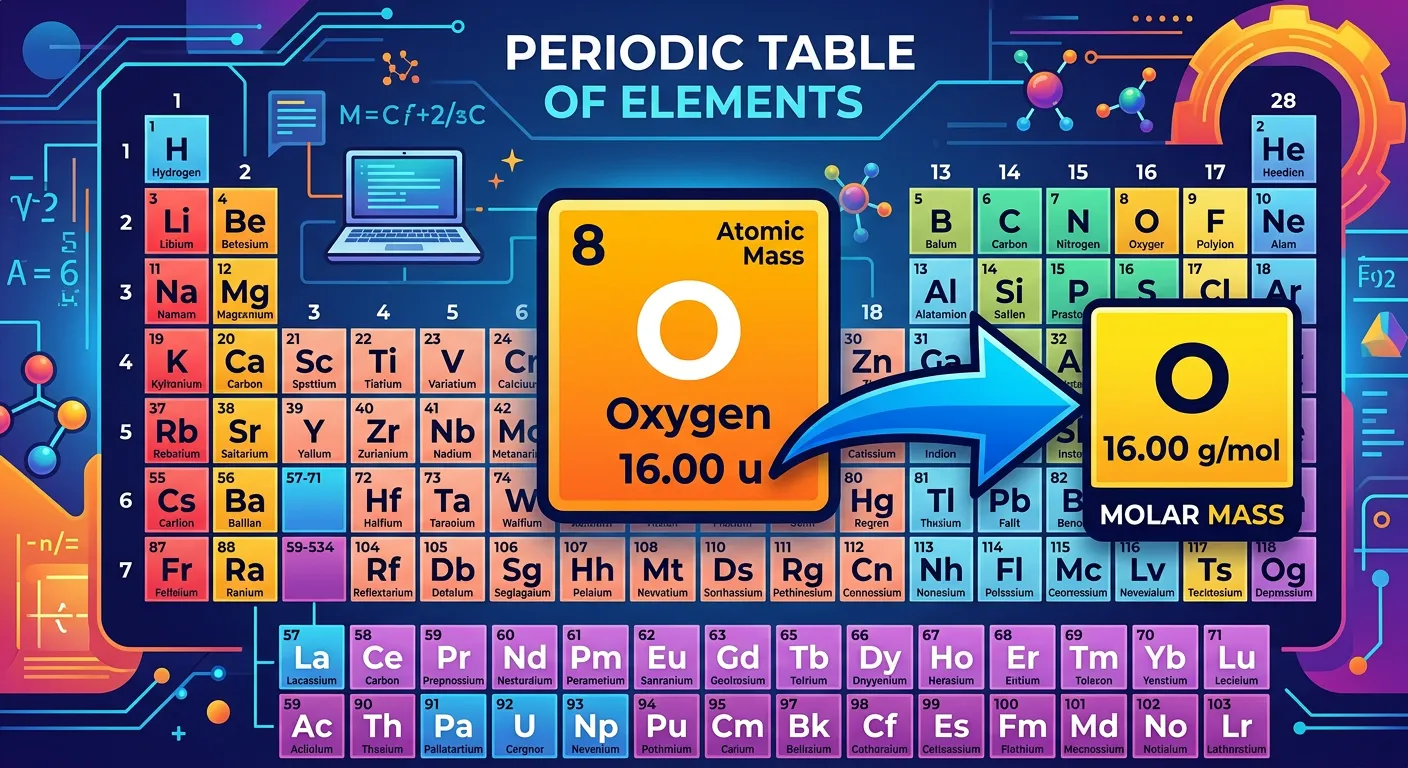
“Here is the NEET shortcut! Look at the Periodic Table. The atomic mass of an element, usually given in atomic mass units or 'u', becomes its Molar Mass just by swapping 'u' for 'grams per mole'. It is that simple—no complex calculation needed!”
During the high-pressure environment of the NEET exam, time is your most precious resource. One of the most effective shortcuts in physical chemistry is the immediate conversion of atomic mass units (u) to molar mass units (g/mol). You do not need to perform complex calculations or use conversion factors. Because of how the mole is defined, the numerical value remains identical. If you see that Sodium (Na) has an atomic mass of 23 u, you can instantly use 23 g/mol for your molar mass calculations.
This shortcut applies across the entire Periodic Table. For elements like Oxygen (16 u), Chlorine (35.5 u), and Calcium (40 u), their molar masses are simply 16 g/mol, 35.5 g/mol, and 40 g/mol respectively. This allows you to extract data from the Periodic Table almost instantly. However, be careful with rounding. While 'u' is a measurement of a single atom's mass, 'g/mol' is the mass of a massive collection of atoms.
To excel in NEET, you should memorize the molar masses of the first 20 elements plus common transition metals like Iron (56), Copper (63.5), and Zinc (65). This mental library will save you minutes during the exam. Physical chemistry problems often hide the molar mass within the text or assume you know it. By internalizing these values, you can focus on the logic of the problem rather than getting bogged down in basic arithmetic.
| Element | Atomic Mass (u) | Molar Mass (g/mol) | Significance for NEET |
|---|---|---|---|
| Hydrogen | 1.008 | 1.008 | Used in almost all organic compounds |
| Carbon | 12.011 | 12.011 | Basis of the mole and organic chemistry |
| Oxygen | 15.999 | 15.999 | Essential for redox and oxide problems |
| Nitrogen | 14.007 | 14.007 | Key in 'Mole Concept' gas problems |
| Sulfur | 32.06 | 32.06 | Frequently appears in $H_2SO_4$ calculations |
Quick Revision Points
- Numerical value of Atomic Mass (u) = Molar Mass (g/mol).
- 'u' refers to the mass of a single atom; 'g/mol' refers to the mass of $6.022 \times 10^{23}$ atoms.
- Swapping units is the fastest way to find molar mass during exams.
- Memorizing common atomic masses is a high-yield strategy for speed.
NEET Exam Angle
- Use rounded values (e.g., O = 16, H = 1) unless the options are extremely close together.
- Look for 'Atomic Weight' in questions; it is synonymous with molar mass in a calculation context.
- Beware of diatomic elements (like $Cl_2$ or $O_2$); their molar mass is double the atomic mass.
04Calculating Molecular Molar Mass: The Summation Method for H2O
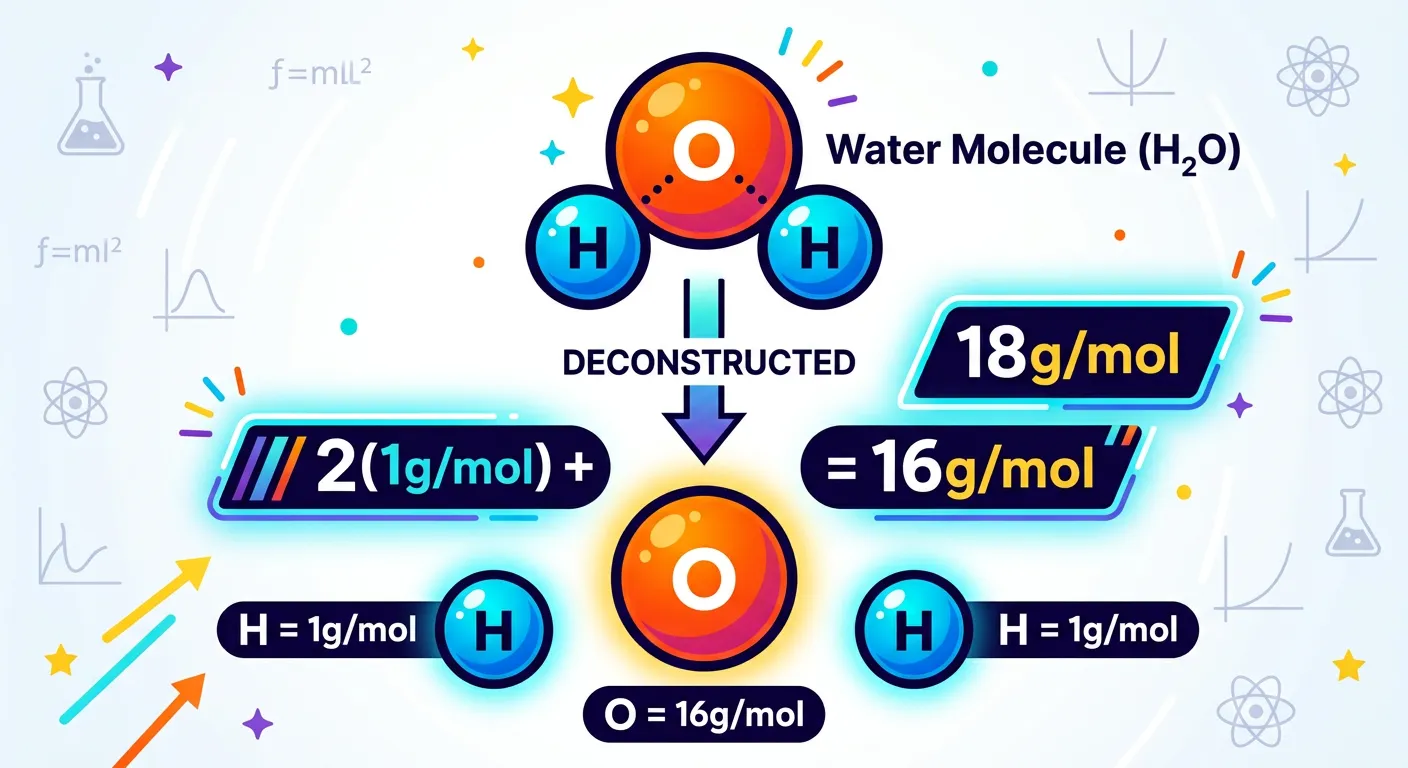
“What about molecules like water? Easy! Just add up the molar masses of every atom present. For H2O, we add two Hydrogens and one Oxygen. 2 plus 16 gives us 18 grams per mole. You’ve just calculated the molar mass of water!”
Moving from individual elements to molecules requires a simple but disciplined approach: the summation method. The molar mass of a molecule (often called molecular molar mass) is simply the sum of the molar masses of all the individual atoms that make up that molecule. Let's take the most common molecule in chemistry: Water ($H_2O$).
A water molecule consists of two Hydrogen atoms and one Oxygen atom. To find its molar mass, we take the molar mass of Hydrogen (approximately 1 g/mol) and multiply it by two, then add the molar mass of Oxygen (approximately 16 g/mol). Calculation: $(2 \times 1.008) + (1 \times 15.999) = 18.015 \text{ g/mol}$. In most NEET problems, we use the rounded value of 18 g/mol. This number is incredibly significant because water is the universal solvent. Whether you are calculating the molarity of an aqueous solution or the mass of water produced in a combustion reaction, '18' will be a recurring figure.
A common mistake students make is forgetting to multiply by the subscript. If a formula says $Al_2(SO_4)_3$, you must multiply everything inside the parentheses by 3. For water, the subscript '2' applies only to Hydrogen. Keeping your calculations organized is key to avoiding 'silly mistakes' that can cost you 4 marks. Always write out the individual components before summing them up to ensure you haven't missed any atoms.
| Molecule | Calculation | Molar Mass (g/mol) | Common Use Case |
|---|---|---|---|
| Water ($H_2O$) | $2(1) + 16$ | 18 | Solvent calculations |
| Ammonia ($NH_3$) | $14 + 3(1)$ | 17 | Haber Process problems |
| Carbon Dioxide ($CO_2$) | $12 + 2(16)$ | 44 | Ideal Gas Law / Combustion |
| Methane ($CH_4$) | $12 + 4(1)$ | 16 | Hydrocarbon stoichiometry |
Quick Revision Points
- Molecular Molar Mass = $\Sigma$ (Atomic Mass $\times$ Number of Atoms).
- Always check subscripts carefully ($H_2O$ means 2 Hydrogens, 1 Oxygen).
- Units remain g/mol.
- For $H_2O$, 18 g/mol is a standard value that should be memorized.
NEET Exam Angle
- $H_2O$ is often a reactant or product; knowing its mass (18) speeds up reaction stoichiometry.
- Watch out for hydrated salts (like $CuSO_4 \cdot 5H_2O$); you must add the mass of the 5 water molecules to the total.
- Practice calculating molar mass for common acids like $HCl$ (36.5) and $H_2SO_4$ (98).
05Complex Molecular Calculations: Determining Molar Mass for Glucose (C6H12O6)
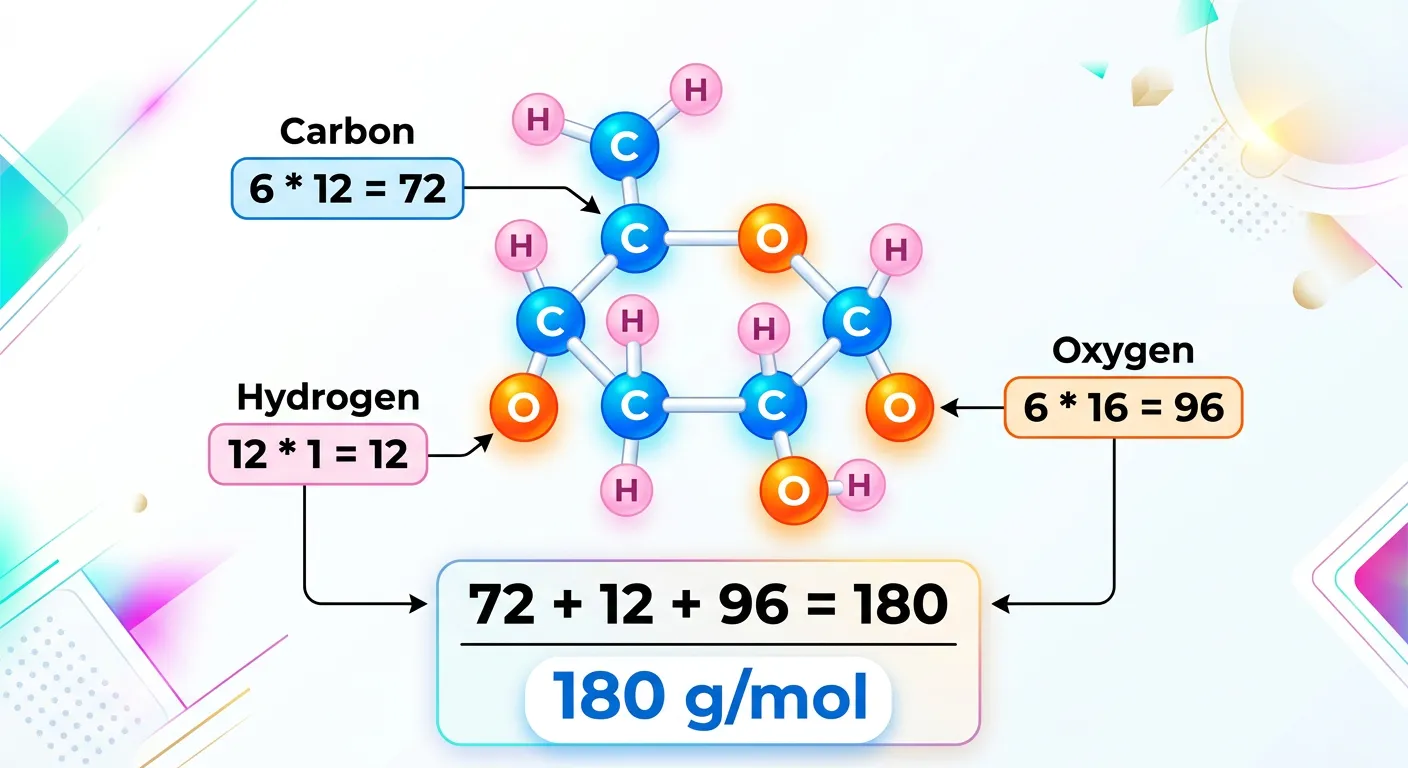
“Let’s level up! Glucose, the energy source for your brain during NEET prep, is C6H12O6. Multiply the number of atoms by their atomic masses and add them up. 6 times 12, 12 times 1, and 6 times 16 equals 180 grams per mole!”
As you progress in your NEET preparation, the molecules will get larger and the formulas more complex. A prime example is Glucose ($C_6H_{12}O_6$), the primary energy source in biological systems. Calculating the molar mass of glucose is a rite of passage for every chemistry student and serves as a bridge between your Chemistry and Biology syllabi. To calculate it, we break it down into its constituent elements: 6 Carbon atoms, 12 Hydrogen atoms, and 6 Oxygen atoms.
Applying our summation rule: Carbon: $6 \times 12.01 = 72.06$ Hydrogen: $12 \times 1.008 = 12.096$ Oxygen: $6 \times 16.00 = 96.00$ Total Molar Mass = $180.156 \text{ g/mol}$ (commonly rounded to 180 g/mol).
Why is 180 such an important number? In NEET Biology, you study cellular respiration where glucose is oxidized. In Chemistry, you might be asked to calculate the molality of a glucose solution. Knowing that 1 mole of glucose equals 180 grams allows you to jump straight into the core of the problem. For large molecules, the algebraic approach is best: $(n_1 \times M_1) + (n_2 \times M_2) + (n_3 \times M_3)$. This systematic method reduces errors when dealing with coefficients and subscripts. Always handle one element at a time, calculate its total contribution, and then sum the final column.
| Compound | Formula | Calculation | Molar Mass (g/mol) |
|---|---|---|---|
| Glucose | $C_6H_{12}O_6$ | $6(12) + 12(1) + 6(16)$ | 180 |
| Sucrose | $C_{12}H_{22}O_{11}$ | $12(12) + 22(1) + 11(16)$ | 342 |
| Urea | $NH_2CONH_2$ | $2(14) + 4(1) + 12 + 16$ | 60 |
| Sulfuric Acid | $H_2SO_4$ | $2(1) + 32 + 4(16)$ | 98 |
Quick Revision Points
- Glucose ($C_6H_{12}O_6$) has a molar mass of 180 g/mol.
- Break down complex formulas element by element to avoid errors.
- Large biological molecules follow the same summation rules as simple ones.
- Sucrose (342) and Glucose (180) are very common in NEET numericals.
NEET Exam Angle
- Multi-element compounds often appear in concentration problems ($Molarity = \text{mass} / (\text{molar mass} \times \text{volume})$).
- Practice calculating the molar mass of Urea (60) and Cane Sugar (342) as they frequently appear in 'Colligative Properties' and 'Solutions' chapters.
- Speed is key; recognizing 180 for Glucose instantly saves about 20-30 seconds of calculation time.
06The Golden Triangle Formula: Mastering Mass-to-Mole Conversions
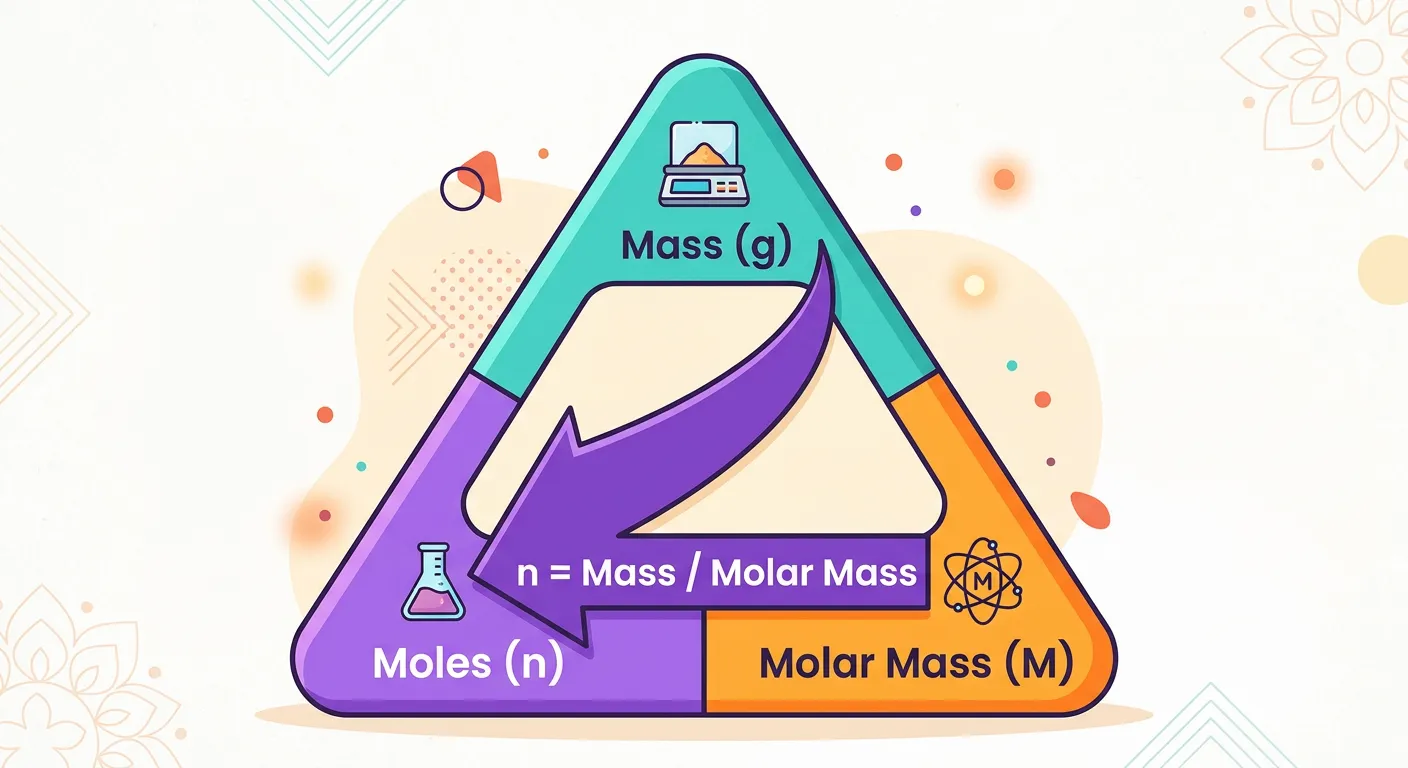
“Need to find how many moles are in a sample? Use this golden triangle! To find moles, simply divide the given mass in grams by the Molar Mass. This relationship is your best friend for solving physical chemistry problems in the exam hall.”
The ultimate goal of learning molar mass is to use it as a conversion factor. In the laboratory and in NEET exam questions, you are rarely given the 'number of moles' directly. Instead, you are given a 'given mass' in grams. To find the amount of substance (n), you must use the most important formula in physical chemistry: $n = m / M$, where '$m$' is the given mass and '$M$' is the molar mass.
Visualizing this as a 'Golden Triangle' helps you rearrange the formula instantly. Place 'mass (m)' at the top of the triangle, and 'moles (n)' and 'Molar Mass (M)' at the bottom.
- To find Moles: $n = m / M$
- To find Mass: $m = n \times M$
- To find Molar Mass: $M = m / n$
This relationship is the 'language' of chemical reactions. For example, if a question asks how many moles are in 36 grams of water, you simply take the given mass (36g) and divide by the molar mass of water (18 g/mol) to get 2 moles. This process is the starting point for almost every problem in the 'Solutions', 'Equilibrium', and 'Thermodynamics' units. Mastering this conversion allows you to move fluently between the quantity you can weigh on a scale and the number of particles participating in a reaction.
| Given Mass (m) | Substance | Molar Mass (M) | Moles (n) | Calculation |
|---|---|---|---|---|
| 10 g | $NaOH$ | 40 g/mol | 0.25 mol | $10 / 40$ |
| 88 g | $CO_2$ | 44 g/mol | 2.0 mol | $88 / 44$ |
| 5 g | $H_2$ | 2 g/mol | 2.5 mol | $5 / 2$ |
| 360 g | $C_6H_{12}O_6$ | 180 g/mol | 2.0 mol | $360 / 180$ |
Quick Revision Points
- The central formula is $n = \text{mass (g)} / \text{molar mass (g/mol)}$.
- Moles are the standard unit for chemical calculations, not grams.
- Use the Golden Triangle to rearrange the formula for mass or molar mass.
- Always ensure your mass is in grams before dividing by molar mass.
NEET Exam Angle
- Many students lose marks by confusing 'mass' with 'moles'. Molar mass is the key that unlocks the conversion.
- Rearranging $m = n \times M$ is frequently required in Gas Law problems ($PV = nRT$).
- Practice rapid divisions (like 1/18 or 1/44) to improve calculation speed.
07Molar Mass as the Currency of Chemistry: Strategy for NEET Success

“You’ve mastered Molar Mass! It is not just a number; it is the currency of chemistry that allows us to count the uncountable. Keep practicing these calculations, stay curious, and you'll definitely crack your NEET goal. See you in the next lesson!”
As we conclude our deep dive into Molar Mass, it is vital to view it not just as a numerical value, but as the 'currency' of chemistry. Just as different countries have different currencies, different substances have different molar masses. To trade or interact, you must know the exchange rate. In a chemical reaction, the balanced equation tells you the ratio in moles, but your lab balance only speaks in grams. Molar mass is the exchange rate that allows these two worlds to communicate.
For NEET success, accuracy in molar mass calculation is non-negotiable. An error of just 1 gram in your molar mass can lead to a completely different answer in a multiple-choice question, often matching one of the 'distractor' options designed to catch students making that specific mistake. Therefore, double-checking your sums for elements like Oxygen and Carbon is a habit that will pay dividends.
Chemistry is often viewed as a bridge between the physical laws of physics and the complex systems of biology. Molar mass sits right at the center of that bridge. Whether you are calculating the dosage of a medicine (Biology/Pharmacology) or the energy released in a fuel cell (Physics/Chemistry), molar mass is the fundamental constant you will rely on. Keep practicing these calculations daily until they become second nature. When you can look at $H_2SO_4$ and immediately think '98', you are well on your way to cracking the NEET physical chemistry section.
Quick Revision Points
- Molar mass is the bridge between mass and number of particles.
- It is an essential prerequisite for Molarity, Molality, and Mole Fraction.
- Accuracy in addition is the most common pitfall in these problems.
- Memorizing the molar masses of the most common 15-20 substances is a 'pro-level' NEET strategy.
NEET Exam Angle
- In Titration problems, the molar mass is used to convert the calculated moles back into mass for the final result.
- 80% of Physical Chemistry questions require a quick molar mass calculation as step one.
- Don't just learn the theory; solve at least 50 varied molar mass problems to build 'muscle memory'.
Recommended Reading
Explore related Biology topics to build deeper chapter connections for NEET.
- Chemical Equations · Topic 1.10
- Dalton's Atomic Theory · Topic 1.2
- Laws of Chemical Combination · Topic 1.4
- Mole Concept · Topic 1.6
- Nature of Electromagnetic Radiation · Topic 2.1
- Photoelectric Effect · Topic 2.2
- Jump to Key Terms (Quick Revision)
- Review Common NEET Mistakes
- Read Topic FAQs
- Check PYQ Pattern Notes
- Practice NEET MCQs
- Solve NEET PYQs
📚 Key Terms
⚠️ Common NEET Mistakes
- 1Forgetting to multiply the atomic mass by the subscript in a molecular formula.
- 2Using the atomic number instead of the atomic mass during calculations.
- 3Ignoring the units and failing to convert kg to g before using the molar mass formula.
- 4Failing to account for water of crystallization in hydrated compounds (e.g., ignoring the ·5H2O).
- 5Confusing the molar mass of an element with the molar mass of its diatomic form (e.g., using 16 instead of 32 for O2).
📝 NEET PYQ Pattern
In NEET 2018–2024, molar mass calculations rarely appear as standalone questions but are critical prerequisites for Molarity (Solutions) and Empirical Formula problems. Analysis shows that 80% of Physical Chemistry questions in NEET require a quick and accurate molar mass calculation as the first step.
❓ Frequently Asked Questions
What is the difference between atomic mass (u) and molar mass (g/mol)?
Atomic mass (u) is the mass of a single atom of an element. Molar mass (g/mol) is the mass of one mole ($6.022 \times 10^{23}$) of those atoms. Numerically, they are the same, but they represent different scales (microscopic vs. macroscopic).
Why is Carbon-12 used as the reference standard for molar mass?
Carbon-12 was chosen for its stability, abundance, and the fact that it allows the atomic mass scale and the gram scale to be linked precisely, making 1 mole of $^{12}C$ exactly 12 grams.
How do I calculate the molar mass of a hydrated salt like CuSO4·5H2O?
You calculate the molar mass of $CuSO_4$ and then add the molar mass of five water molecules ($5 \times 18 = 90$ g/mol) to the total sum.
Does the molar mass of a substance change with temperature or pressure?
No, molar mass is an intrinsic property of a substance based on its atomic composition. It does not change with temperature or pressure, unlike volume or density.
Can molar mass be expressed in units other than grams per mole?
The SI unit is actually kg/mol, but g/mol is the standard convention in almost all chemical calculations and NEET exam problems.
What is the relationship between molar mass and the number of molecules in a sample?
Molar mass allows you to find the number of moles ($n = m/M$). Once you have moles, you multiply by Avogadro's number ($N_A$) to find the total number of molecules.
Written By
NEET Content Strategist & Biology Expert
Sangita Kumari is a NEET educator and content strategist with over 6 years of experience teaching Biology, Chemistry, and Physics to Class 11 and 12 aspirants. She helps bridge the gap between traditional NCERT preparation and modern AI-powered learning. Her content is trusted by thousands of NEET aspirants across India.