Adiabatic Process
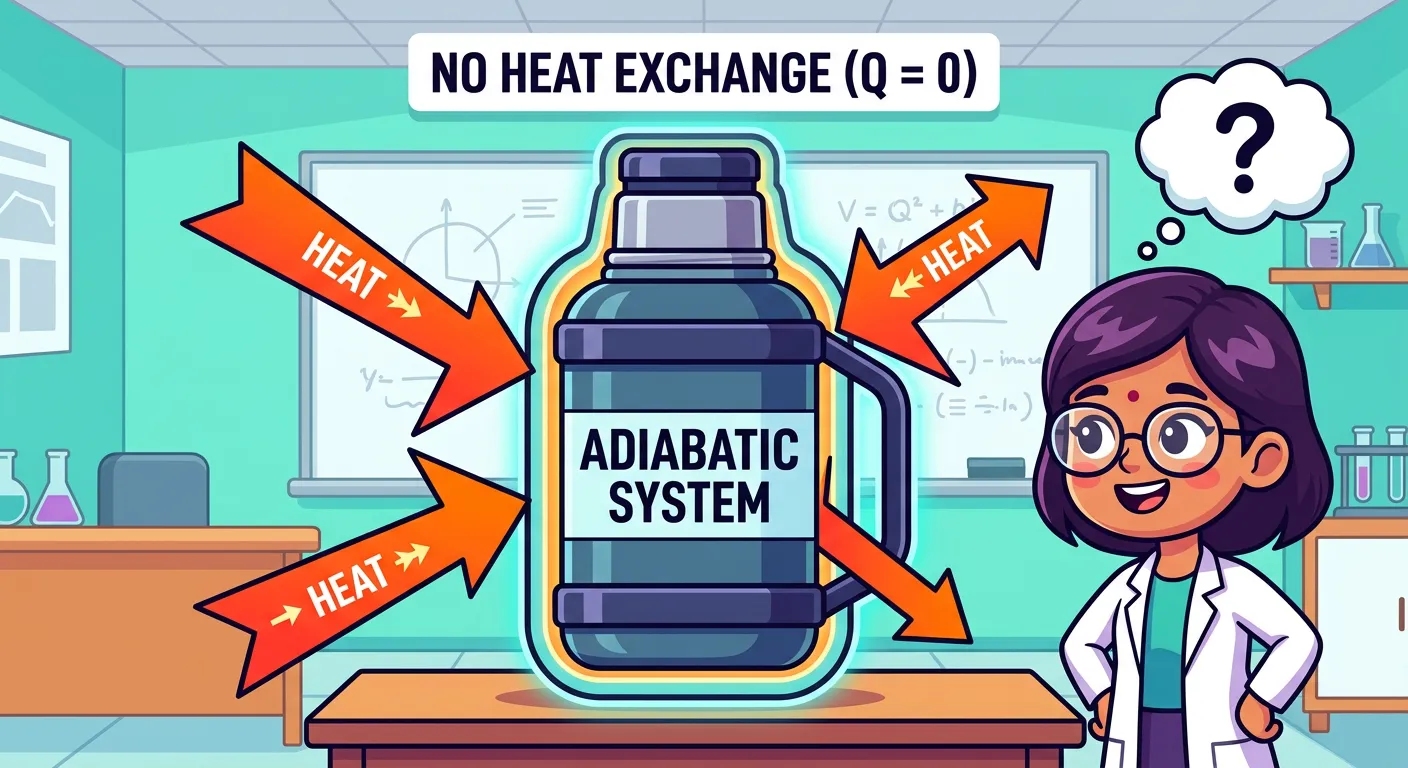
Hello future doctors! Today we're diving into the 'Adiabatic Process.' Imagine a perfect thermos flask where no heat can enter or escape. When a gas undergoes a change in such an isolated system, we call it an Adiabatic Process. The rule is simple: Heat exchange, Q, is always zero!
1
7